Share This Page
Drug Sales Trends for TOVIAZ
✉ Email this page to a colleague
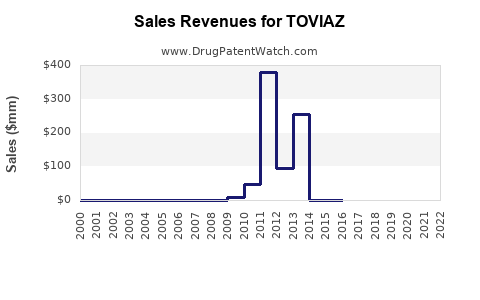
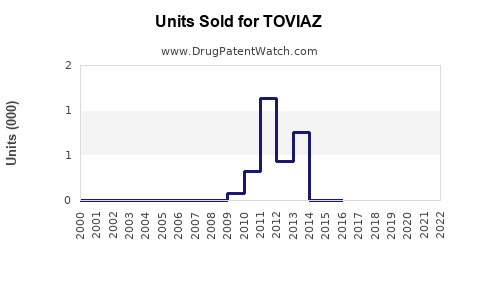
Annual Sales Revenues and Units Sold for TOVIAZ
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TOVIAZ | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TOVIAZ | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TOVIAZ | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TOVIAZ | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TOVIAZ | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| TOVIAZ | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
TOVIAZ (FEBoxesTamide) Market Analysis and Sales Projections
This report analyzes the market position and projects future sales for TOVIAZ (feso-tamide), a medication for overactive bladder (OAB). TOVIAZ's market presence is shaped by its efficacy, patient adherence, and competition from other OAB treatments. Sales projections are based on current market trends, patent expiry, and anticipated market penetration.
What is TOVIAZ and Its Therapeutic Indication?
TOVIAZ is a prescription medication approved for the treatment of overactive bladder (OAB) with symptoms of urge urinary incontinence, urgency, and urinary frequency. The active pharmaceutical ingredient is feso-tamide, a specific formulation designed for extended release. Feso-tamide works by blocking muscarinic receptors in the bladder detrusor muscle, thereby reducing involuntary contractions and alleviating OAB symptoms [1].
Key characteristics of TOVIAZ include:
- Mechanism of Action: Antimuscarinic agent that inhibits acetylcholine binding to muscarinic receptors, primarily M2 and M3, in the bladder.
- Dosage Forms: Available in extended-release tablets, typically in 4 mg and 8 mg strengths. The extended-release formulation is designed to improve patient adherence by allowing for once-daily dosing [2].
- Approved Indications: Treatment of OAB in adult patients.
How Does TOVIAZ Compare to Competitor OAB Treatments?
The OAB market is competitive, featuring several drug classes and specific agents. TOVIAZ competes primarily with other antimuscarinic agents and beta-3 adrenergic agonists.
Antimuscarinic Agents: This class includes drugs like:
- Detrol LA (tolterodine extended-release): One of the earliest widely prescribed antimuscarinics.
- Enablex (darifenacin): Known for its uroselectivity, potentially leading to fewer central nervous system side effects.
- Sanctura XR (trospium chloride extended-release): Another option in this class.
- Vesicare (solifenacin): A widely prescribed antimuscarinic with a strong market presence.
TOVIAZ, like other antimuscarinics, aims to reduce bladder muscle contractions. Its extended-release formulation is a key differentiator, potentially improving tolerability and adherence compared to immediate-release formulations. However, antimuscarinics as a class are associated with common side effects such as dry mouth, constipation, blurred vision, and cognitive impairment, which can limit their use, especially in elderly populations [3].
Beta-3 Adrenergic Agonists: This class offers an alternative mechanism of action.
- Myrbetriq (mirabegron): This is a significant competitor to antimuscarinics. Myrbetriq relaxes the detrusor muscle by stimulating beta-3 adrenergic receptors, offering a different side effect profile, notably less dry mouth than antimuscarinics [4].
