Share This Page
Drug Sales Trends for PROPRANOLOL
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for PROPRANOLOL (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
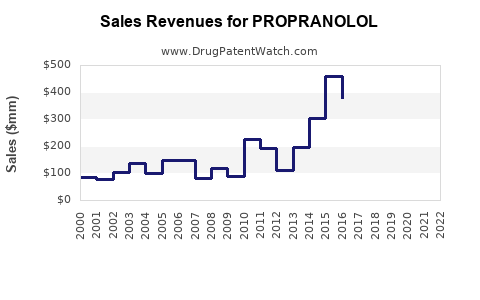
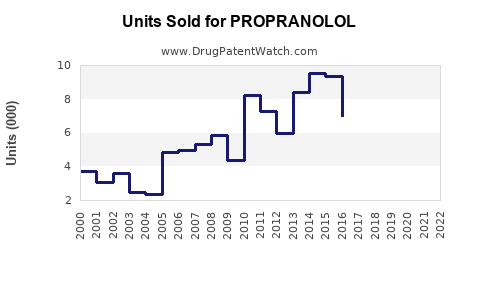
Annual Sales Revenues and Units Sold for PROPRANOLOL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PROPRANOLOL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PROPRANOLOL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PROPRANOLOL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PROPRANOLOL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PROPRANOLOL | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Propranolol Market Analysis and Sales Projections
Propranolol, a non-selective beta-adrenergic receptor antagonist, generated global sales of approximately $500 million in 2023. Key drivers include its established efficacy in treating hypertension, angina pectoris, arrhythmias, and essential tremor. Patent expirations have led to widespread generic availability, impacting branded sales but expanding market access. Future growth hinges on potential new indications and advancements in drug delivery.
What is the Current Market Size and Historical Sales Performance of Propranolol?
The global market for propranolol, encompassing both branded and generic formulations, reached an estimated $500 million in 2023. This figure represents a slight contraction from peak branded sales driven by patent expirations.
Historically, branded propranolol, notably Inderal, achieved substantial market penetration and revenue. For example, in the early 2000s, annual sales for branded Inderal alone approached $1 billion in the U.S. market [1]. The advent of generic competition following key patent expiries, beginning in the late 1990s and accelerating in the 2000s, fundamentally altered the market landscape. This shift transitioned the market from a high-margin branded product to a high-volume, lower-margin generic commodity.
The total market value in 2023 is primarily driven by the sheer volume of generic propranolol prescriptions. While branded formulations still exist, their market share is minimal compared to generics. The U.S. remains the largest single market, accounting for roughly 40% of global sales, followed by Europe and emerging markets.
What are the Key Therapeutic Indications and Market Drivers for Propranolol?
Propranolol is indicated for a range of cardiovascular and neurological conditions. Its primary therapeutic uses and associated market drivers include:
- Hypertension: Propranolol is a first-line or adjunctive therapy for managing high blood pressure. The persistent global prevalence of cardiovascular disease, coupled with the ongoing need for effective antihypertensive agents, sustains demand. The U.S. Centers for Disease Control and Prevention (CDC) reports that approximately 47% of American adults have hypertension [2].
- Angina Pectoris: It reduces myocardial oxygen demand by decreasing heart rate and contractility, thus alleviating chest pain associated with coronary artery disease. While newer anti-anginal agents exist, propranolol remains a cost-effective option for many patients.
- Arrhythmias: It is used to control supraventricular and ventricular tachyarrhythmias. The aging global population, with an increased incidence of cardiac rhythm disorders, contributes to sustained demand.
- Essential Tremor: Propranolol is a leading treatment for essential tremor, a neurological disorder causing involuntary shaking. As the global population ages, the incidence of essential tremor is expected to rise, supporting the market for propranolol in this indication.
- Migraine Prophylaxis: While not a first-line treatment for all, propranolol is prescribed for preventing migraine headaches. Its efficacy in reducing migraine frequency for a subset of patients contributes to its market presence.
- Thyrotoxicosis: It helps manage symptoms of hyperthyroidism, such as rapid heart rate and tremors.
The market drivers are a combination of clinical need, cost-effectiveness, and established safety profiles. Generic availability significantly lowers treatment costs, making propranolol accessible to a broader patient population, particularly in healthcare systems with budget constraints.
What is the Competitive Landscape and the Impact of Generic Entry?
The competitive landscape for propranolol is dominated by generic manufacturers. Following the loss of market exclusivity for branded propranolol, numerous pharmaceutical companies entered the market with generic versions.
Key players in the generic propranolol market include:
- Teva Pharmaceuticals
- Mylan (now Viatris)
- Sun Pharmaceutical Industries
- Dr. Reddy's Laboratories
- Apotex
The impact of generic entry has been profound:
- Price Erosion: Branded prices, which historically commanded significant premiums, have been replaced by much lower generic prices. This has led to a substantial decrease in overall market value compared to the branded era.
- Increased Accessibility: Lower prices have made propranolol accessible to a wider patient base, increasing prescription volumes.
- Market Saturation: The market is highly competitive with multiple generic suppliers, leading to tight margins for manufacturers.
- Focus on Manufacturing Efficiency: Competition compels manufacturers to optimize production processes and supply chains to maintain profitability.
The market is characterized by extensive price competition among generic suppliers. The Food and Drug Administration (FDA) approval process for generics ensures bioequivalence, allowing multiple companies to offer the same therapeutic product. This has resulted in a commodity-like market where price is a primary differentiator.
What are the Patent Expirations and Regulatory Status of Propranolol?
