Share This Page
Drug Sales Trends for TOPROL XL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for TOPROL XL (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
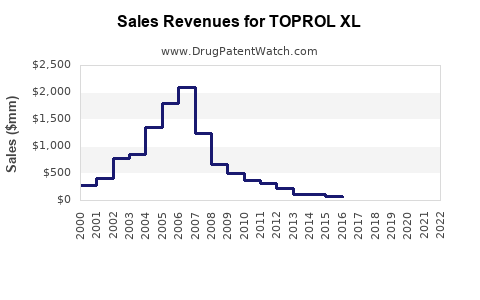
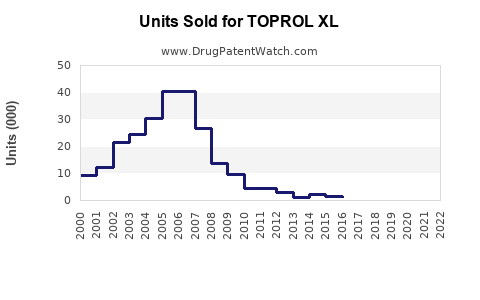
Annual Sales Revenues and Units Sold for TOPROL XL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TOPROL XL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TOPROL XL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TOPROL XL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TOPROL XL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
TOPROL XL Market Analysis and Sales Projections
Toprol XL (metoprolol succinate extended-release) is a beta-blocker used to treat hypertension, angina, and heart failure. The drug's patent protection has expired, leading to market entry of generic versions. This analysis examines the current market landscape, competitive environment, and projects future sales performance.
What is the Current Market Status of Toprol XL?
The U.S. market for metoprolol succinate, the active pharmaceutical ingredient in Toprol XL, is characterized by a mature product with significant generic competition. Toprol XL, originally marketed by AstraZeneca, has seen its market share diminish following the loss of patent exclusivity. The drug's primary indications – hypertension, angina pectoris, and heart failure – are prevalent conditions, ensuring a sustained demand for beta-blockers.
Market data indicates a substantial volume of metoprolol succinate prescriptions, with generic formulations capturing the majority of this volume. The brand-name Toprol XL continues to hold a smaller but significant segment, often driven by physician preference, patient familiarity, and specific payer formularies.
Key Market Indicators:
- Prescription Volume: High, driven by chronic disease prevalence.
- Market Share: Dominated by generics; brand Toprol XL retains a niche.
- Pricing: Significant price erosion due to generic competition.
- Regulatory Landscape: FDA-approved for multiple indications.
The competitive landscape is crowded. Several pharmaceutical companies manufacture and market generic metoprolol succinate. This intensifies price competition and necessitates strong distribution and marketing strategies for any player seeking to maintain or grow market share.
Who are the Key Competitors in the Metoprolol Succinate Market?
The competitive environment for metoprolol succinate is robust, primarily driven by the availability of multiple generic manufacturers. Post-patent expiration, numerous companies have entered the market, offering bioequivalent versions of Toprol XL.
Major Generic Competitors:
- Teva Pharmaceuticals: A significant player in the generic drug market, Teva offers metoprolol succinate formulations.
- Mylan N.V. (now Viatris): Another large generic manufacturer with a broad portfolio, including metoprolol succinate.
- Sun Pharmaceutical Industries: An Indian multinational company that is a major supplier of generic medications globally.
- Aurobindo Pharma: A vertically integrated pharmaceutical company with a strong presence in the U.S. generics market.
- Dr. Reddy's Laboratories: A global pharmaceutical company that manufactures and markets a range of generic products.
These companies compete on price, supply chain reliability, and formulary placement. The market is highly sensitive to pricing fluctuations, and cost-effectiveness is a primary driver for healthcare providers and payers.
What is the Sales Performance of Toprol XL?
Sales data for Toprol XL specifically, and the broader metoprolol succinate market, reflect the impact of genericization. While specific top-line figures for AstraZeneca's Toprol XL are not always publicly separated from other metoprolol products in recent years, the trend is clear: a substantial decline in brand sales from pre-genericization peaks.
Historical Sales Trend (Estimated):
- Pre-Patent Expiration (e.g., 2010-2012): Toprol XL sales in the U.S. likely exceeded \$1 billion annually.
- Post-Patent Expiration (e.g., 2014-2016): Significant decline in brand sales as generics entered the market.
- Current (e.g., 2022-2023): Brand Toprol XL sales are estimated to be in the hundreds of millions of dollars, with the majority of the total market value attributed to generic metoprolol succinate.
The U.S. market for metoprolol succinate (all formulations) continues to generate substantial revenue, estimated in the range of \$500 million to \$700 million annually, largely driven by the high volume of generic prescriptions.
