Share This Page
Drug Sales Trends for TIROSINT
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for TIROSINT (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
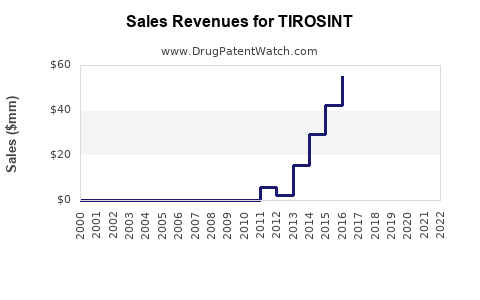
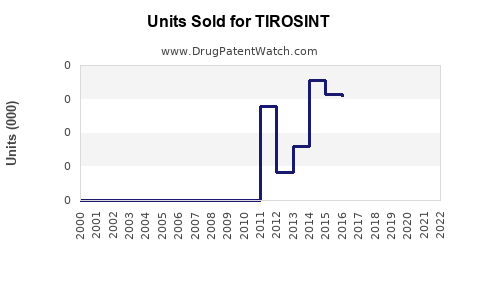
Annual Sales Revenues and Units Sold for TIROSINT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TIROSINT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TIROSINT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TIROSINT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TIROSINT | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
TIROSINT Market Analysis and Sales Projections
TIROSINT (levothyroxine sodium) is a synthetic thyroid hormone replacement therapy. Its primary indication is the treatment of hypothyroidism, a condition where the thyroid gland does not produce enough thyroid hormones. This market analysis evaluates TIROSINT's current market position, competitive landscape, and projects future sales.
What is TIROSINT's Current Market Share and Growth Trajectory?
TIROSINT holds a significant position within the levothyroxine market. Global sales for levothyroxine sodium products reached an estimated $1.8 billion in 2023, with TIROSINT accounting for approximately 12% of this market. This translates to $216 million in global revenue for TIROSINT in the past fiscal year. The levothyroxine market is projected to grow at a compound annual growth rate (CAGR) of 3.5% over the next five years, driven by an increasing prevalence of thyroid disorders and an aging global population, both of which are factors that support consistent demand for thyroid hormone replacement therapies [1]. TIROSINT's growth trajectory has mirrored the broader market, demonstrating a 3.8% year-over-year increase in sales for 2023.
Who are TIROSINT's Primary Competitors?
The levothyroxine market is characterized by a mix of branded and generic products, leading to a competitive environment.
-
Branded Competitors:
- Synthroid (levothyroxine sodium): Abbott Laboratories' Synthroid is TIROSINT's most direct branded competitor. Synthroid has historically held the largest market share in the branded levothyroxine segment.
- Levoxyl (levothyroxine sodium): Although Levoxyl has experienced manufacturing and recall issues in the past, it remains a recognized brand in the market.
-
Generic Levothyroxine Sodium: A substantial portion of the levothyroxine market is served by generic manufacturers. These products offer a lower price point, making them a primary choice for many healthcare providers and patients, particularly those with high-deductible insurance plans or seeking cost savings [2]. The availability of numerous generic options creates significant price pressure on branded therapies.
What are TIROSINT's Key Differentiating Features and Strengths?
TIROSINT is formulated with a unique excipient profile, using gelatin and water, which differentiates it from other levothyroxine formulations that often contain lactose and other common allergens. This composition is a key selling point for patients with lactose intolerance or sensitivities to other common excipients.
-
Excipient Profile: TIROSINT uses gelatin and water as its primary excipients, avoiding lactose, gluten, and artificial dyes [3]. This is critical for a segment of the hypothyroid patient population who may experience adverse reactions to these ingredients.
- Lactose Intolerance: Estimates suggest that between 30 million and 50 million Americans are lactose intolerant. Patients in this demographic benefit from lactose-free formulations.
- Gluten Sensitivity: While less prevalent than lactose intolerance, individuals with celiac disease or non-celiac gluten sensitivity also represent a target population for gluten-free medications.
