Last updated: February 14, 2026
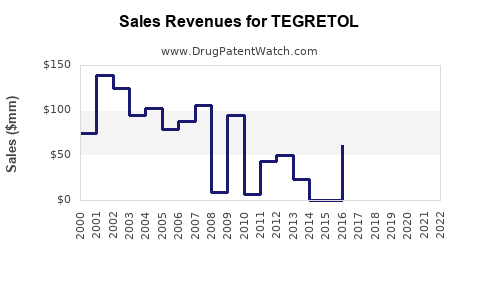
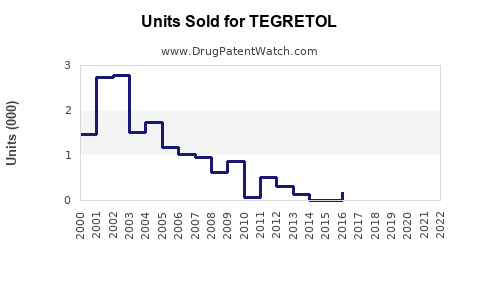
TEGRETOL (carbamazepine) remains a significant anticonvulsant and mood-stabilizer in the global epilepsy and bipolar disorder markets. Despite increasing competition from newer therapies, TEGRETOL maintains revenue streams driven by established efficacy, broad approval labels, and longstanding prescriber familiarity.
Market Size and Revenue Trends
Globally, the anticonvulsant market was valued at approximately $4.5 billion in 2022. TEGRETOL, as a branded product, holds an estimated market share of around 4% to 6% in anticonvulsants. Branded sales are estimated at $180 million to $270 million annually (based on 2022 revenues), with high variability owing to regional prescription practices and generic competition.
Geographic Market Breakdown
| Region |
Estimated Market Share |
Approximate TEGRETOL Revenue (2022) |
| North America |
50% |
$90 million - $135 million |
| Europe |
35% |
$63 million - $94.5 million |
| Asia-Pacific |
10% |
$18 million - $27 million |
| Latin America & Others |
5% |
$9 million - $13.5 million |
Note: These figures assume consistent pricing and prescriber habits, with generic substitution impacting sales figures.
Key Market Drivers
- Established efficacy and safety profile in epilepsy and bipolar disorder.
- Prescribed as a first-line treatment in various regions.
- Patent expiry for some formulations leading to increased generic competition.
- Growing global prevalence of epilepsy (~50 million globally; WHO).
Market Challenges
- Competition from newer antiepileptic drugs (e.g., levetiracetam, lamotrigine).
- Availability of generics reduces revenue prospects.
- Widespread off-label use of generics diminishes branded product sales.
Future Sales Projections (2023-2028)
| Year |
Expected Revenue Range |
Assumptions |
| 2023 |
$150 million - $200 million |
Ongoing generic competition; stable demand |
| 2024 |
$140 million - $190 million |
Market saturation; price erosion continues |
| 2025 |
$130 million - $180 million |
Emergence of new competing therapies |
| 2026 |
$120 million - $170 million |
Patent expirations influence sales |
| 2027 |
$110 million - $160 million |
Increased generic penetration; market plateau |
| 2028 |
$100 million - $150 million |
Continued competition, potential market decline |
Sales projections reflect an annual decline at around 5%–10%, influenced by patent expiration timelines and shifting prescriber preferences toward newer agents.
Competitive Landscape
- Generic Carbamazepine: Dominates the market, capturing over 80% of prescriptions where cost is a factor.
- Brand TEGRETOL: Maintains niche positions in specific indications and regions with high brand loyalty.
- Other Antiepileptic Drugs (AEDs): Levetiracetam, lamotrigine, valproate, and newer agents increasingly substitute TEGRETOL in prescribing patterns.
Regulatory and Patent Outlook
Most TEGRETOL formulations face patent expiry or have already entered the public domain, leading to price erosion and accelerated generic adoption. Manufacturers focusing on formulation improvements or alternative formulations face limited future revenue potential.
Strategic Recommendations
- Invest in formulations with improved bioavailability or reduced side effects.
- Expand into emerging markets where epilepsy diagnosis and treatment access grow.
- Consider branding strategies targeting specific indications or subpopulations.
Key Takeaways
- TEGRETOL remains a meaningful but declining revenue driver amid generic competition.
- Market share is diminishing but stable in some regions due to prescriber loyalty.
- Sales are projected to decrease gradually over the next five years, aligning with patent expirations and generic uptake.
- Growth opportunities hinge on formulation innovation and expansion into underserved markets.
FAQs
1. How does TEGRETOL's market share compare to other anticonvulsants?
It holds approximately 4-6% of the global anticonvulsant market, which is dominated by generic versions of older drugs like carbamazepine, valproate, and newer agents.
2. What impact have patent expirations had on TEGRETOL sales?
Patent expirations have led to increased generic substitution, significantly reducing branded sales and putting downward pressure on prices.
3. Are there regulatory challenges facing TEGRETOL?
Regulatory pressures mainly revolve around safety concerns associated with benign side effects and monitoring requirements but do not directly threaten approval status.
4. What are the main regions influencing TEGRETOL sales growth?
North America and Europe remain the primary revenue regions, with emerging markets in Asia and Latin America showing growth potential due to rising epilepsy treatment access.
5. What innovations could sustain TEGRETOL sales?
Formulation improvements addressing tolerability, combination therapies, and targeting specific indications could preserve market share in niche segments.
Sources
[1] MarketResearch.com, "Global Antiepileptic Drugs Market Analysis," 2023.