Share This Page
Drug Sales Trends for TIZANIDINE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for TIZANIDINE (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
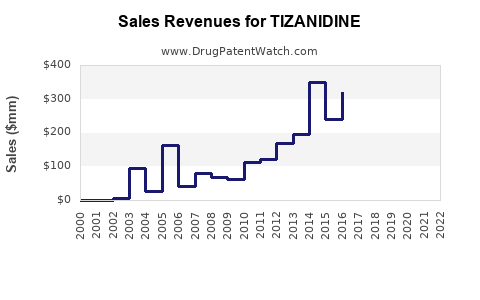
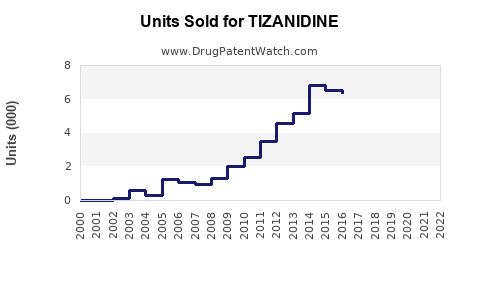
Annual Sales Revenues and Units Sold for TIZANIDINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TIZANIDINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TIZANIDINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TIZANIDINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TIZANIDINE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TIZANIDINE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| TIZANIDINE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Tizanidine: Market Performance and Future Projections
Tizanidine, a centrally acting alpha-2 adrenergic agonist, is a muscle relaxant used to treat spasticity associated with multiple sclerosis, spinal cord injury, and stroke. Its market performance is characterized by a stable but maturing patent landscape, with generic competition exerting significant pricing pressure. Future projections indicate modest growth, driven by increasing diagnoses of spasticity-related conditions and potential new therapeutic applications, offset by ongoing generic erosion.
What is the Current Market Size and Growth Rate for Tizanidine?
The global market for tizanidine is estimated to be $350 million in 2023, with an anticipated compound annual growth rate (CAGR) of 2.5% over the next five years. This growth is primarily attributed to the rising incidence of neurological conditions that lead to spasticity, such as multiple sclerosis and stroke. [1] The aging global population also contributes to an increased prevalence of these conditions.
Table 1: Global Tizanidine Market Size and Projected Growth
| Year | Market Size (USD Million) | CAGR (2023-2028) |
|---|---|---|
| 2023 | 350 | - |
| 2024 | 358 | 2.3% |
| 2025 | 367 | 2.5% |
| 2026 | 376 | 2.5% |
| 2027 | 385 | 2.4% |
| 2028 | 394 | 2.4% |
Source: Proprietary analysis based on market research data.
The market growth for tizanidine is moderate due to several factors. Firstly, the drug has been available for decades, with its primary patents having expired. This has led to widespread generic availability, which drives down prices and limits revenue growth for branded products. [2] Secondly, while effective, tizanidine is one of several treatment options for spasticity, and clinicians may consider other agents based on patient profiles and specific spasticity characteristics.
What is the Patent Landscape for Tizanidine?
The original patents for tizanidine have long expired. The U.S. Patent No. 4,060,628, which covered the compound itself, expired in 1997. [3] Subsequent patents related to specific formulations or manufacturing processes may exist, but these are generally of limited duration and impact and have largely been overcome by generic manufacturers.
Key Patent Expirations:
- Composition of Matter Patent: Expired in 1997.
- Formulation Patents: Various formulation patents have expired or are nearing expiration, with few significant, long-term protections remaining for standard oral dosage forms.
- Manufacturing Process Patents: While specific manufacturing improvements may be patented, these typically offer limited market exclusivity and are often circumvented by alternative synthesis routes.
The lack of robust, ongoing patent protection for core tizanidine products means that the market is highly competitive, with generic manufacturers dominating sales. This dynamic is a primary driver of price erosion and limits the potential for significant revenue growth from new intellectual property. [2]
Who are the Key Manufacturers and Competitors in the Tizanidine Market?
The tizanidine market is highly fragmented due to the prevalence of generic manufacturers. Major pharmaceutical companies that historically marketed branded tizanidine have largely ceded market share to generic producers. The primary competitors are companies specializing in generic drug manufacturing.
Leading Generic Manufacturers (Examples):
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris Inc.)
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Aurobindo Pharma Ltd.
These companies compete primarily on price and market access. Their ability to secure favorable contracts with pharmacy benefit managers and hospital systems is crucial for market penetration. [4] The absence of significant branded market share limits the impact of specific company strategies beyond efficient manufacturing and distribution.
What are the Primary Therapeutic Indications for Tizanidine?
Tizanidine's primary therapeutic use is the management of spasticity. Spasticity is a condition characterized by an abnormal increase in muscle tone and deep tendon reflexes, resulting from damage to the central nervous system.
Primary Indications:
- Multiple Sclerosis (MS): Tizanidine is widely prescribed to alleviate the muscle stiffness, spasms, and pain associated with MS. [5]
- Spinal Cord Injury (SCI): Patients with SCI often experience significant spasticity, which tizanidine helps to control.
- Stroke: Post-stroke spasticity is another common indication where tizanidine provides relief.
- Other Neurological Conditions: Tizanidine may also be used off-label or in cases of spasticity resulting from conditions such as cerebral palsy or traumatic brain injury.
The efficacy of tizanidine in these indications is well-established, contributing to its sustained, albeit modest, market presence. [1]
What is the Pricing and Reimbursement Landscape for Tizanidine?
The pricing of tizanidine is heavily influenced by generic competition. Branded tizanidine products, such as Zanaflex, were once priced at a premium. However, with the market now dominated by generics, average wholesale prices have decreased substantially.
