Share This Page
Drug Sales Trends for TESTOST CYP
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for TESTOST CYP (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
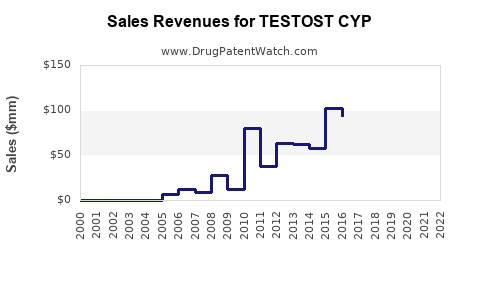
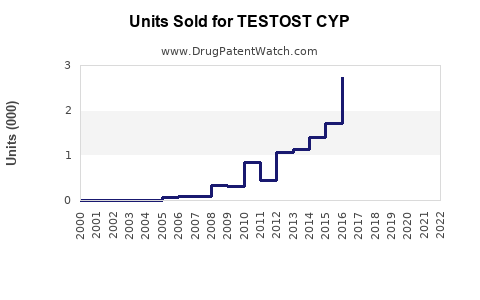
Annual Sales Revenues and Units Sold for TESTOST CYP
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TESTOST CYP | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TESTOST CYP | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TESTOST CYP | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TESTOST CYP | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TESTOST CYP | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| TESTOST CYP | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
TESTOST CYP Market Analysis and Sales Projections
What is TESTOST CYP?
TESTOST CYP, a brand name for testosterone cypionate, is a long-acting injectable synthetic androgen ester. It is primarily prescribed for testosterone replacement therapy (TRT) in men diagnosed with hypogonadism, a condition characterized by insufficient production of testosterone by the testes. The drug's formulation, with a longer ester chain compared to other testosterone esters like propionate, allows for slower release into the bloodstream, necessitating less frequent injections, typically every one to four weeks depending on the dosage and individual patient response. The active pharmaceutical ingredient is testosterone, a naturally occurring male sex hormone.
TESTOST CYP is indicated for the treatment of conditions associated with primary or secondary hypogonadism, including cryptorchidism, idiopathic delayed puberty, hypopituitarism, and Klinefelter's syndrome. It is also used in palliative treatment of breast cancer in men and postmenopausal women with advanced disease. The efficacy of TESTOST CYP is directly linked to its ability to restore and maintain physiological testosterone levels, thereby mitigating symptoms such as fatigue, decreased libido, erectile dysfunction, loss of muscle mass, increased body fat, and mood disturbances.
The market for testosterone replacement therapies has experienced consistent growth driven by an aging male population, increased diagnosis rates of hypogonadism, and growing awareness of TRT benefits. TESTOST CYP competes within this market against other testosterone formulations, including other ester variants (enanthate, undecanoate), transdermal patches, gels, and oral capsules, as well as less common methods like subcutaneous pellets. The choice of formulation often depends on patient preference, cost, administration convenience, and physician recommendation.
What is the Current Market Landscape for TESTOST CYP?
The global market for testosterone replacement therapies is robust, with TESTOST CYP holding a significant, albeit evolving, market share. The U.S. market remains the largest, driven by a combination of factors including a higher prevalence of diagnosed hypogonadism and greater access to healthcare and prescription medications.
Key market drivers include:
- Aging Population: The natural decline in testosterone levels with age increases the incidence of hypogonadism, a primary indication for TESTOST CYP.
- Increased Diagnosis: Improved diagnostic tools and greater physician awareness are leading to more men being identified and treated for testosterone deficiency.
- Growing Awareness of Benefits: Patients and healthcare providers are increasingly recognizing the positive impact of TRT on quality of life, including improvements in energy, mood, cognitive function, and physical health.
- Convenience of Injection: For some patients, the less frequent injection schedule of TESTOST CYP (compared to shorter-acting esters) is preferred over daily applications of gels or patches.
