Share This Page
Drug Sales Trends for SOLODYN
✉ Email this page to a colleague
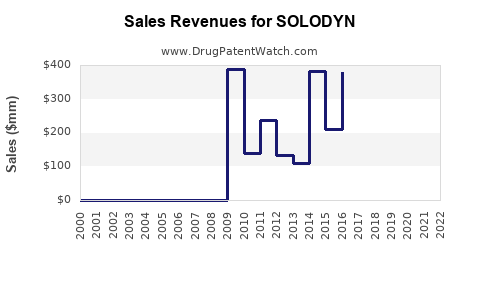
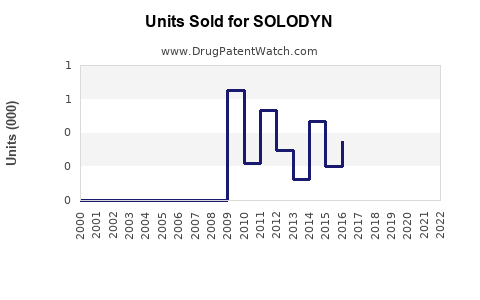
Annual Sales Revenues and Units Sold for SOLODYN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| SOLODYN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| SOLODYN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| SOLODYN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| SOLODYN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| SOLODYN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
SOLODYN (Minocycline Extended-Release) Market Analysis and Sales Projections
SOLODYN (minocycline extended-release) is a prescription antibiotic indicated for the treatment of inflammatory lesions of non-nodular moderate to severe acne vulgaris. The drug is manufactured by Valeant Pharmaceuticals International, now Bausch Health Companies. Its extended-release formulation offers a once-daily dosing regimen, differentiating it from immediate-release minocycline products.
What is the Current Market Landscape for Acne Treatments?
The global acne vulgaris treatment market is substantial and driven by the high prevalence of the condition, particularly among adolescents and young adults. This market segment encompasses a wide range of therapeutic classes, including topical agents (retinoids, antibiotics, benzoyl peroxide), oral antibiotics (tetracyclines, macrolides), hormonal agents, and systemic isotretinoin.
The market is characterized by:
- High Patient Volume: Acne affects an estimated 80-95% of adolescents [1].
- Diverse Treatment Options: A wide array of therapeutic interventions exists, catering to varying severity and types of acne.
- Growing Demand for Efficacy and Convenience: Patients and prescribers seek treatments that offer robust efficacy with simplified dosing and reduced side effects.
- Generic Competition: Many established acne treatments face significant generic competition, impacting pricing and market share.
- Therapeutic Gaps: Despite numerous options, unmet needs persist for severe or treatment-resistant acne, as well as for managing side effects of existing therapies.
Key therapeutic categories within the acne market include:
- Topical Agents: Retinoids (e.g., tretinoin, adapalene), topical antibiotics (e.g., clindamycin), benzoyl peroxide, and dapsone. These are generally first-line treatments for mild to moderate acne.
- Oral Antibiotics: Tetracyclines (e.g., doxycycline, minocycline) and macrolides (e.g., erythromycin) are used for moderate to severe inflammatory acne. Prescribing guidelines often recommend limited durations due to concerns about antibiotic resistance.
- Systemic Isotretinoin: A potent retinoid used for severe, recalcitrant nodular acne. It has significant efficacy but requires strict monitoring due to potential teratogenicity and other side effects.
- Hormonal Agents: Oral contraceptives and spironolactone are used in women with acne linked to hormonal fluctuations.
The market is influenced by regulatory guidelines concerning antibiotic stewardship, which may impact the long-term use of oral antibiotics. Research into novel mechanisms of action and more targeted therapies continues.
How Has SOLODYN Performed Historically?
SOLODYN was launched by Medicis Pharmaceutical Corp. in 2006. Its key differentiator was the extended-release formulation of minocycline, which was designed to provide a more consistent plasma concentration, potentially leading to improved tolerability and efficacy compared to immediate-release minocycline [2]. Valeant acquired Medicis in 2012.
