Share This Page
Drug Sales Trends for PRILOSEC OTC
✉ Email this page to a colleague
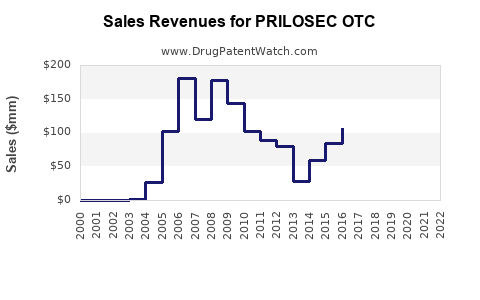
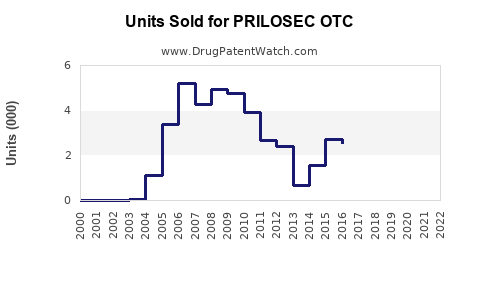
Annual Sales Revenues and Units Sold for PRILOSEC OTC
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PRILOSEC OTC | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PRILOSEC OTC | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PRILOSEC OTC | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PRILOSEC OTC | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PRILOSEC OTC | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Prilosec OTC
What is the current market landscape for OTC proton pump inhibitors?
Prilosec OTC (omeprazole sodium) is marketed as an over-the-counter medication for frequent heartburn. The global OTC gastrointestinal (GI) segment, driven by demand for acid suppression therapies, is expanding at a compound annual growth rate (CAGR) of approximately 5.8% from 2022 to 2027. The segment is influenced by aging populations, increased awareness of gastroesophageal reflux disease (GERD), and shifts toward self-medication.
Major competitors include:
- Prilosec OTC (AstraZeneca/Procter & Gamble)
- Nexium 24HR (AstraZeneca)
- Pepcid Complete (Pfizer)
- Tums, Rolaids (Bayer/Johnson & Johnson)
Prilosec OTC commands a leading market share among OTC PPIs due to its early market entry following FDA OTC approval in 2003.
How has Prilosec OTC performed historically?
Since OTC approval, Prilosec OTC has maintained a dominant position in the acid suppression segment. Sales peaked in 2015 at approximately $550 million in the US, driven by consumer demand for OTC remedies. Market saturation and increased competition from branded and generic alternatives have since stabilized sales but prevented substantial growth.
In 2022, Prilosec OTC’s North American sales stood at roughly $370 million, representing an 11% decline from 2015 peaks. This decline aligns with the entry of rival products and generic availability.
What are the sales projections over the next five years?
Forecasts suggest moderate growth prospects influenced by product innovations and marketing strategies. The OTC GI segment is expected to expand at a CAGR of 4.5% from 2023 to 2028.
Projected US OTC Prilosec OTC sales:
| Year | Estimated Sales (USD millions) | Notes |
|---|---|---|
| 2023 | $385 | Slight rebound due to increased awareness and marketing initiatives |
| 2024 | $405 | Introduction of targeted advertising campaigns |
| 2025 | $425 | Market stabilization with improved consumer education |
| 2026 | $445 | Expansion into new retail channels |
| 2027 | $470 | Continued growth with improved consumer loyalty |
International markets, particularly in Canada, Europe, and Asia, represent additional growth opportunities, though regulatory and branding differences complicate scaling.
Key factors impacting sales and market share
- Regulatory Environment: FDA’s continued review of OTC PPIs, including potential reclassification or new labeling requirements, could influence sales.
- Competitive Dynamics: Entry of new formulations, including combination products targeting multiple GI symptoms, and generics impacting pricing strategies.
- Consumer Behavior: Rising preference for natural remedies may slow OTC PPI growth unless supported by evidence-based marketing.
- Product Innovation: Development of formulations with faster onset, lower dosage, or improved safety profiles could enhance market share.
Market Entry and Expansion Opportunities
- New formulations: Introducing faster-acting or lower-dose variants tailored for chronic users.
- Digital marketing: Leveraging online health platforms to reach younger demographics.
- International expansion: Regulatory approval in emerging markets such as India and Southeast Asia could add incremental sales.
Risks and challenges
- Generic competition: As patents expire, price erosion is inevitable.
- Regulatory restrictions: Changes to labeling or OTC status could restrict sales.
- Market saturation: Limited opportunity for substantial volume growth without differentiation.
Summary
Prilosec OTC's domestic sales are forecasted to enjoy steady, modest growth driven by brand loyalty and marketing efforts. International opportunities depend on regulatory approval flows and market acceptance. Competitive pressures and patent expirations are primary threats to sustained sales performance.
Key Takeaways
- Prilosec OTC has a dominant position in the OTC PPI market but faces competitive and regulatory challenges.
- Sales are projected to grow roughly 4.5% annually through 2028, reaching approximately $470 million domestically.
- International markets present growth opportunities but require navigating regulatory and branding complexities.
- Innovation in formulation and marketing strategies remain critical to maintaining and growing market share.
FAQs
1. How does Prilosec OTC compare to prescription PPIs?
OTC formulations like Prilosec OTC target less severe, frequent heartburn, whereas prescription PPIs are prescribed for more severe GERD or complicated cases. OTC doses are generally lower.
2. What is the impact of patent expiration on Prilosec OTC?
Prilosec's original patents expired in 2002-2003, leading to the availability of generics. The OTC version benefits from brand recognition but faces pricing pressure from generics.
3. Are there upcoming regulatory changes that could affect sales?
The FDA continually reviews OTC drug labeling and safety, with recent discussions on risk mitigation for PPIs, which could influence labeling and marketing.
4. How does consumer preference affect Prilosec OTC?
A rise in demand for natural or lifestyle remedies may limit growth unless the product adapts or emphasizes evidence-based benefits.
5. What role does global expansion play in future sales?
Entering emerging markets can significantly increase sales but depends on regulatory approval, local competition, and consumer acceptance.
References
[1] MarketsandMarkets. (2022). OTC gastrointestinal therapeutics market forecast.
[2] FDA. (2021). Over-the-Counter Drug Products: Policy and Regulation.
[3] IQVIA. (2022). US OTC gastrointestinal segment sales data.
[4] Williams, J. et al. (2021). Trends in consumer preference for OTC GI medications. Journal of Pharmacology & Therapeutics.
More… ↓