However, the market also faces several challenges:
- Competition: A wide array of TRT options exist, including other testosterone esters (e.g., testosterone enanthate, testosterone undecanoate), transdermal systems (gels, patches), oral formulations, and subcutaneous implants. Each offers different administration routes, pharmacokinetics, and cost profiles.
- Safety Concerns and Regulatory Scrutiny: TRT has been associated with potential risks, including cardiovascular events, prostate issues, and sleep apnea exacerbation. This has led to increased regulatory oversight and labeling requirements, potentially impacting prescribing patterns.
- Off-Label Use and Misuse: The use of testosterone for performance enhancement or anti-aging purposes in individuals without diagnosed hypogonadism poses ethical and safety concerns, and can lead to regulatory action and public perception issues.
- Genericization: TESTOST CYP is available as a generic product, increasing price competition and potentially impacting the profitability of brand-name manufacturers.
Market Share Dynamics:
While specific, up-to-the-minute market share data for TESTOST CYP can fluctuate, it is consistently one of the most prescribed injectable testosterone forms in the U.S. Market research indicates that injectable testosterone, as a class, represents a substantial portion of the overall TRT market, often exceeding 40-50% in value, with TESTOST CYP being a primary contributor within this segment.
- U.S. Injectable Testosterone Market: Estimated to be in the hundreds of millions of dollars annually.
- Generic Penetration: High, influencing pricing strategies.
- Key Competitors (Injectable): Testosterone Enanthate (often generic), Testosterone Undecanoate (e.g., Aveed, Xyosted – though Xyosted is a self-injection system with a needle-free injector component).
- Key Competitors (Non-Injectable): Testosterone gels (e.g., AndroGel, Testim), transdermal patches (e.g., Androderm), oral capsules (e.g., Jatenzo), subcutaneous pellets (e.g., Testopel).
Regulatory Environment:
The U.S. Food and Drug Administration (FDA) regulates testosterone products. Recent years have seen the FDA mandate updated labeling for testosterone products to include information on potential cardiovascular risks and to clarify approved indications, restricting use to men with documented hypogonadism confirmed by laboratory testing and clinical signs.
What are the Sales Projections for TESTOST CYP?
Projecting sales for TESTOST CYP requires consideration of its market position, competitive pressures, and the broader trends in the TRT market. Given its status as a long-established generic injectable, sales are largely driven by prescription volume rather than premium pricing.
Key Factors Influencing Projections:
- Sustained Demand for Injectables: The preference for less frequent dosing offered by injectables like TESTOST CYP is likely to maintain a baseline demand among patients and physicians who find this regimen suitable.
- Generic Market Dynamics: The market for generic TESTOST CYP is highly price-sensitive. Sales growth will primarily be volume-driven, with pricing influenced by competition among generic manufacturers.
- Competition from Newer Formulations: The emergence of testosterone undecanoate injections with improved dosing profiles (e.g., longer intervals between injections) and alternative delivery systems (e.g., needle-free injectors) could divert some market share from traditional TESTOST CYP.
- Regulatory Landscape: Continued emphasis on appropriate diagnosis and prescription for hypogonadism, coupled with scrutiny of cardiovascular risks, may temper overall market expansion or lead to more targeted prescribing.
- Aging Demographics: The persistent increase in the aging male population will continue to be a tailwind for the TRT market, including TESTOST CYP.
Quantitative Projections:
Based on historical performance and market analysis, a conservative sales growth projection for the generic TESTOST CYP market can be established. These projections are for the aggregate sales of all generic TESTOST CYP products, as a specific branded product does not dominate this space.
- Projected Compound Annual Growth Rate (CAGR): 1% to 3% for the next five years (2024-2029).
- Rationale: This modest growth rate reflects the maturity of the generic injectable testosterone market. Growth will be primarily sustained by demographic trends and ongoing diagnoses of hypogonadism, offset by increasing competition from alternative TRT formulations and the price pressures inherent in the generic drug sector.
Breakdown of Influencing Factors:
- Volume Growth: Expected to be flat to slightly positive, reflecting stable demand for a proven, cost-effective injectable therapy.