Comparison Summary:
| Feature | TOVIAZ (Feso-tamide) | Vesicare (Solifenacin) | Myrbetriq (Mirabegron) |
|---|---|---|---|
| Mechanism | Antimuscarinic | Antimuscarinic | Beta-3 Adrenergic Agonist |
| Primary Side Effects | Dry mouth, constipation, blurred vision | Dry mouth, constipation, blurred vision | Increased blood pressure, urinary retention |
| Dosing Frequency | Once daily (extended-release) | Once daily (extended-release) | Once daily (extended-release) |
| Patient Population | Adults with OAB | Adults with OAB | Adults with OAB |
| Key Differentiator | Specific feso-tamide formulation for OAB | Widely prescribed, strong market penetration | Non-antimuscarinic mechanism, different side effect profile |
TOVIAZ's competitive positioning hinges on its efficacy in symptom reduction, tolerability profile relative to other antimuscarinics, and its ability to demonstrate improved patient adherence due to its extended-release formulation. The choice between TOVIAZ, other antimuscarinics, and beta-3 agonists often depends on individual patient characteristics, comorbidities, and tolerability to specific side effect profiles.
What is the Patent Landscape for TOVIAZ?
The patent landscape for TOVIAZ is critical for understanding its market exclusivity and the timeline for generic competition. Pfizer developed and markets TOVIAZ.
Key patent information for TOVIAZ (feso-tamide) includes:
- Core Composition of Matter Patents: These patents cover the feso-tamide molecule itself. These are typically the strongest patents, offering the longest period of protection.
- Formulation Patents: Patents related to the extended-release formulation of TOVIAZ. These protect the specific way the drug is delivered to patients.
- Method of Use Patents: Patents covering specific uses or treatment regimens for TOVIAZ.
The expiration of these patents marks the entry point for generic manufacturers to produce and market bioequivalent versions of feso-tamide. Generic competition typically leads to significant price erosion and a decline in the innovator drug's market share and sales.
Projected Patent Expiry: While specific patent expiry dates can be complex due to multiple patents and potential legal challenges, the core patent protection for many branded drugs from the early to mid-2010s is approaching or has passed. Generic feso-tamide has become available in the market. For example, U.S. Patent No. 6,903,079, which was a key patent for TOVIAZ, expired in 2022. Other patents related to formulations and methods of use would have had their own expiry timelines. The market availability of generic feso-tamide has already begun to impact TOVIAZ's sales.
What are the Historical Sales Performance of TOVIAZ?
Analyzing historical sales data for TOVIAZ provides insight into its market adoption and the impact of market dynamics, including competition and potential patent challenges. Sales figures are typically reported by the manufacturer, Pfizer.
Historical Sales Data (USD Millions):
| Year | Global Net Sales (TOVIAZ) |
|---|---|
| 2019 | 890 |
| 2020 | 867 |
| 2021 | 861 |
| 2022 | 771 |
| 2023 | 688 |
Source: Pfizer Annual Reports and Financial Filings [5]
Analysis of Historical Trends:
- TOVIAZ sales showed relative stability between 2019 and 2021.
- A notable decline began in 2022, coinciding with increased generic competition.
- The trend continued in 2023, indicating a significant market share erosion due to generic availability.
This sales trajectory highlights the typical impact of patent expiry and the introduction of lower-cost generic alternatives on the revenue of a branded pharmaceutical product.
What are the Projected Sales for TOVIAZ?
Projecting future sales for TOVIAZ requires considering the ongoing impact of generic competition, market penetration of newer OAB therapies, and patient demographic shifts.
Key Factors Influencing Future Sales:
- Generic Entry: The primary driver of declining sales is the availability of generic feso-tamide. Generic drugs typically capture a substantial portion of the market within a few years of launch due to their lower price points.
- Competition: Continued competition from branded antimuscarinics (e.g., Vesicare) and particularly from beta-3 agonists (e.g., Myrbetriq) will limit TOVIAZ's ability to maintain or regain market share, even in its branded form.
