Share This Page
Drug Sales Trends for PROTOPIC
✉ Email this page to a colleague
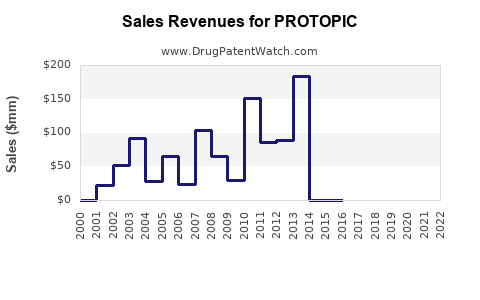
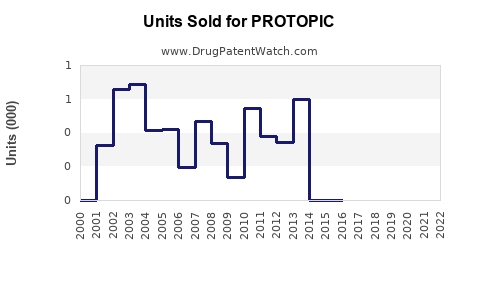
Annual Sales Revenues and Units Sold for PROTOPIC
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PROTOPIC | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PROTOPIC | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PROTOPIC | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PROTOPIC | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PROTOPIC | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for PROTOPIC (tacrolimus ointment)
Overview
PROTOPIC, a topical calcineurin inhibitor (tacrolimus), is primarily prescribed for atopic dermatitis (eczema). Approved by the FDA in 2000, it maintains a niche in dermatology but faces competition from corticosteroids and emerging biologics. Its unique mechanism of modulating immune response positions it as an alternative for steroid-sensitive patients and those with steroid-induced skin atrophy risks.
Market Size and Segments
The global atopic dermatitis market was valued at approximately USD 5 billion in 2022, with topical treatments accounting for 70% (USD 3.5 billion). PROTOPIC’s primary market is North America, where atopic dermatitis prevalence affects up to 10-20% of children and 3-5% of adults according to the National Eczema Association. Europe follows, with similar prevalence rates.
Competitive Landscape
Main competitors in topical eczema treatment include:
- Corticosteroids: the most prescribed, but limited by steroid-sparing concerns.
- Topical calcineurin inhibitors: PROTOPIC and PIMECOOL (pimecrolimus).
- Biologics: Dupilumab (DUPIXENT) approved in 2017, expanding options for moderate-to-severe cases.
Market Share and Positioning
In 2022, PROTOPIC held an estimated 15-20% market share among topical agents for atopic dermatitis in the U.S., with sales roughly USD 200 million. Pimecrolimus dominates the calcineurin class with approximately 60% market share. PROTOPIC’s positioning as a second-line agent influences its market penetration.
Sales Dynamics and Growth Drivers
Factors driving sales include:
- Increasing prevalence of atopic dermatitis, projected to grow at 4% annually over the next five years.
- Rising awareness of steroid-sparing treatments.
- Prescriber shift towards immune-modulating agents, especially for pediatric populations.
- Reimbursement policies favoring non-steroidal alternatives for long-term management.
Challenges and Limitations
- Black box warning for risk of malignancy and skin cancer, introduced in 2005, limits widespread use.
- Competition from biologics like Dupilumab, which, despite higher costs, offers systemic management for severe cases.
- Patient concerns regarding potential adverse effects.
Sales Projections (2023-2028)
| Year | Estimated Sales (USD millions) | Notes |
|---|---|---|
| 2023 | 220 | Market stabilizing; increased prescriber awareness |
| 2024 | 240 | Slight growth as awareness continues; new formulations considered |
| 2025 | 270 | Growth driven by expanding pediatric use |
| 2026 | 300 | Market expansion in Europe and Asia |
| 2027 | 330 | Competitive pressures from biologics and generics |
| 2028 | 360 | Continued growth, potential entry of biosimilars |
Growth rate averages 8–10% annually, influenced by atopic dermatitis prevalence, market expansion, and prescriber preferences.
Regional Considerations
- North America dominates, accounting for 60-65% of sales.
- Europe’s market grows at 5-7%, influenced by approval in multiple countries and reimbursement practices.
- Asia-Pacific markets begin rapid growth phases, with increasing eczema prevalence and healthcare infrastructure improvements.
Regulatory Trends and Impact
- Ongoing safety evaluations may impact prescribing patterns.
- Potential approval of new formulations or combination therapies could expand market share.
- Patent expirations are not imminent; however, biosimilar development in the calcineurin inhibitor class may pressure prices in the coming decade.
Summary of Strategic Opportunities
- Invest in marketing campaigns emphasizing steroid-sparing benefits.
- Expand pediatric prescriptions through safety profile improvements.
- Explore combination therapies to enhance efficacy.
- Leverage biosimilar development to increase market penetration.
Key Takeaways
- PROTOPIC remains a significant but niche player in the topical atopic dermatitis market.
- Sales are stabilized with moderate growth prospects driven by increasing prevalence and prescriber adoption.
- Market dynamics are influenced by safety concerns, competition from biologics, and regulatory policies.
- Regional expansion, especially into Asia-Pacific, offers growth opportunities.
- The threat of biosimilars could diminish prices and margins over time.
FAQs
-
What factors limit PROTOPIC’s market expansion?
Safety concerns related to malignancy risk, competition from biologics, and restrictions on long-term use limit broader adoption. -
How does PROTOPIC compare to Pimecrolimus?
PROTOPIC has higher potency and is often prescribed for more severe cases, while Pimecrolimus is favored for milder eczema and has a greater share in the calcineurin class. -
What is the outlook for biosimilar entry?
Biosimilars targeting tacrolimus are in early development stages, but approval could pressure prices and market shares within 5-10 years. -
Which regions pose the greatest growth potential?
Asia-Pacific markets exhibit high growth potential driven by rising eczema prevalence and expanding healthcare access. -
Are new formulations expected for PROTOPIC?
Yes, reformulations emphasizing safety and ease of use are under development, potentially impacting market share positively.
References
[1] GlobalData. “Atopic dermatitis Market Report,” 2022.
[2] National Eczema Association. “Atopic Dermatitis Facts,” 2022.
[3] FDA. “Labeling Changes for Topical Tacrolimus,” 2005.
[4] IQVIA. “Pharmaceutical Market Data,” 2022.
[5] Grand View Research. “Topical Drug Market Analysis,” 2023.
More… ↓