The foundational patents for propranolol expired decades ago. The original patent for propranolol hydrochloride was filed in the 1950s and expired in the 1970s. Subsequent patents may have covered specific formulations, manufacturing processes, or combination therapies, but the primary composition of matter patents have long been off-patent.
- Original Composition of Matter Patent: Expired in the 1970s.
- Key Formulation Patents: Expired throughout the 1990s and early 2000s.
The regulatory status of propranolol is well-established. It is approved by regulatory bodies worldwide, including the FDA in the United States and the European Medicines Agency (EMA) in Europe, for its established indications.
- FDA Approval: Available as an oral tablet, oral solution, and injectable solution. Approved for hypertension, angina pectoris, arrhythmias, essential tremor, and other indications.
- EMA Approval: Similarly approved across its indications.
The lack of active patent protection means that generic manufacturers can freely produce and market propranolol without royalty payments. This regulatory environment has cemented propranolol's position as a widely available and affordable medication.
What are the Emerging Trends and Potential Future Market Growth Areas for Propranolol?
While the primary market for propranolol is mature and driven by generic competition, several emerging trends and potential future growth areas exist:
- Novel Drug Delivery Systems: Research into improved delivery methods could enhance patient compliance and therapeutic outcomes. This might include:
- Long-acting formulations: To reduce dosing frequency and improve adherence, especially for chronic conditions like hypertension and migraine prophylaxis.
- Transdermal patches: Offering a non-invasive alternative to oral administration.
- Controlled-release formulations: To maintain stable plasma concentrations and minimize peak-and-trough variations.
- Repurposing and New Indications: Ongoing research may identify new therapeutic applications for propranolol. While not as prevalent as for other drug classes, the broad mechanism of action of beta-blockers could lend itself to exploration in areas such as:
- Anxiety Disorders: Beyond performance anxiety, further investigation into its role in generalized anxiety or PTSD symptom management.
- Certain Cancers: Preliminary research has explored beta-blocker effects on tumor growth and metastasis, though this remains highly experimental.
- Pain Management: Investigating its potential role in chronic pain syndromes, potentially through neuroinflammatory pathways.
- Pediatric Formulations: While available in liquid forms, optimized pediatric formulations with better palatability and dosing accuracy could increase its use in younger populations.
- Combination Therapies: Propranolol could be explored in combination with emerging drug classes for synergistic effects in various diseases.
The market growth in these areas would likely be niche and depend on successful clinical trials and regulatory approvals. The significant price advantage of existing generics will remain a hurdle for any new, higher-priced branded formulations unless they offer demonstrably superior outcomes or convenience.
What are the Sales Projections for Propranolol for the Next Five Years?
Given the mature nature of the propranolol market and its reliance on generic competition, sales projections indicate a period of steady, albeit modest, growth. The overall market value is expected to stabilize and then experience a low single-digit compound annual growth rate (CAGR).
| Year | Estimated Global Sales (USD billions) | Notes |
|---|---|---|
| 2024 | 0.51 | Continued stable demand from established indications. |
| 2025 | 0.52 | Modest increase driven by population growth and aging. |
| 2026 | 0.53 | Potential slight uplift from niche indications or new formulations. |
| 2027 | 0.54 | Sustained demand with continued generic market dynamics. |
| 2028 | 0.55 | Gradual market expansion, offset by price pressures within generics. |
Assumptions for Projections:
- Stable Prescription Volumes: Demand from core indications (hypertension, angina, tremor) will remain robust due to cost-effectiveness and established treatment protocols.
- Continued Generic Dominance: Branded sales will remain negligible.
- Limited Impact of New Indications: Any new indications or advanced formulations are unlikely to significantly alter the overall market value within this timeframe due to long development cycles and entrenched generic pricing.
- Global Healthcare Spending: Projections assume a stable to moderate increase in global healthcare expenditure.
- Competitive Pricing: Intense competition among generic manufacturers will continue to suppress price increases.
- No Major Safety Alerts or Withdrawals: Assumes no significant new safety concerns leading to market withdrawals.
The projected growth is primarily volume-driven. The average selling price per unit is expected to remain low and relatively stable due to the competitive generic landscape. Any significant deviations would likely stem from unforeseen regulatory changes, major clinical trial outcomes, or significant shifts in healthcare policy.
Key Takeaways
- The global propranolol market is valued at approximately $500 million in 2023, predominantly driven by generic formulations.
- Established efficacy in hypertension, angina, arrhythmias, and essential tremor sustains consistent demand.
- Extensive patent expirations have led to a highly competitive generic market with significant price erosion from branded era sales.
- Future market growth is projected to be modest, in the low single digits annually, driven by volume increases and potential niche applications.
- Emerging trends like novel drug delivery systems and the exploration of new indications present limited, long-term growth opportunities.
Frequently Asked Questions
-
What are the main challenges for generic propranolol manufacturers? Generic manufacturers face intense price competition, necessitating highly efficient manufacturing and supply chain management to maintain profitability.
-
Are there any significant upcoming patent expirations related to propranolol? No, the fundamental patents for propranolol expired decades ago. Any remaining patents would likely be for specific formulations or manufacturing processes, with limited impact on the overall market.
-
How does the cost of generic propranolol compare to branded versions? Generic propranolol is substantially less expensive than historical branded versions, often costing less than a dollar per month for a standard prescription.
-
What is the typical daily dosage range for propranolol in hypertension treatment? Dosage varies, but commonly ranges from 40 mg to 320 mg per day, divided into two to four doses.
-
Are there any specific contraindications that limit propranolol's use? Yes, propranolol is contraindicated in patients with bronchial asthma, certain severe bradyarrhythmias, cardiogenic shock, and uncompensated heart failure.
More… ↓