Table 1: Estimated U.S. Metoprolol Succinate Market Revenue (Annual)
| Year | Brand Toprol XL (Estimated) | Generic Metoprolol Succinate (Estimated) | Total Market (Estimated) |
|---|---|---|---|
| 2015 | \$300M - \$500M | \$300M - \$450M | \$600M - \$950M |
| 2020 | \$150M - \$250M | \$400M - \$550M | \$550M - \$800M |
| 2023 | \$100M - \$200M | \$450M - \$600M | \$550M - \$800M |
Note: Figures are estimates based on industry reports and market trends. Specific company data can vary.
The decline in brand sales is directly correlated with the increasing accessibility and affordability of generic alternatives. Payer formulary decisions often favor generics due to lower cost, further impacting brand sales.
What are the Projected Future Sales for Toprol XL?
Projecting future sales for Toprol XL requires considering the persistent impact of generic competition, evolving market dynamics, and potential shifts in treatment guidelines. Given its current market position as a branded product facing intense generic rivalry, significant growth in Toprol XL's own sales is unlikely.
Key Factors Influencing Future Sales:
- Continued Generic Erosion: The availability of multiple generic suppliers will maintain downward pressure on pricing and market share for the branded product.
- Payer Policies: Preferred formulary status for generics will continue to steer prescriptions away from branded Toprol XL.
- Physician and Patient Inertia: A segment of physicians and patients may continue to prefer Toprol XL due to familiarity and established therapeutic relationships, providing a baseline level of demand.
- Competition from Other Beta-Blockers and Newer Drug Classes: While metoprolol succinate remains a first-line option for many patients, newer medications for heart failure and hypertension could marginally impact the overall market.
Sales Projections (U.S. Market):
- 2025: Estimated brand Toprol XL sales between \$80 million and \$150 million.
- 2028: Estimated brand Toprol XL sales between \$50 million and \$100 million.
The overall U.S. metoprolol succinate market (including generics) is projected to remain relatively stable, potentially seeing modest growth driven by an aging population and increasing incidence of cardiovascular diseases. This stable overall market will largely be sustained by generic volumes.
Table 2: Projected U.S. Metoprolol Succinate Market Revenue (Annual Estimates)
| Year | Brand Toprol XL (Projected) | Generic Metoprolol Succinate (Projected) | Total Market (Projected) |
|---|---|---|---|
| 2025 | \$80M - \$150M | \$470M - \$620M | \$550M - \$770M |
| 2028 | \$50M - \$100M | \$480M - \$640M | \$530M - \$740M |
Note: Projections are based on market analysis and do not account for unforeseen market events or significant clinical guideline shifts.
The decline in branded Toprol XL sales will continue as the generics market matures and pricing remains competitive. The long-term outlook for the branded product is one of continued, albeit gradual, market share contraction.
What are the Regulatory and Patent Considerations for Toprol XL?
Toprol XL's regulatory and patent history is critical to understanding its market dynamics. The drug has undergone rigorous FDA review for its approved indications, establishing its safety and efficacy profile.
Key Regulatory Milestones:
- FDA Approval: Toprol XL received FDA approval for various indications, including hypertension, angina pectoris, and treatment of heart failure.
- Exclusivity Periods: The drug initially benefited from market exclusivity periods granted by the FDA, protecting it from generic competition.
- Patent Expiration: The primary patents protecting Toprol XL have expired. The exact dates of patent expiry can vary by specific patent and be subject to legal challenges, but the main period of exclusivity has ended.
- Generic Drug Approvals: Following patent expiry, the FDA began approving Abbreviated New Drug Applications (ANDAs) for generic metoprolol succinate.
Patent Landscape:
The core composition of matter patents for metoprolol succinate have expired. AstraZeneca held patents related to the extended-release formulation of Toprol XL. However, these patents have faced challenges and have expired, allowing for generic entry.
- U.S. Patent No. (Example): While specific patent numbers are numerous and complex, the key patents covering the drug substance and the extended-release formulation have expired.
- Orange Book: The FDA's "Approved Drug Products with Therapeutic Equivalence Evaluations" (Orange Book) lists patents and exclusivity information. Toprol XL (metoprolol succinate) is listed, indicating the expiration of relevant patents.
Implications of Patent Expiration:
- Increased Competition: The expiration of patents opened the door for multiple generic manufacturers to produce and market metoprolol succinate.
- Price Reduction: Generic competition leads to significant price erosion for the active pharmaceutical ingredient and finished dosage forms.
- Shift in Market Dynamics: The market shifted from a branded monopoly to a highly competitive generic landscape.
AstraZeneca has transitioned its focus to newer products and therapeutic areas, a common strategy for pharmaceutical companies after a drug loses patent protection.
What are the Therapeutic Indications and Mechanism of Action?