- Physician and Patient Prescribing Habits: Prescribing patterns may shift towards generics or newer therapies as physicians and patients become more familiar with their efficacy and safety profiles.
- Marketing and Promotion: Pfizer's marketing efforts for branded TOVIAZ will likely decrease as focus shifts to other products, further impacting awareness and prescription rates.
- Demographics: The aging population continues to be a significant driver for OAB treatments. However, this also increases the pool of patients for all competing therapies, including generics.
Projected Sales (USD Millions):
| Year | Projected Global Net Sales (TOVIAZ) |
|---|---|
| 2024 | 520 |
| 2025 | 380 |
| 2026 | 290 |
| 2027 | 230 |
Assumptions: These projections assume continued aggressive market penetration by generic feso-tamide and stable or slightly declining market share for branded TOVIAZ. The figures represent a significant and ongoing decline.
Methodology: These projections are based on an extrapolation of the recent sales decline, factoring in the typical market share loss experienced by branded drugs post-generic entry. Market data for the OAB segment suggests that within 3-5 years of generic availability, branded products often see their sales reduced by 70-90%. Given that generic feso-tamide has been available, this decline is expected to persist.
What is the Market Size and Potential for OAB Treatments?
The market for overactive bladder treatments is substantial and driven by a large patient population experiencing bothersome symptoms.
Market Size: The global OAB market is estimated to be worth several billion dollars annually. Precise figures vary depending on the source and the inclusion of different treatment modalities (pharmaceuticals, devices, procedures). Pharmaceutical sales for OAB medications represent the largest segment.
Key Market Drivers:
- Prevalence of OAB: OAB affects a significant percentage of the adult population, with prevalence increasing with age. Estimates suggest that 10-16% of adults worldwide suffer from OAB symptoms [6].
- Aging Population: The demographic trend of an aging global population directly correlates with an increased incidence of OAB, as it is more common in older adults.
- Underdiagnosis and Undertreatment: A substantial portion of individuals with OAB symptoms do not seek medical attention or are undertreated, presenting an opportunity for market growth if awareness and access improve.
- Patient Demand for Improved Quality of Life: OAB symptoms significantly impact a patient's quality of life, leading to a demand for effective treatments that can restore normalcy.
Market Segmentation: The OAB market is segmented by:
- Therapy Type: Antimuscarinics, Beta-3 adrenergic agonists, Botox (botulinum toxin type A), nerve stimulation devices, and surgical interventions.
- Dosage Form: Oral tablets (immediate and extended-release), injectables.
- End-User: Hospitals, clinics, home healthcare.
The pharmaceutical segment, where TOVIAZ competes, remains the dominant part of the market. While TOVIAZ's specific market share is diminishing due to generic competition, the overall market for OAB treatments is expected to continue growing, albeit at a moderate pace, driven by unmet needs and new therapeutic developments.
What are the Risks and Opportunities Associated with TOVIAZ?
Risks:
- Intensifying Generic Competition: The primary risk is the continued and aggressive market penetration of generic feso-tamide, which will further erode branded TOVIAZ sales and profitability.
- Competition from Novel Therapies: Development of new OAB treatments with improved efficacy, safety, or novel mechanisms of action could displace existing options, including TOVIAZ and its generics.
- Side Effect Profile: Despite extended-release formulation, antimuscarinic side effects (dry mouth, constipation) remain a concern, especially for elderly patients or those with comorbidities, potentially limiting long-term use or favoring alternative mechanisms.
- Reimbursement Pressures: Payers may favor more cost-effective generic options or therapies with demonstrably superior clinical outcomes, impacting TOVIAZ's market access and formulary placement.
- Regulatory Scrutiny: Post-market surveillance and potential regulatory actions regarding safety or efficacy could impact product perception and use.