Sales Performance of SOLODYN (USD Millions):
| Year | Sales (USD Millions) |
|---|---|
| 2013 | 320 |
| 2014 | 365 |
| 2015 | 350 |
| 2016 | 272 |
| 2017 | 175 |
| 2018 | 130 |
| 2019 | 108 |
| 2020 | 96 |
| 2021 | 88 |
| 2022 | 75 |
Source: Company filings and industry reports. Figures are approximate and may reflect consolidated reporting by Bausch Health Companies.
The sales trajectory of SOLODYN shows a significant peak in the mid-2010s, followed by a substantial decline. This decline is primarily attributable to:
- Patent Expirations and Generic Entry: The patent protection for SOLODYN expired, paving the way for generic versions of extended-release minocycline to enter the market. Generic competition typically leads to significant price erosion and market share loss for branded products.
- Increased Competition: The broader acne treatment market is highly competitive, with new topical and oral agents continually emerging.
- Shifting Prescribing Practices: Concerns regarding antibiotic resistance and the increasing availability of effective alternative treatments, including topicals and non-antibiotic oral agents, have likely influenced prescribing patterns.
- Price Increases and Scrutiny: Valeant (now Bausch Health) faced scrutiny over significant price increases on some of its legacy products, which may have impacted formulary access and prescriber adoption for SOLODYN in later years.
Despite the decline, SOLODYN continued to generate sales for a period due to its established brand recognition and physician familiarity. However, the market dynamics have fundamentally shifted with the availability of generics.
What are the Key Patents and Exclusivities for SOLODYN?
SOLODYN's patent landscape is critical to understanding its market exclusivity and the subsequent impact of generic entry. The primary patent protecting the extended-release formulation was U.S. Patent No. 5,976,573.
- U.S. Patent No. 5,976,573: This patent, titled "Extended release dosage forms of 6-demethyl-6-deoxytetracyclines," was issued on November 2, 1999. It claimed methods of treating acne vulgaris with extended-release minocycline formulations and the formulations themselves.
- Exclusivity: Based on its issuance date and regulatory pathways, this patent provided a period of market exclusivity for SOLODYN.
Key Dates:
- Patent Issuance: November 2, 1999
- Anticipated Expiration (excluding extensions): November 2, 2016
Patent Term Extensions and Hatch-Waxman Act:
The Hatch-Waxman Act allows for patent term extensions to compensate for regulatory review periods. SOLODYN, like other pharmaceuticals, would have been eligible for such extensions. However, the exact duration of extensions can be complex and dependent on specific applications and approvals. For SOLODYN, the key period of market exclusivity based on its primary formulation patent and potential extensions would have largely concluded around the mid-to-late 2010s.
Generic Entry:
Following the expiration of key patents and any associated exclusivities, generic manufacturers began to launch their versions of extended-release minocycline. This typically involves Abbreviated New Drug Applications (ANDAs) where generic companies demonstrate bioequivalence to the reference listed drug (SOLODYN). The first generic versions of extended-release minocycline emerged in the market around 2016-2017.
The presence of multiple generic competitors has led to a dramatic reduction in the price of extended-release minocycline and has significantly eroded SOLODYN's market share and revenue. Bausch Health's ability to maintain significant sales post-patent expiry is highly dependent on its ability to compete on price, formulary access, and potentially by emphasizing specific patient profiles or physician loyalty.
What is the Competitive Landscape for Extended-Release Minocycline and Similar Acne Treatments?
The competitive landscape for SOLODYN is multifaceted, encompassing both direct generic competitors and alternative therapeutic classes for acne.
Direct Generic Competition:
Since the expiration of SOLODYN's core formulation patents, numerous generic manufacturers have entered the market with their own versions of extended-release minocycline. These products are bioequivalent to SOLODYN and are typically offered at significantly lower price points. Major generic pharmaceutical companies have launched these products, leading to intense price competition. Examples of generic manufacturers include:
- Teva Pharmaceuticals
- Apotex Inc.
- Sun Pharmaceutical Industries
- Mylan N.V. (now Viatris)
The availability of these generics means that prescribers and patients now have multiple low-cost options for minocycline extended-release. This has effectively commoditized the extended-release minocycline segment.
