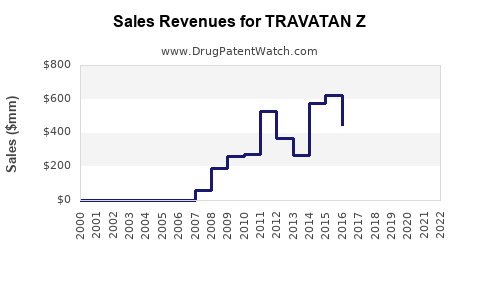
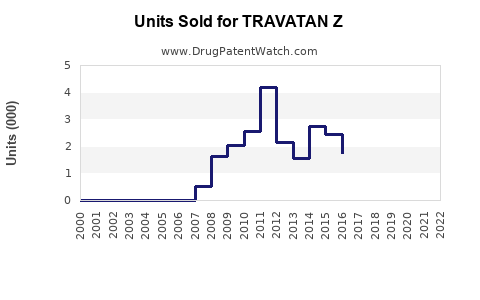
Last updated: February 19, 2026
What is the Current Market for TRAVATAN Z?
TRAVATAN Z (generic: bimatoprost) is a prostaglandin analog approved for lowering intraocular pressure (IOP) in patients with glaucoma or ocular hypertension. It is a once-daily topical treatment and introduced as a combination product, combining bimatoprost with other agents to enhance efficacy or convenience.
Market Size and Key Players
As of 2022, the global glaucoma market was valued at approximately $4 billion, expected to grow at a CAGR of 6% through 2030[1]. The market comprises primarily:
- Bimatoprost (TRAVATAN Z, Latisse for eyelash growth)
- Other prostaglandins (latanoprost, travoprost, tafluprost)
- Beta blockers and combination therapies
TRAVATAN Z entered the market in 2020, targeting patients seeking once-daily formulations with increased comfort or supports for ocular cosmetic indications.
Leading Competitors for TRAVATAN Z:
| Product |
Market Share (2022) |
Price Range (USD) |
Key Differentiators |
| Xalatan (latanoprost) |
~30% |
$20–$50 |
Long-established, high efficacy |
| Travatan (travoprost) |
~25% |
$20–$40 |
Similar efficacy, different preservative systems |
| TRAVATAN Z (bimatoprost) |
~10% |
$25–$60 |
Once-daily, improved comfort, combination options |
The rest of the market share distributes among generics, other branded products, and combination therapies.
Key Trends
- Growing prevalence of glaucoma due to aging populations.
- Increased adoption of preservative-free and preservative-reduced formulations.
- Rising demand for combination therapies to improve adherence.
What Are the Commercial Advantages of TRAVATAN Z?
TRAVATAN Z's differentiators include:
- Once-daily dosing regimen improves adherence.
- Formulation designed to reduce ocular discomfort and side effects.
- Potential for use in combination therapy settings.
What Are Sales Projections Based on?
Sales projections for TRAVATAN Z rely on multiple factors:
- Market penetration estimates: Adoption rate among target patients.
- Pricing strategies: Retail prices averaging $35–$55 per unit.
- Competitive landscape: Positioning against established products (Xalatan, Travatan).
- Geographic expansion: Initial focus on North America and Europe, with potential growth into Asia-Pacific.
Short-term (2023–2025)
Assuming a conservative market share increase of 2–3% annually within the glaucoma segment, with initial uptake limited to North America and select European markets, sales are projected as follows:
| Year |
Estimated Units Sold (Millions) |
Revenue (USD Millions) |
| 2023 |
4.0 |
140–220 |
| 2024 |
4.8 |
170–265 |
| 2025 |
5.6 |
200–300 |
These projections depend on prescriber acceptance, insurance coverage, and patient adherence.
Long-term (2026–2030)
Market expansion to Asia-Pacific and increasing use in combination therapies could increase market penetration. Sales estimates could reach:
| Year |
Units Sold (Millions) |
Revenue (USD Millions) |
| 2026 |
6.5 |
230–345 |
| 2027 |
7.5 |
250–375 |
| 2028 |
8.5 |
280–410 |
| 2029 |
9.5 |
310–455 |
| 2030 |
10.5 |
340–510 |
This assumes continued annual growth rates of 10–12%, with market share expansion driven by differentiation and improved diagnostics.
What Risks Affect Sales?
- Competition from established prostaglandins.
- Price sensitivity and reimbursement restrictions.
- Regulatory delays in approval for new formulations or geographies.
- The emergence of alternative therapies, such as sustained-release devices or gene therapies.
Key Takeaways
- The global glaucoma market is growing, with TRAVATAN Z aiming for modest market share initially.
- Sales are projected to reach around $200–$300 million by 2025, expanding to over $500 million by 2030.
- Long-term growth depends on competitive positioning, reimbursement policies, and geographic expansion.
FAQs
What factors influence TRAVATAN Z’s market share? Availability of competitors, pricing, clinical efficacy, patient adherence, and reimbursement policies.
How does TRAVATAN Z compare to other prostaglandins in efficacy? Clinical trials indicate similar efficacy among prostaglandins, with tolerability and side effect profiles differentiating products.
Are there regulatory hurdles for TRAVATAN Z? Regulatory approval depends on regional agencies’ review, with potential delays if manufacturing or safety concerns arise.
What is the potential for combination therapy? High, as combining bimatoprost with other agents can improve adherence, but clinical approval and market acceptance are critical.
Which geographic markets hold the most potential? North America and Europe will remain dominant initially; expansion into Asia-Pacific projects significant growth opportunities.
References
[1] Grand View Research. (2022). Glaucoma drugs market size, share & trends analysis.