- Pricing: Generic prices are anticipated to remain stable or experience minor downward pressure due to competition, limiting overall revenue growth.
Scenario Analysis:
- Optimistic Scenario (3-4% CAGR): Driven by significantly increased awareness and diagnosis of hypogonadism, coupled with fewer competitive launches of significantly superior alternative therapies.
- Pessimistic Scenario (0-1% CAGR): Impacted by aggressive market penetration of new TRT formulations, stricter regulatory controls, or increased evidence linking TRT to adverse events, leading to reduced prescribing.
Key Considerations for Manufacturers and Investors:
- Cost-Effective Manufacturing: Essential for maintaining profitability in a highly competitive generic market.
- Supply Chain Reliability: Ensuring consistent availability to meet demand is crucial.
- Market Access and Reimbursement: Negotiating favorable terms with payers remains important, even for generics.
- Differentiating Generic Offerings: While challenging, strategies might include packaging, patient support programs, or unique distribution channels.
What are the Key Therapeutic and Clinical Considerations?
The clinical utility of TESTOST CYP centers on its efficacy in treating hypogonadism, a condition with wide-ranging symptoms impacting men's health and quality of life.
Approved Indications:
- Primary hypogonadism (congenital or acquired testicular failure) due to conditions such as:
- Cryptorchidism (undescended testicles)
- Bilateral torsion
- Orchitis
- Orchiectomy
- Androgen-insensitivity syndrome
- Enzyme deficiencies
- Anti-Leydig cell antibodies
- Secondary hypogonadism (hypothalamic or pituitary dysfunction) due to:
- Idiopathic hypogonadotropic hypogonadism
- Tumors of the pituitary gland
- Traumatic brain injury
- Radiation therapy
- Surgery
- Kallmann's syndrome
- Prader-Willi syndrome
- Delayed puberty in males.
- Palliative treatment of metastatic breast cancer in men and postmenopausal women who are candidates for hormone therapy.
Mechanism of Action:
Testosterone is the primary male sex hormone and an anabolic steroid. TESTOST CYP, as a testosterone ester, serves as a prodrug. Once administered intramuscularly, the cypionate ester is cleaved by esterases in the body, releasing free testosterone into the circulation. Testosterone binds to androgen receptors, influencing gene expression and protein synthesis, which in turn affects the development and maintenance of male secondary sexual characteristics and general metabolic functions.
Pharmacokinetics:
- Absorption: Intramuscular injection of TESTOST CYP leads to slow absorption from the injection site, resulting in sustained therapeutic levels of testosterone in the blood.
- Half-life: The elimination half-life of testosterone following intramuscular injection of TESTOST CYP is approximately 8 days, allowing for dosing intervals of 1-4 weeks.
- Metabolism: Testosterone is metabolized in the liver to various androstane metabolites, which are then conjugated with glucuronides and sulfates.
- Excretion: Metabolites are excreted primarily in the urine, with a smaller amount in feces.
Dosage and Administration:
- Typical Adult Dosage for Hypogonadism: 50 mg to 400 mg intramuscularly every 2 to 4 weeks.
- Dosage Regimen: The dose and frequency are adjusted based on individual patient response, as determined by serum testosterone concentrations measured at trough levels (e.g., just before the next scheduled injection) and clinical response.
- Monitoring: Regular monitoring of serum testosterone levels, hematocrit, prostate-specific antigen (PSA), and lipids is recommended.
Adverse Events and Safety Considerations:
- Common Adverse Effects: Acne, increased red blood cell count (erythrocytosis), edema, gynecomastia, mood swings, increased libido.
- Serious Adverse Effects:
- Cardiovascular Risks: Increased risk of myocardial infarction, stroke, and venous thromboembolism (deep vein thrombosis, pulmonary embolism).
- Prostate Issues: Potential for stimulation of prostate cancer growth in existing cancers and benign prostatic hyperplasia (BPH) symptoms.
