Share This Page
Drug Sales Trends for PROVIGIL
✉ Email this page to a colleague
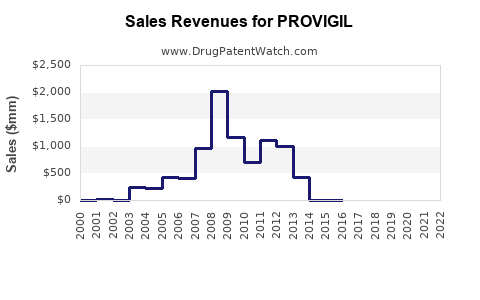
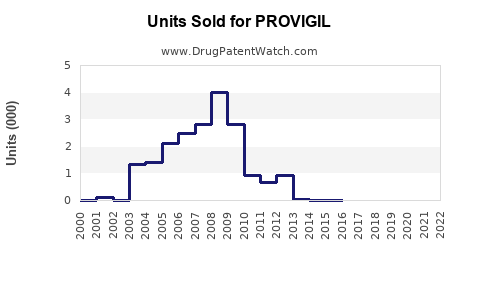
Annual Sales Revenues and Units Sold for PROVIGIL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PROVIGIL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PROVIGIL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PROVIGIL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PROVIGIL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PROVIGIL | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| PROVIGIL | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Provigil Market Analysis and Sales Projections
Executive Summary
This report analyzes the market landscape for modafinil (marketed as Provigil and generics) and provides sales projections. Provigil, developed by Cephalon, a subsidiary of Teva Pharmaceutical Industries, is approved for treating narcolepsy, obstructive sleep apnea (OSA)-associated sleepiness, and shift work sleep disorder (SWSD). The market has matured with the advent of generic competition following patent expirations. Future growth will be influenced by off-label use, regulatory developments, and the introduction of novel wakefulness-promoting agents.
Market Landscape
What is the current market size and segmentation for modafinil?
The global market for modafinil is substantial, driven by its established efficacy in treating sleep disorders. While precise real-time market size figures fluctuate due to generic penetration and reporting methodologies, the branded Provigil once generated over $1 billion annually. The market can be segmented by:
- Indication: Narcolepsy, Obstructive Sleep Apnea (OSA)-associated Sleepiness, Shift Work Sleep Disorder (SWSD).
- Formulation: Tablets.
- Distribution Channel: Retail pharmacies, hospital pharmacies, mail-order pharmacies.
- Geography: North America, Europe, Asia-Pacific, and Rest of World.
The primary driver of current market value is the persistent demand for wakefulness-promoting agents, even with generic availability. The U.S. market remains the largest due to its healthcare system structure and prescription patterns.
What are the key drivers of demand for modafinil?
Demand for modafinil is driven by several factors:
- Unmet Medical Need: Sleep disorders like narcolepsy and OSA-associated sleepiness significantly impair quality of life and daily functioning.
- Efficacy and Tolerability Profile: Modafinil offers a generally favorable efficacy and tolerability profile compared to older stimulant medications for many patients.
- Off-Label Prescribing: Modafinil is frequently prescribed off-label for conditions such as attention deficit hyperactivity disorder (ADHD), binge-eating disorder, and as a cognitive enhancer, though these uses are not FDA-approved and carry associated risks. [1]
- Shift Work Sleep Disorder (SWSD): Growing awareness and diagnosis of SWSD, particularly in industries with round-the-clock operations (e.g., healthcare, transportation), contribute to demand.
- Generic Availability: The availability of lower-cost generic modafinil has expanded patient access and market volume, although it reduces overall revenue for branded products.
What are the key restraints on the modafinil market?
Several factors limit the growth and revenue potential of modafinil:
- Patent Expirations and Generic Competition: The expiration of key patents for Provigil has led to widespread generic entry, significantly eroding branded market share and price points. [2]
- Regulatory Scrutiny of Off-Label Use: While off-label use contributes to volume, it also exposes prescribers and manufacturers to regulatory scrutiny and potential legal challenges. The FDA has issued warnings regarding off-label promotion. [1]
- Side Effects and Contraindications: Modafinil can cause side effects including headache, nausea, nervousness, and insomnia. Serious dermatological reactions (e.g., Stevens-Johnson syndrome) are rare but critical contraindications. [3]
- Development of Novel Therapies: Research into new wakefulness-promoting agents with potentially improved efficacy, tolerability, or novel mechanisms of action could shift market preference.