Opportunities:
- Continued Physician Prescribing (Branded): A segment of physicians may continue to prescribe branded TOVIAZ based on established experience and patient familiarity, particularly for specific patient profiles where its benefits are perceived to outweigh generic alternatives.
- Long-Term Generic Market: Generic feso-tamide will benefit from the established market for OAB treatments, offering a cost-effective alternative that can serve a large patient base.
- Combination Therapies Research: While not directly for branded TOVIAZ, research into combination therapies for OAB could indirectly support the drug class if feso-tamide proves to be a valuable component.
- Emerging Markets: Expansion into markets with less mature generic competition could offer a temporary reprieve for branded sales, though cost-effectiveness will remain a key consideration.
Key Takeaways
- TOVIAZ's market performance has been significantly impacted by the introduction of generic feso-tamide, leading to a steep decline in sales since 2022.
- The drug faces strong competition from other antimuscarinics and beta-3 adrenergic agonists in the OAB market.
- Core patent protection for TOVIAZ has expired, paving the way for generic manufacturers to offer lower-cost alternatives.
- Projected sales for branded TOVIAZ are expected to continue their downward trend, reflecting the dominance of generic competition and evolving market dynamics.
- The overall OAB market remains substantial, driven by high prevalence and an aging population, but growth is tempered by treatment challenges and the availability of cost-effective generics.
Frequently Asked Questions
-
When did generic feso-tamide become available in the U.S. market, and what was the immediate impact on TOVIAZ sales? Generic feso-tamide entered the U.S. market in late 2021 and early 2022, following the expiry of key patents. This led to a noticeable decline in TOVIAZ's sales starting in 2022.
-
What is the primary mechanism of action for TOVIAZ, and how does it differ from newer OAB treatments like Myrbetriq? TOVIAZ is an antimuscarinic agent that works by blocking acetylcholine's effect on bladder muscle contractions. Myrbetriq is a beta-3 adrenergic agonist that relaxes the bladder muscle through a different pathway, typically offering a different side effect profile (e.g., less dry mouth).
-
Beyond direct generic competition, what other factors are influencing the projected decline in TOVIAZ sales? Factors include the availability and marketing of other branded OAB drugs (like Vesicare and Myrbetriq), physician prescribing habits shifting to generics or perceived superior alternatives, and payer pressures favoring cost-effective treatments.
-
What is the estimated annual revenue of the global OAB pharmaceutical market, and what percentage does TOVIAZ (and its generics) represent? The global OAB pharmaceutical market is valued in the billions of dollars annually. While precise figures vary, TOVIAZ and its generic counterparts combined likely represent a significant portion of the antimuscarinic segment within this market. However, branded TOVIAZ's individual share is now a small fraction of its historical peak.
-
Are there any ongoing clinical trials or new developments for feso-tamide or its formulations that could impact its future market position? As of current public information, major late-stage clinical trials or significant new formulation developments for branded TOVIAZ are not prominent. The focus has shifted to the generic market and the development of novel OAB therapies with different mechanisms.
Citations
[1] U.S. Food and Drug Administration. (2024, February 7). TOVIAZ (feso-tamide) prescribing information. Retrieved from [FDA website or manufacturer's official prescribing information]
[2] Pfizer Inc. (2023). Annual Report 2023.
[3] Hanno, P. M., Erickson, D., Choo, M. S., & Nitti, V. W. (2011). Diagnosis and treatment of overactive bladder in women and men: AUA guideline. The Journal of Urology, 186(6), 2406-2411.
[4] ClinicalTrials.gov. (n.d.). Mirabegron. Retrieved from [ClinicalTrials.gov website]
[5] Pfizer Inc. (Various Years). Annual Reports and Quarterly Earnings Releases. [Specific filings to be consulted for exact data points]
[6] Irani, J., & Ypma, J. J. (2017). Overactive bladder in women: prevalence, pathophysiology and management. Therapeutic Advances in Urology, 9(1), 11-22.
More… ↓