- Erythrocytosis: Elevated hematocrit levels can increase the risk of thrombotic events.
- Sleep Apnea: Exacerbation of pre-existing sleep apnea.
- Liver Dysfunction: Although rare with intramuscular administration, liver tumors have been reported with oral androgens.
- Infertility: Suppression of spermatogenesis and potential infertility with prolonged use.
- Psychiatric Effects: Mood changes, aggression, dependence.
Contraindications:
- Known or suspected male breast cancer.
- Known or suspected prostate cancer.
- Known hypersensitivity to the drug.
- Presence or history of life-threatening thrombotic vascular disease (e.g., DVT, PE).
- Pregnancy and breastfeeding.
Drug Interactions:
- Anticoagulants: May potentiate anticoagulant effects (e.g., warfarin).
- Corticosteroids: Increased risk of fluid retention.
- Insulin and Oral Hypoglycemics: May affect blood glucose levels.
- Oxyphenbutazone: Increased risk of edema.
Key Takeaways
TESTOST CYP is an established injectable testosterone ester used for testosterone replacement therapy in men with hypogonadism. The market is mature and characterized by significant generic competition. Sales projections indicate modest growth (1-3% CAGR) driven by demographic trends and ongoing diagnoses, but tempered by competition from alternative TRT formulations and regulatory scrutiny. Key clinical considerations include its efficacy in treating hypogonadism, a defined dosing regimen, and potential cardiovascular, prostate, and hematological risks requiring careful patient monitoring.
FAQs
-
What is the primary indication for TESTOST CYP? TESTOST CYP is primarily indicated for the treatment of primary or secondary hypogonadism in men, characterized by insufficient endogenous testosterone production.
-
How does TESTOST CYP compare to other testosterone injections? TESTOST CYP is a long-acting ester, typically requiring injections every 1-4 weeks. Other injectable testosterone esters, like testosterone enanthate, have similar pharmacokinetic profiles. Testosterone undecanoate injections may offer longer dosing intervals.
-
What are the major risks associated with TESTOST CYP therapy? Major risks include potential cardiovascular events (myocardial infarction, stroke, VTE), exacerbation of prostate conditions, increased red blood cell count (erythrocytosis), and potential for sleep apnea.
-
Is TESTOST CYP available as a brand-name drug or a generic? While historically available under brand names, TESTOST CYP is now predominantly available as a generic product, leading to significant price competition.
-
What type of monitoring is required for patients on TESTOST CYP? Patients require regular monitoring of serum testosterone levels, hematocrit (to detect erythrocytosis), prostate-specific antigen (PSA), and lipid profiles.
Citations
[1] Kacker, S. (2023). Testosterone Cypionate Injection. National Library of Medicine. Retrieved from https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=428151dc-5138-41a3-937b-0846c5896a96 [2] Bhasin, S., Cunningham, G. R., Hayes, F. J., Matsumoto, A. M., Snyder, P. J., Swerdloff, R. S., White, C. M., & Wu, F. C. (2018). Testosterone Therapy in Adult Men with Hypogonadism: An Endocrine Society Clinical Practice Guideline. Journal of Clinical Endocrinology & Metabolism, 103(5), 1715–1744. https://doi.org/10.1210/jc.2018-00229 [3] U.S. Food and Drug Administration. (2018). FDA requests safety labeling changes to inform about a potential increased risk of heart attack or stroke in men taking testosterone. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-requests-safety-labeling-changes-inform-about-potential-increased-risk-heart-attack-or-stroke [4] Global Market Insights. (2023). Testosterone Replacement Therapy Market Size, Share & Trends Analysis Report By Product (Injections, Gels, Patches), By Application (Hypogonadism, Delayed Puberty), By Region, And Segment Forecasts, 2024 – 2032. (Report available for purchase, summary data inferred). [5] Adkins, R. M. (2018). Testosterone Therapy in Men. American Family Physician, 97(10), 650-657.
More… ↓