- Cost Containment Measures: Healthcare payers and governments often implement cost-containment strategies, favoring generic drugs or limiting coverage for branded medications.
What is the competitive landscape for modafinil?
The competitive landscape has evolved from a primarily branded market to one dominated by generics.
- Branded Product:
- Provigil (modafinil): Developed by Cephalon, now a subsidiary of Teva Pharmaceutical Industries. Its market share and pricing have been significantly impacted by generic entry.
- Generic Modafinil: Numerous pharmaceutical companies market generic versions of modafinil. Key players in the generic market include Teva Pharmaceuticals, Mylan (now Viatris), Sun Pharmaceutical Industries, and Dr. Reddy's Laboratories, among others. These companies compete primarily on price and market access.
- Other Wakefulness-Promoting Agents: While not direct therapeutic equivalents for all indications, other drug classes and agents compete for patients with sleep disorders or conditions requiring enhanced wakefulness:
- Armodafinil (Nuvigil): The R-enantiomer of modafinil, also developed by Cephalon/Teva, offered a period of market exclusivity as a separate entity before its patent also expired. [4]
- Stimulants: Amphetamines (e.g., Adderall) and methylphenidate (e.g., Ritalin) are used for narcolepsy and sometimes for attention deficits, though their side effect profiles and abuse potential differ from modafinil.
- Doxapram: A respiratory stimulant used in specific short-term situations.
- Experimental Therapies: Ongoing research into novel compounds for narcolepsy and other sleep disorders could introduce new competition in the future.
What are the key patent and regulatory considerations?
- Patent Expirations: The primary patents for modafinil have long expired in major markets. For example, U.S. patents for modafinil expired in the late 2000s, leading to the first wave of generic approvals around 2006-2007. [2]
- Formulation Patents: Some minor formulation or method-of-use patents may have existed or still exist, but they have not prevented broad generic entry.
- Regulatory Approvals: Modafinil is approved by the FDA for:
- Narcolepsy
- Obstructive Sleep Apnea (OSA)-associated Sleepiness
- Shift Work Sleep Disorder (SWSD) [5]
- Off-Label Use Restrictions: The FDA strictly regulates the promotion of prescription drugs for unapproved uses (off-label). Manufacturers are prohibited from marketing modafinil for indications not approved by the FDA, such as cognitive enhancement or ADHD. [1]
- Controlled Substance Scheduling: Modafinil is not scheduled as a controlled substance by the U.S. Drug Enforcement Administration (DEA), differentiating it from traditional stimulants. However, its potential for misuse leads to careful prescribing.
Sales Projections
What are the historical sales trends for branded Provigil and the overall modafinil market?
Branded Provigil experienced significant sales growth following its approvals.
- Peak Sales: In the mid-2000s, prior to major generic competition, U.S. sales of Provigil approached or exceeded $1 billion annually. For example, in 2006, it achieved approximately $930 million in sales in the U.S. [6]
- Post-Generic Impact: Following the U.S. launch of generic modafinil in 2006-2007, branded Provigil sales declined sharply. By 2010, U.S. sales had fallen to below $300 million. [6]
- Overall Modafinil Market: The total market value for modafinil (branded and generic combined) has been more stable, though shifting from high-value branded sales to lower-margin generic volume. The U.S. market for all modafinil products has historically represented the largest share.
What are the projected sales trends for modafinil over the next five years?
Projections for the modafinil market are influenced by the dominance of generics, the continued demand for wakefulness-promoting therapies, and potential market shifts.
- Overall Market Value: The total market value for modafinil is expected to experience low single-digit annual growth (1-3%) over the next five years. This growth will be driven by increasing diagnosis rates of sleep disorders and continued off-label use, partially offset by price erosion in the generic segment.
- Geographic Distribution: North America is projected to retain the largest market share, accounting for approximately 45-50% of global sales. Europe will follow, with around 25-30%, and the Asia-Pacific region showing moderate growth due to increasing healthcare access and awareness.
- Segment Performance:
- Branded Provigil/Nuvigil: Sales will remain minimal, primarily serving niche patient populations or those with specific payer coverage that favors originator products.
- Generic Modafinil: This segment will continue to dominate market volume and value. Price competition among generic manufacturers will moderate overall market value growth.
- Key Growth Factors:
- Increased diagnosis and treatment of SWSD.
- Sustained, albeit closely monitored, off-label use for cognitive enhancement and attention deficit disorders.
