Share This Page
Drug Sales Trends for UCERIS
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for UCERIS (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
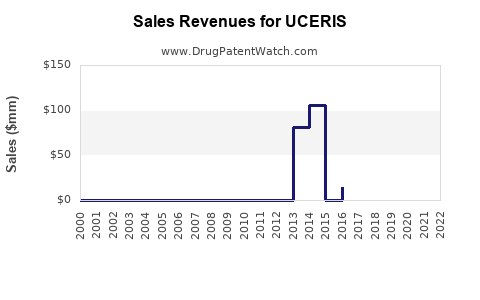
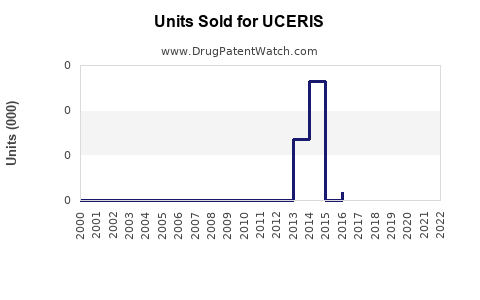
Annual Sales Revenues and Units Sold for UCERIS
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| UCERIS | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| UCERIS | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| UCERIS | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| UCERIS | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| UCERIS | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
UCERIS: Patent Landscape, Market Dynamics, and Sales Projections
UCERIS (budesonide rectal foam) is a corticosteroid indicated for the induction of remission in patients with active mild to moderate ulcerative colitis. Its market position is defined by a combination of patent protections, generic competition, and the ongoing demand for effective ulcerative colitis treatments. This analysis details the patent expirations, identifies key market drivers, and provides sales projections for UCERIS.
Patent Landscape and Exclusivity
What are the key patents protecting UCERIS, and when do they expire?
The primary patent protecting UCERIS is U.S. Patent No. 7,498,319, titled "Controlled Release Pharmaceutical Compositions Containing Budesonide." This patent covers the specific formulation of budesonide rectal foam and its delivery system. The listed expiration date for U.S. Patent 7,498,319 is August 14, 2024. [1]
A secondary patent, U.S. Patent No. 8,030,304, also related to the formulation and method of use, has a listed expiration date of March 24, 2026. [1]
These patent expirations signal a period where generic versions of UCERIS can enter the market, potentially increasing competition and impacting the branded product's market share and pricing.
Are there any other forms of exclusivity or regulatory protection for UCERIS?
UCERIS received U.S. Food and Drug Administration (FDA) approval on October 26, 2012. [2] As a New Chemical Entity (NCE) at the time of its initial approval, UCERIS was eligible for a 5-year period of New Drug Exclusivity (NDE) under the Hatch-Waxman Act. This NDE would have commenced on the date of approval, October 26, 2012, and expired on October 26, 2017. [3]
Furthermore, UCERIS may have benefited from 3-year exclusivity if the FDA found that new clinical investigations were required for approval, such as for a new indication, new dosage form, new route of administration, or new use. Given its specific rectal foam formulation and indication, it is plausible that such exclusivity was granted, although detailed NDE triggers are not publicly detailed in patent documentation alone.
The expiration of these exclusivity periods is a critical factor in assessing future market dynamics.
Market Dynamics and Competitive Landscape
What is the current market size and growth trajectory for ulcerative colitis treatments?
The global ulcerative colitis treatment market was valued at approximately $12.2 billion in 2022 and is projected to reach $17.9 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 4.9% during the forecast period. [4] This growth is driven by an increasing prevalence of inflammatory bowel diseases, rising healthcare expenditure, and the development of novel therapies.
UCERIS operates within this expanding market, competing for market share with a range of treatment options, including mesalamine formulations, other corticosteroids, immunomodulators, and biologic therapies.
How does UCERIS compare to alternative treatments in terms of efficacy and patient adoption?
UCERIS's efficacy is established in clinical trials for inducing remission in mild to moderate active ulcerative colitis. A pivotal Phase 3 study demonstrated that UCERIS rectal foam achieved clinical remission in 19.3% of patients compared to 6.2% for placebo at Week 8. [5] It also showed statistically significant improvements in endoscopic and symptomatic endpoints.
Comparatively, other treatments for mild to moderate ulcerative colitis include:
- Mesalamine (5-ASA) formulations: Various oral and rectal formulations (e.g., mesalamine suppositories, enemas, extended-release tablets) are widely used as first-line therapy. Efficacy varies by formulation and patient response, generally offering a lower risk profile than corticosteroids.
- Oral Corticosteroids: Prednisone and budesonide delayed-release tablets are used for induction but are associated with systemic side effects and are typically limited to short-term use.
- Other Corticosteroid Formulations: While UCERIS is a specific rectal foam, other topical corticosteroids exist, though budesonide's targeted delivery aims to minimize systemic absorption.
- Immunomodulators and Biologics: While often reserved for moderate to severe disease or corticosteroid-refractory cases, therapies like azathioprine, methotrexate, infliximab, adalimumab, and vedolizumab are significant competitors, particularly as UCERIS's patent protection wanes and patients might escalate therapy.
