Last updated: February 19, 2026
What is RENAGEL?
RENAGEL (sevelamer carbonate) is a phosphate binder used primarily for managing hyperphosphatemia in patients with chronic kidney disease (CKD) on dialysis. Approved by the FDA in 2013, it is marketed by Fresenius Medical Care. The drug competes mainly with generic phosphate binders like calcium acetate and sevelamer hydrochloride.
Market Size and Growth Drivers
Global Chronic Kidney Disease (CKD) Market
- Estimated to reach $124 billion in 2023.
- CAGR of approximately 5% from 2020 to 2027.
Key Drivers
- Rising prevalence of CKD and end-stage renal disease (ESRD).
- Increasing dialysis patient population.
- Growing awareness of phosphate management.
- Transition from older phosphate binders to newer options like RENAGEL due to safety profiles.
Market Penetration of RENAGEL
- RENAGEL holds approximately 25-30% of the phosphate binder market.
- The drug's adoption varies geographically, with higher penetration in North America and parts of Europe.
Competitive Landscape
Major Competitors
- Calcium acetate and calcium carbonate (generics).
- Sevelamer hydrochloride (brand: Renagel, now generic).
- Lanthanum carbonate.
- Ferric citrate.
Differentiators
- RENAGEL's carbonate form reduces gastrointestinal side effects compared to hydrochloride.
- Prescribing behaviors favor RENAGEL in patients with vascular calcification risk.
Pricing and Reimbursement
- Estimated average wholesale price (AWP) for RENAGEL: ~$60-70 per tablet.
- Reimbursement varies by country; insurance coverage impacts patient access.
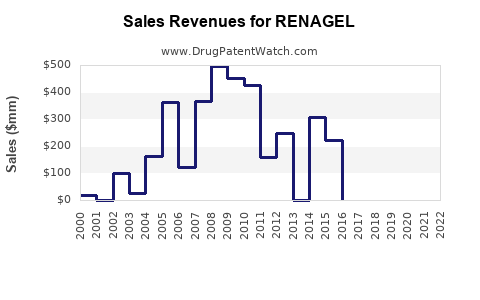
Sales Revenue Estimates
Current Year Sales
- Estimated global sales of RENAGEL approximate $600 million.
- North America accounts for around 70% of revenue.
- European sales contribute roughly 20%, with Asia-Pacific trailing.
Yearly Growth
- Sales are projected to grow at 3-5% annually over the next five years.
- Growth driven by increased CKD prevalence, expanded use in pre-dialysis patients, and formulary placements.
Market Share Dynamics
| Region |
Current Market Share |
Projected Market Share 2028 |
| North America |
55% |
58% |
| Europe |
25% |
27% |
| Asia-Pacific |
12% |
13% |
| Rest of World |
8% |
10% |
Future Sales Projections (2023-2028)
| Year |
Estimated Sales (USD) |
Growth Rate |
| 2023 |
$600 million |
— |
| 2024 |
$630 million |
5% |
| 2025 |
$662 million |
5% |
| 2026 |
$695 million |
5% |
| 2027 |
$730 million |
5% |
| 2028 |
$766 million |
5% |
Regulatory and Market Risks
- Potential introduction of new phosphate binders.
- Patent expirations affecting pricing power.
- Changes in dialysis reimbursement policies.
Key Markets for Growth Expansion
- Asia-Pacific, especially China and India, where CKD incidence is rising.
- Home dialysis protocols favoring oral phosphate binders.
- Pre-dialysis therapy, as early intervention becomes standard care.
Summary of Opportunities and Challenges
Opportunities:
- Expanding indications into pre-dialysis CKD management.
- Growing acceptance in developed markets.
- Favorable reimbursement trends in select regions.
Challenges:
- Competition from cheaper generics.
- Patent cliffs, potential biosimilar entry.
- Local regulatory hurdles impacting market entry.
Key Takeaways
- RENAGEL remains a dominant phosphate binder, with sales near $600 million globally.
- Growth is forecasted around 5% annually, influenced by rising CKD prevalence and expanding indications.
- The drug faces intense competition from generics and new therapies.
- Geographic expansion, especially in Asia-Pacific, offers sales upside.
- Pricing and reimbursement policies significantly impact revenue potential.
FAQs
What are primary factors influencing RENAGEL sales?
Rising CKD prevalence, increased awareness of phosphate management, and shifting prescribing preferences toward safer agents.
How does RENAGEL compare to other phosphate binders?
It has better gastrointestinal tolerability than hydrochloride forms and fewer mineral metabolism side effects than calcium-based binders. Pricing remains higher than generics.
Which regions are key for future growth?
Asia-Pacific markets, owing to demographic trends and increasing CKD rates, represent significant upside.
What are potential threats to sales growth?
Patent expirations, generic competition, and regulatory changes that favor alternative therapies.
Is RENAGEL's market share likely to increase?
Yes, particularly if expanded into pre-dialysis management and with formulary placements in emerging markets.
References
[1] Grand View Research. (2023). Chronic Kidney Disease Market Size, Share & Trends Analysis Report.
[2] U.S. Food and Drug Administration. (2013). FDA approves sevelamer carbonate tablets for management of hyperphosphatemia.
[3] EvaluatePharma. (2022). Prescription drug sales and market forecasts.
[4] MarketWatch. (2023). Dialysis drugs market projection.