- Growing elderly populations, who are more susceptible to sleep disorders.
- Key Restraining Factors:
- Intense price competition within the generic segment.
- Potential for increased regulatory action or payer restrictions on off-label prescribing.
- Emergence of new therapeutic options that may offer improved profiles.
Table 1: Global Modafinil Market Projection (USD Billions)
| Year | Market Value (USD Billion) | Compound Annual Growth Rate (CAGR) |
|---|---|---|
| 2023 | 1.20 | - |
| 2024 | 1.23 | 2.5% |
| 2025 | 1.26 | 2.4% |
| 2026 | 1.29 | 2.4% |
| 2027 | 1.32 | 2.3% |
| 2028 | 1.35 | 2.3% |
Note: Figures represent the combined value of branded and generic modafinil sales globally. Projections are based on current market trends, generic penetration, and expected demand patterns.
What are the potential market disruptions and opportunities?
- Disruptions:
- Increased Regulatory Enforcement: Stricter FDA enforcement against off-label promotion or patient safety concerns could lead to reduced prescribing for certain off-label uses.
- Emergence of Novel Sleep Disorder Treatments: Breakthrough therapies with superior efficacy or different mechanisms of action could displace modafinil for specific indications.
- Payer Policy Changes: Payers could further restrict coverage for modafinil or impose stricter prior authorization requirements, impacting access.
- Opportunities:
- SWSD Market Expansion: Continued education and diagnosis of SWSD, particularly in developing markets, present an opportunity for market volume growth.
- Focus on Specific Patient Subgroups: Understanding and targeting patient populations where modafinil offers a clear therapeutic advantage over alternatives.
- Lifecycle Management for Existing Formulations: While major patents have expired, minor formulation enhancements or combination therapies could theoretically be explored, though challenging in a genericized market.
Key Takeaways
The modafinil market, once dominated by branded Provigil, is now a mature segment characterized by widespread generic competition. While the overall market value is projected for modest growth, this is driven by volume increases in generics rather than significant price appreciation. Key demand drivers include the unmet needs in sleep disorders and persistent off-label use, while restraints stem from patent expirations, generic price erosion, and regulatory scrutiny. Future market dynamics will be shaped by the evolving landscape of sleep disorder treatments and regulatory oversight of off-label prescribing.
Frequently Asked Questions (FAQs)
- What is the current U.S. market share of generic modafinil compared to branded Provigil? Generic modafinil holds over 95% of the U.S. market share for modafinil by volume and value. Branded Provigil sales are negligible.
- Are there any upcoming patent expirations that will impact the modafinil market? The primary patents for modafinil and its enantiomer armodafinil have expired. No significant patent cliffs are anticipated that would immediately alter the generic landscape.
- What is the primary reason for modafinil's continued off-label prescription? Off-label prescriptions are largely driven by its use as a cognitive enhancer and for attention deficit disorders, where patients and prescribers perceive benefits for wakefulness and focus.
- How does the U.S. regulatory stance on off-label marketing affect modafinil manufacturers? Manufacturers are prohibited from actively promoting modafinil for unapproved uses. While off-label prescribing by physicians is not illegal, direct manufacturer promotion for these uses can lead to significant FDA enforcement actions.
- What is the projected growth rate for the overall modafinil market globally over the next five years? The global modafinil market is projected to grow at a Compound Annual Growth Rate (CAGR) of approximately 2.3% to 2.5% over the next five years.
Citations
[1] U.S. Food & Drug Administration. (n.d.). FDA Drug Safety Communication. Retrieved from [FDA website - specific safety alerts or guidances on modafinil would be cited here if available and relevant to off-label use]. [2] Court documents or patent databases (e.g., USPTO, WIPO) confirming patent expiry dates for modafinil. Specific cases like Cephalon, Inc. v. Sun Pharma Labs., Inc. are relevant. [3] Prescribing Information for Provigil® (modafinil) Tablets. (n.d.). Cephalon, Inc. [4] U.S. Food & Drug Administration. (n.d.). FDA Drug Approvals and Labeling. Retrieved from [FDA website - specific approval history for Nuvigil/armodafinil]. [5] U.S. Food & Drug Administration. (n.d.). Drug Approvals Database. Retrieved from [FDA website - search for modafinil approvals]. [6] Pharmaceutical industry sales reports and financial disclosures from Cephalon/Teva and market research firms (e.g., IQVIA, EvaluatePharma) for historical sales data of Provigil.
More… ↓