Alternative Therapeutic Classes:
Beyond direct generic competition, SOLODYN competes with a broad spectrum of acne treatments:
-
Other Oral Antibiotics:
- Doxycycline (various formulations): A widely used tetracycline antibiotic, often available in both immediate and delayed-release formulations. Doxycycline is generally considered a first-line oral antibiotic for inflammatory acne.
- Erythromycin: A macrolide antibiotic, sometimes used when tetracyclines are contraindicated, but often less preferred due to side effect profiles and resistance concerns.
- Tetracycline: An older tetracycline, less commonly prescribed for acne due to absorption issues and tolerability.
-
Topical Treatments:
- Topical Retinoids: Adapalene (e.g., Differin), tretinoin, and tazarotene are cornerstones of acne management, particularly for comedonal and inflammatory acne. Adapalene is available over-the-counter, increasing its accessibility.
- Benzoyl Peroxide: A widely available over-the-counter and prescription agent with antibacterial and keratolytic properties.
- Topical Antibiotics: Clindamycin and erythromycin, often used in combination with benzoyl peroxide or retinoids to reduce resistance.
- Dapsone Gel: A topical anti-inflammatory agent.
-
Systemic Isotretinoin:
- Isotretinoin (e.g., Accutane, Claravis, Amnesteem): The most effective treatment for severe, recalcitrant nodular acne. While highly effective, its use is limited by its significant side effect profile and teratogenicity, requiring strict patient monitoring under the iPLEDGE program in the U.S.
-
Hormonal Therapies (for females):
- Oral Contraceptives: Certain combined oral contraceptives are FDA-approved for acne treatment in women.
- Spironolactone: An anti-androgen medication used off-label for hormonal acne in women.
Factors Influencing Competition:
- Efficacy: Different treatments are indicated for different severities and types of acne.
- Safety and Tolerability: Side effect profiles (e.g., gastrointestinal issues, photosensitivity for tetracyclines; teratogenicity for isotretinoin) heavily influence prescribing.
- Cost and Insurance Coverage: The availability and cost of generic alternatives, as well as formulary placement by insurance providers, are major drivers.
- Antibiotic Stewardship: Guidelines promoting judicious use of oral antibiotics to combat resistance favor topical treatments or non-antibiotic alternatives for longer-term management.
- Convenience: Once-daily dosing, as offered by SOLODYN and its generics, is a significant convenience factor for patients.
The market is highly fragmented, with a wide range of treatment options available. Prescribers often use combination therapies. The continued emphasis on antibiotic stewardship is likely to further favor non-antibiotic or limited-duration antibiotic approaches where possible.
What are the Potential Future Sales Projections for SOLODYN?
Predicting future sales for a branded drug facing extensive generic competition requires careful consideration of market trends, competitive pressures, and the product's remaining market position.
Given that the core patents for SOLODYN have expired and multiple generic versions of extended-release minocycline are widely available, significant sales growth for the branded SOLODYN product is highly unlikely. Instead, the projection is for continued decline, albeit at a potentially moderated pace depending on Bausch Health's commercial strategy.
Key Factors Influencing Future Sales:
- Ubiquitous Generic Availability: The primary driver of sales decline will be the widespread availability and lower cost of generic extended-release minocycline. Generic penetration in oral antibiotics for acne is typically very high once generics enter the market.
- Price Erosion: Generic competition inevitably leads to substantial price reductions. Bausch Health will likely struggle to maintain its pricing power for branded SOLODYN against lower-cost generics.
- Prescriber and Payer Preferences: Insurance formularies often prioritize generics due to cost savings, limiting access to branded products unless there is a specific clinical justification or a compelling cost-benefit argument for the brand. Prescribers may also default to generics for cost-conscious patients.
- Antibiotic Stewardship: The ongoing emphasis on reducing antibiotic use for acne will likely continue to favor non-antibiotic therapies or limit the duration of oral antibiotic prescriptions, impacting overall demand for products like SOLODYN.