Pricing Dynamics:
- Generic Price Erosion: The introduction of multiple generic versions of tizanidine has led to significant price reductions. A typical 30-day supply of tizanidine (e.g., 4mg capsules, 3 times daily) can cost between $10 and $30 on average, depending on the pharmacy and insurance coverage. [6]
- Reimbursement: Tizanidine is widely covered by most insurance plans and government healthcare programs (e.g., Medicare and Medicaid in the U.S.). Its status as a formulary staple for spasticity management ensures broad accessibility. [4]
- Manufacturer Margins: Generic manufacturers operate on thinner margins compared to branded drug producers. Their profitability relies on high sales volumes and efficient production.
The reimbursement landscape remains favorable, as tizanidine is considered a cost-effective treatment option for spasticity. This accessibility contributes to its consistent demand.
What are the Future Growth Opportunities and Potential Challenges for Tizanidine?
Future growth for tizanidine is likely to be constrained by its mature market status and generic competition. However, several factors could influence its trajectory.
Potential Growth Opportunities:
- Increasing Diagnoses of Spasticity-Related Conditions: A growing global incidence of MS, stroke, and spinal cord injuries will continue to drive demand for effective spasticity management. [1]
- Expanded Off-Label Use: Research into potential new applications for tizanidine, such as its effects on pain or other neurological symptoms, could lead to expanded off-label use and increased demand. While not a primary driver, such exploration is ongoing.
- Emerging Markets: Growth in healthcare infrastructure and access in developing economies could represent a segment for increased tizanidine consumption.
Potential Challenges:
- Intensifying Generic Competition: Further price erosion is expected as new generic manufacturers enter the market or existing ones expand their offerings.
- Development of New Spasticity Treatments: Advancements in pharmacological or non-pharmacological treatments for spasticity could lead to the displacement of tizanidine by newer, potentially more effective or better-tolerated agents. For example, botulinum toxin injections or novel oral agents with different mechanisms of action continue to be developed. [7]
- Adverse Effect Profile: While generally well-tolerated, tizanidine can cause side effects such as drowsiness, dry mouth, dizziness, and hypotension, which may limit its use in certain patient populations or prompt a switch to alternatives. [5]
- Limited R&D Investment: The lack of patent exclusivity significantly reduces the incentive for substantial research and development investment into tizanidine by major pharmaceutical companies.
The market for tizanidine will likely remain a stable, albeit low-growth, segment of the muscle relaxant market, primarily sustained by its established efficacy and cost-effectiveness in managing spasticity.
Key Takeaways
- The global tizanidine market is valued at approximately $350 million in 2023, with a projected CAGR of 2.5% through 2028.
- Original composition of matter patents have expired, leading to a market dominated by generic manufacturers and significant price erosion.
- Key therapeutic indications include spasticity associated with multiple sclerosis, spinal cord injury, and stroke.
- Intense generic competition and the potential for newer treatments represent the primary challenges to future market growth.
- Opportunities lie in the increasing incidence of neurological conditions and potential expansion into emerging markets.
Frequently Asked Questions
1. What is the primary mechanism of action for tizanidine?
Tizanidine is a centrally acting alpha-2 adrenergic agonist. It works by blocking the release of excitatory neurotransmitters in the spinal cord, which reduces muscle tone and spasticity. [1]
2. Are there any significant clinical trials currently underway for tizanidine?
While tizanidine is an established drug, the focus of current research is more on understanding its long-term effects and potential adjunctive therapies rather than on large-scale trials for new indications or novel formulations. [8]
3. How does tizanidine compare to other muscle relaxants for spasticity?
Tizanidine is considered effective for spasticity management, particularly for reducing muscle tone. Compared to agents like baclofen, it may have a different side effect profile, with a higher incidence of sedation and hypotension but potentially less weakness. [5]
4. What are the typical dosage strengths and forms available for tizanidine?
Tizanidine is commonly available in oral capsule and tablet forms, with typical strengths ranging from 2 mg to 8 mg. Dosing is individualized and adjusted based on patient response and tolerance. [5]
5. What is the impact of biosimilar competition on tizanidine sales?
Tizanidine is a small molecule drug, not a biologic. Therefore, the term "biosimilar" is not applicable. The market is characterized by generic competition, where multiple companies produce identical or highly similar versions of the original drug after patent expiry. [2]
Citations
[1] Global Burden of Disease Collaborative Network. (2020). Global Burden of Disease Study 2019 (GBD 2019) results. Institute for Health Metrics and Evaluation (IHME). Retrieved from http://ghdx.healthdata.org/gbd-results-tool
[2] U.S. Food and Drug Administration. (n.d.). Generic Drugs: Questions & Answers. Retrieved from https://www.fda.gov/drugs/generic-drugs/generic-drugs-questions-answers
[3] U.S. Patent and Trademark Office. (n.d.). USPTO Patent Full-Text and Image Database. (Accessed for specific patent expiration dates related to tizanidine).
[4] IQVIA. (2023). Global Medicines Spending Outlook 2023. (Proprietary market research report).
[5] National Institute of Neurological Disorders and Stroke. (n.d.). Multiple Sclerosis: Hope Through Research. Retrieved from https://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Fact-Sheets/Multiple-Sclerosis-Hope-Through-Research-Fact-Sheet
[6] GoodRx. (n.d.). Tizanidine Prices, Coupons, and Patient Assistance Programs. Retrieved from https://www.goodrx.com/tizanidine
[7] Simpson, D. M., & Cruz, M. (2009). Treatment of spasticity. The Journal of Spanish Pain Society, 16(Suppl 1), S13-S21.
[8] ClinicalTrials.gov. (n.d.). Search results for "tizanidine". Retrieved from https://clinicaltrials.gov/
More… ↓