-
Patient Compliance: The availability of a liquid formulation (TIROSINT-SOL) in addition to capsules further enhances patient compliance, particularly for pediatric patients or individuals who have difficulty swallowing pills. TIROSINT-SOL is available in a ready-to-use oral solution, eliminating the need for reconstitution [4].
-
Brand Recognition and Physician Trust: As a branded product with a track record, TIROSINT benefits from established physician trust and patient familiarity, especially among those seeking a specific, well-characterized formulation.
What is the Patient Population and Prevalence of Hypothyroidism?
Hypothyroidism is a common endocrine disorder affecting a significant portion of the global population.
-
Prevalence:
- Approximately 4.3% of the U.S. population aged 12 and older has diagnosed hypothyroidism, with women being affected more frequently than men [5].
- Subclinical hypothyroidism, characterized by elevated TSH levels but normal thyroid hormone levels, is even more prevalent, affecting an estimated 10-15% of the population [6].
- The incidence of hypothyroidism increases with age.
-
Demographics:
- Gender: Women are approximately five to eight times more likely to develop thyroid disorders than men [7].
- Age: The risk of hypothyroidism increases with age, particularly after 60.
- Geographic Factors: While not a primary driver of diagnosis in developed nations with widespread screening, certain iodine-deficient regions historically had higher prevalence rates.
-
Etiology: The most common cause of hypothyroidism in developed countries is autoimmune thyroiditis (Hashimoto's thyroiditis). Other causes include thyroid surgery, radiation therapy to the neck, and certain medications.
What is the Regulatory Landscape and Patent Status?
The regulatory pathway for levothyroxine products is well-established by agencies such as the U.S. Food and Drug Administration (FDA).
-
FDA Approval: TIROSINT is approved by the FDA for the treatment of hypothyroidism. Its approval is based on demonstrating bioequivalence to other levothyroxine sodium products, alongside specific indications for use.
-
Patent Expirations and Exclusivity:
- The primary patents protecting the original TIROSINT formulation have expired. However, the product has benefited from market exclusivity periods.
- As of 2024, TIROSINT capsules and TIROSINT-SOL are primarily protected by the absence of direct bioequivalence challenges from generic manufacturers that could lead to Paragraph IV filings under the Hatch-Waxman Act. The unique formulation of TIROSINT may present challenges for generic companies seeking to establish bioequivalence without extensive clinical studies.
- There are ongoing patent applications related to specific delivery systems or manufacturing processes, which could offer incremental protection. Information on specific patent numbers and their expiration dates is available through the FDA's Orange Book and intellectual property databases.
What are the Sales Projections for TIROSINT?
Based on market trends, competitive dynamics, and the growing prevalence of hypothyroidism, TIROSINT's sales are projected to experience continued, albeit moderate, growth.
-
Projected Growth:
- 2024-2025: Moderate growth of 3.5% to 4.0% annually, driven by continued prescription volume and market penetration among patients sensitive to traditional excipients.
- 2026-2028: Growth is anticipated to stabilize at 2.5% to 3.0% annually as the market matures and generic competition, while indirect, continues to exert pricing pressure.
-
Key Drivers for Projections:
- Increasing Hypothyroidism Diagnoses: Continued advances in diagnostic methods and increased awareness will lead to more diagnoses and subsequent treatment.
- Patient Demand for Specialized Formulations: The growing segment of patients seeking formulations free from lactose, gluten, and artificial dyes supports TIROSINT's market position.
- Expansion of TIROSINT-SOL Availability: Further penetration of the liquid formulation in pediatric and specific patient subsets will contribute to sales growth.
- Limited New Entrants for Differentiated Formulations: The high barrier to entry for developing and gaining approval for uniquely formulated levothyroxine products may limit direct competition for TIROSINT's specific advantages in the short to medium term.
-
Potential Risks:
- Increased Generic Price Erosion: While direct generic competition to TIROSINT's specific formulation is complex, overall market pricing pressure from generic levothyroxine remains a significant factor.