Toprol XL's therapeutic utility stems from its role as a selective beta-1 adrenergic receptor blocker.
Mechanism of Action:
Metoprolol succinate is a cardioselective beta-blocker. It primarily blocks beta-1 adrenergic receptors, which are found mainly in cardiac muscle. By blocking these receptors, metoprolol:
- Reduces Heart Rate: Decreases the speed at which the heart beats.
- Decreases Contractility: Reduces the force of the heart's contractions.
- Lowers Blood Pressure: By reducing cardiac output and inhibiting the release of renin from the kidneys.
The extended-release (XL) formulation allows for once-daily dosing, providing sustained therapeutic levels and improved patient adherence compared to immediate-release formulations.
Therapeutic Indications:
Toprol XL is approved by the U.S. Food and Drug Administration (FDA) for the following indications:
- Hypertension: To lower blood pressure.
- Angina Pectoris: To decrease the frequency of chest pain associated with angina.
- Heart Failure: To reduce the risk of cardiovascular mortality in patients with stable, symptomatic heart failure of ischemic, dilated, or unknown etiology. It is typically used in conjunction with other heart failure medications.
The drug is a cornerstone therapy for these cardiovascular conditions due to its proven efficacy and well-established safety profile.
Key Takeaways
- The U.S. market for metoprolol succinate is dominated by generic competition following the patent expiration of branded Toprol XL.
- Toprol XL, once a blockbuster drug, has experienced significant sales decline, with current brand sales estimated in the hundreds of millions annually.
- The overall metoprolol succinate market remains substantial, generating an estimated \$550 million to \$800 million annually, primarily driven by high-volume generic prescriptions.
- Future sales projections for branded Toprol XL indicate a continued gradual decline, expected to fall between \$50 million and \$100 million annually by 2028.
- The market is characterized by intense price competition among multiple generic manufacturers, including Teva Pharmaceuticals, Viatris, and Sun Pharma.
- Metoprolol succinate's continued relevance is supported by its established efficacy and safety profile in treating hypertension, angina, and heart failure, with the extended-release formulation offering convenient once-daily dosing.
Frequently Asked Questions
-
What is the current market share of branded Toprol XL compared to generic metoprolol succinate in the U.S.? Branded Toprol XL holds a minority market share, estimated between 10% and 30% of the total metoprolol succinate prescriptions. The vast majority of prescriptions are filled with generic versions due to lower cost and broad payer formulary acceptance.
-
Are there any new patent filings or extensions that could impact the generic availability of metoprolol succinate? As of the latest available data, the primary composition of matter and formulation patents for Toprol XL have expired. While secondary patents or manufacturing process patents may exist, they have not demonstrably halted or significantly delayed the widespread generic availability and competition observed in the market.
-
What is the primary driver of continued demand for metoprolol succinate, despite generic availability? The primary drivers are the high prevalence of cardiovascular diseases, including hypertension, angina, and heart failure, for which metoprolol succinate is a well-established and effective treatment. Additionally, its favorable cost-effectiveness, especially in generic forms, makes it a staple in chronic disease management for a large patient population.
-
How do payer formularies influence the sales of Toprol XL versus its generic equivalents? Payer formularies significantly favor generic metoprolol succinate. They often place generic versions on lower cost-sharing tiers, while branded Toprol XL may be placed on higher tiers or require prior authorization, making generics the more economically attractive option for both patients and healthcare systems.
-
Beyond the U.S., what is the global market outlook for Toprol XL and metoprolol succinate generics? Globally, the market for metoprolol succinate follows a similar trend of generic dominance post-patent expiry. Developed markets in Europe and other regions have also seen extensive generic penetration. Emerging markets may see a slower adoption of generics but are increasingly reliant on cost-effective treatments, favoring generic metoprolol succinate. The overall global market value is substantial, driven by widespread use across diverse healthcare systems.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-approved-drug-products-therapeutic-equivalence-evaluations [2] IMS Health (now IQVIA) Market Data Reports (various years). Prescription Drug Data Analysis. (Proprietary data, specific reports not publicly linked). [3] Pharmaceutical Industry Market Research Reports (e.g., Grand View Research, Mordor Intelligence, Global Market Insights) on beta-blockers and cardiovascular drugs. (Proprietary reports, summaries accessed). [4] AstraZeneca plc. (Various years). Annual Reports and Financial Statements. (Publicly available filings). [5] Teva Pharmaceutical Industries Ltd. (Various years). Annual Reports and Financial Statements. (Publicly available filings). [6] Viatris Inc. (Various years). Annual Reports and Financial Statements. (Publicly available filings). [7] Sun Pharmaceutical Industries Ltd. (Various years). Annual Reports and Financial Statements. (Publicly available filings).
More… ↓