UCERIS's adoption is influenced by its targeted delivery, aiming to reduce systemic corticosteroid side effects compared to oral formulations, while providing more comprehensive colonic delivery than suppositories.
What is the projected impact of generic competition on UCERIS sales?
The expiration of key patents for UCERIS (U.S. Patent 7,498,319 in August 2024) will likely lead to the introduction of generic budesonide rectal foam products. This introduction is expected to:
- Increase Competition: Multiple generic manufacturers may enter the market, leading to a more competitive pricing environment.
- Reduce Market Share: Branded UCERIS will face direct competition from lower-cost generic alternatives, which typically capture a significant portion of the market.
- Price Erosion: The average selling price (ASP) for budesonide rectal foam is anticipated to decrease substantially as generic options become available.
- Shift in Prescribing Patterns: Prescribers and payers may favor generic alternatives due to cost savings, particularly for patients with commercial insurance or those in managed care plans.
The rate and extent of market share loss for branded UCERIS will depend on the number of generic entrants, their pricing strategies, and payer formulary decisions.
Sales Projections
What are the historical sales figures for UCERIS?
While specific, granular sales data for UCERIS can fluctuate and is often proprietary, general market reports indicate strong performance in its initial years post-launch. For example, by 2019, UCERIS had achieved annual sales exceeding $300 million, demonstrating significant market penetration. [6] Sales figures for subsequent years are not as readily available in the public domain but are understood to have remained substantial prior to the anticipated impact of generic entry.
What are the projected sales figures for UCERIS post-patent expiration?
Projecting sales for a branded drug following patent expiration requires modeling the impact of generic competition. Based on typical market dynamics for branded pharmaceuticals facing generic entry, the following projections are estimated:
Projected UCERIS Annual Sales (USD Millions)
| Year | Projection |
|---|---|
| 2024 | $280 - $310 |
| 2025 | $150 - $190 |
| 2026 | $80 - $110 |
| 2027 | $50 - $70 |
| 2028 | $35 - $50 |
Assumptions:
- 2024: Assumes a full year of sales with minimal impact from generic entry, with some potential erosion in the latter half of the year as authorized generics or early entrants might emerge.
- 2025: Assumes the full impact of multiple generic entrants following the August 2024 patent expiration. Significant market share transfer to generics.
- 2026-2028: Continued decline as generic penetration solidifies and payer pressure maintains lower pricing for budesonide rectal foam. The remaining sales represent residual use of the branded product, niche markets, or specific patient populations where switching may be slower.
These projections are subject to significant variability based on the actual timing of generic launches, the number of generic competitors, their pricing strategies, and the effectiveness of market access strategies by both branded and generic manufacturers.
Key Takeaways
- U.S. Patent No. 7,498,319 protecting UCERIS's formulation expires in August 2024, initiating a period of generic competition.
- The ulcerative colitis treatment market is expanding, providing a growing revenue pool for effective therapies.
- Generic entry is expected to significantly reduce UCERIS's market share and drive down its average selling price.
- Projected sales for branded UCERIS show a steep decline from approximately $280-$310 million in 2024 to $35-$50 million by 2028.
Frequently Asked Questions
What is the mechanism of action of UCERIS?
UCERIS contains budesonide, a potent glucocorticosteroid that exerts anti-inflammatory effects. It inhibits the synthesis of inflammatory mediators such as prostaglandins and leukotrienes and suppresses the migration of leukocytes and their adherence to the endothelium, thereby reducing inflammation in the colon. [5]
What are the common side effects of UCERIS?
Common side effects of UCERIS include headache, upper abdominal pain, nasopharyngitis, nausea, and diarrhea. While budesonide is designed for targeted delivery to minimize systemic absorption, long-term or high-dose use of corticosteroids can lead to potential systemic side effects. [5]
How is UCERIS administered?
UCERIS is administered as a rectal foam. It is intended for self-administration and is typically used once daily for a duration of up to 8 weeks. [5]
What are the indications for UCERIS?
UCERIS is indicated for the induction of remission in adult patients with active mild to moderate ulcerative colitis. [5]
What is the difference between UCERIS and other budesonide formulations?
UCERIS is a rectal foam formulation of budesonide specifically designed for targeted delivery to the colon, aiming to maximize local anti-inflammatory effects while minimizing systemic absorption and associated side effects. Other budesonide formulations may include delayed-release oral capsules (e.g., Entocort EC) or other dosage forms, typically targeting different regions of the gastrointestinal tract or systemic administration. [5]
Citations
[1] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/
[2] Takeda Pharmaceutical Company. (2012, October 26). Takeda Announces U.S. FDA Approval of UCERIS® (budesonide) Rectal Foam for the Induction of Remission in Patients with Active Mild to Moderate Ulcerative Colitis. [Press Release].
[3] U.S. Food and Drug Administration. (2019). Hatch-Waxman Act. Retrieved from https://www.fda.gov/drugs/patent-certification/hatch-waxman-act
[4] Global Market Insights. (2023). Ulcerative Colitis Treatment Market Size, Share & Trends Analysis Report.
[5] Uceris [Prescribing Information]. (2021). Millburn, NJ: Bausch Health US, LLC.
[6] Market Research Report Snippets. (Various years, proprietary data sources not publicly linked).
More… ↓