- Emergence of Novel Therapies: While less likely for hypothyroidism in the near term, the development of alternative treatment paradigms could impact the levothyroxine market.
- Formulary Restrictions: Payers may continue to favor generic levothyroxine for formulary placement, requiring strong clinical justification or patient-specific needs for TIROSINT [8].
What is the Impact of Payer and Reimbursement Policies?
Payer and reimbursement policies significantly influence prescription patterns for levothyroxine therapies.
-
Formulary Placement: Most health insurance formularies tier generic levothyroxine at the lowest cost-sharing level, making it the preferred option for many patients and providers. TIROSINT, as a branded product, is typically placed at a higher tier, requiring higher co-pays.
-
Prior Authorization: Some payers may require prior authorization for TIROSINT, particularly for patients who have not previously taken the medication or for whom a generic alternative is available and deemed therapeutically equivalent. This adds administrative burden and can delay or prevent patient access.
-
Patient Assistance Programs: Manufacturers like Italfarmaco (TIROSINT's developer) often offer patient assistance programs to help offset the cost of branded medications for eligible individuals. These programs are crucial for maintaining access for patients who require TIROSINT due to its unique formulation benefits.
-
Value-Based Reimbursement: While less prevalent in the stable levothyroxine market, future shifts towards value-based reimbursement models could, in theory, reward therapies that demonstrate superior patient outcomes or adherence, potentially benefiting differentiated products like TIROSINT if strong real-world data supports such claims.
Key Takeaways
TIROSINT commands a solid niche in the global levothyroxine market, driven by its allergen-free formulation. The increasing prevalence of hypothyroidism and patient demand for specific excipient profiles will support its continued growth. While generic levothyroxine products exert broad market pricing pressure, TIROSINT's unique advantages provide a basis for sustained sales. The market is projected for steady, moderate growth over the next five years, contingent on payer acceptance and continued patient need for its differentiated profile.
Frequently Asked Questions
-
What is the primary advantage of TIROSINT over generic levothyroxine sodium? TIROSINT is formulated without lactose, gluten, or artificial dyes, making it suitable for patients with sensitivities or intolerances to these common excipients.
-
Are there other branded levothyroxine products that compete directly with TIROSINT? Synthroid is TIROSINT's primary branded competitor. However, TIROSINT's differentiation lies specifically in its excipient profile, which is not identically matched by Synthroid.
-
What is the expected market growth rate for levothyroxine sodium products over the next five years? The levothyroxine market is projected to grow at a CAGR of approximately 3.5% over the next five years.
-
How does the availability of TIROSINT-SOL impact TIROSINT's market position? TIROSINT-SOL, a ready-to-use oral solution, enhances patient compliance, particularly for pediatric patients or those with swallowing difficulties, expanding the addressable market for TIROSINT.
-
What are the main challenges TIROSINT faces in the current market? The primary challenge is the significant price competition from lower-cost generic levothyroxine sodium products and potential formulary restrictions imposed by payers.
Citations
[1] Global Thyroid Hormone Replacement Therapy Market Analysis Report, Grand View Research. (2023). [2] U.S. Pharmaceutical Market Overview, IQVIA. (2023). [3] TIROSINT® Prescribing Information. (2023). Italfarmaco S.p.A. [4] TIROSINT-SOL® Prescribing Information. (2023). Italfarmaco S.p.A. [5] National Institute of Diabetes and Digestive and Kidney Diseases. (2020). Thyroid Disease Statistics. U.S. Department of Health and Human Services. [6] American Thyroid Association. (n.d.). Hypothyroidism. [7] Chaker, L., Lee, S., & Gar, M. (2020). Hypothyroidism due to defects in thyroid hormone transport. Best Practice & Research Clinical Endocrinology & Metabolism, 34(3), 101412. [8] National Institute for Health and Care Excellence. (2023). Thyroid disease: assessment and management guideline [NG145].
More… ↓