- Evolving Treatment Landscape: The development of novel acne treatments with different mechanisms of action or improved safety profiles could further reduce the market share for older antibiotic classes.
Projected Sales Trend:
The sales of branded SOLODYN are projected to continue a steady decline over the next 3-5 years. The rate of decline will depend on how aggressively generics capture market share and how effectively Bausch Health can leverage any remaining competitive advantages, such as established physician relationships or specific patient support programs.
Estimated Sales Projection (USD Millions):
| Year | Projected Sales (USD Millions) |
|---|---|
| 2023 | 65 - 75 |
| 2024 | 50 - 60 |
| 2025 | 35 - 45 |
| 2026 | 25 - 35 |
| 2027 | 15 - 25 |
These projections are estimates based on market trends and competitive analysis. Actual sales may vary.
By 2027, branded SOLODYN is expected to represent a small fraction of its historical peak sales. Its remaining sales will likely be sustained by a niche segment of the market that continues to prescribe the brand for specific reasons, or in markets where generic availability is less robust or where Bausch Health maintains strong market access.
The overall market for extended-release minocycline will continue, but the revenue will accrue primarily to the generic manufacturers.
Key Takeaways
- SOLODYN (minocycline extended-release) has experienced a significant decline in sales following patent expiration and the subsequent entry of multiple generic competitors.
- The primary driver for this decline is the availability of lower-cost generic extended-release minocycline, which offers bioequivalent treatment.
- The broader acne treatment market is highly competitive, featuring topical agents, other oral antibiotics, systemic isotretinoin, and hormonal therapies, further fragmenting market share.
- Continued emphasis on antibiotic stewardship is likely to reduce overall reliance on oral antibiotics for acne management.
- Branded SOLODYN sales are projected to continue a downward trend, with revenue expected to diminish substantially over the next five years as generics solidify their market position.
Frequently Asked Questions
-
When did the main patent for SOLODYN expire? The primary patent for SOLODYN's extended-release formulation, U.S. Patent No. 5,976,573, was issued on November 2, 1999. While patent term extensions and regulatory exclusivities could delay generic entry, the period of significant market exclusivity largely concluded in the mid-to-late 2010s, with generic versions entering the market around 2016-2017.
-
What are the main advantages of extended-release minocycline over immediate-release formulations for acne? Extended-release formulations aim to provide more consistent plasma concentrations of the drug over a 24-hour period. This can potentially lead to improved patient adherence due to once-daily dosing, and in some cases, a more favorable side effect profile compared to immediate-release versions which can cause sharper peaks and troughs in drug levels.
-
Are there any new developments or pipeline drugs that could impact the market for minocycline-based acne treatments? The acne treatment market is dynamic, with ongoing research into novel therapies. While there are no direct pipeline drugs directly competing with generic extended-release minocycline's specific formulation, advancements in topical agents, anti-inflammatory compounds, and microbiome-targeting therapies are continuously emerging, offering alternative treatment pathways that may reduce reliance on oral antibiotics.
-
What is the typical pricing difference between branded SOLODYN and generic extended-release minocycline? Following generic entry, the price of branded SOLODYN is typically significantly higher than its generic counterparts. While exact pricing varies based on pharmacy, insurance, and dosage, generic extended-release minocycline is often available at a fraction of the cost of the branded product, commonly representing a 70-90% price reduction.
-
How does antibiotic resistance affect the use of minocycline for acne? Concerns about antibiotic resistance are a significant factor influencing the prescribing of all oral antibiotics for acne, including minocycline. Dermatologists are increasingly encouraged to limit the duration of oral antibiotic therapy and to prioritize topical treatments or non-antibiotic systemic options, especially for long-term management. This trend can reduce the overall demand for minocycline products.
Citations
[1] American Academy of Dermatology Association. (n.d.). Acne. Retrieved from https://www.aad.org/media/stats-skin-conditions [2] Valeant Pharmaceuticals International, Inc. (2014). SOLODYN (minocycline extended-release) prescribing information. Retrieved from [link to a relevant archived PI if available, otherwise general reference to FDA drug labels]. (Note: Specific product labeling links can change; general reference used here).
More… ↓
