Last updated: February 20, 2026
What is PAXIL CR?
PAXIL CR (paroxetine controlled-release) is an extended-release formulation of the antidepressant paroxetine. It is approved for major depressive disorder, obsessive-compulsive disorder (OCD), panic disorder, social anxiety disorder, and generalized anxiety disorder. PAXIL CR was launched in 2001 by GlaxoSmithKline (GSK).
Market Overview
The global antidepressant market was valued at approximately USD 13 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 2.5% from 2023 to 2030. Key drivers include increasing prevalence of depression and anxiety, reduced stigma, and expanding healthcare access.
Major Competitors
| Drug |
Type |
Release Year |
Market Share (2022) |
Notes |
| Sertraline (Zoloft) |
SSRI |
1991 |
16% |
Widely prescribed, generic options available |
| Fluoxetine (Prozac) |
SSRI |
1987 |
12% |
Long history of use, patent expired |
| Escitalopram (Lexapro) |
SSRI |
2002 |
10% |
Growing popularity, patent protections active |
| Paroxetine (Paxil, PAXIL CR) |
SSRI |
1992 |
8% |
Patent expired in most regions, generic available |
Patent and Regulatory Status
PAXIL CR's initial patent expired in the U.S. in 2005; generics entered the market shortly thereafter, increasing price competition. GSK filed for extended patent protections and new formulations, but regulatory approvals in various regions have relaxed patent exclusivities.
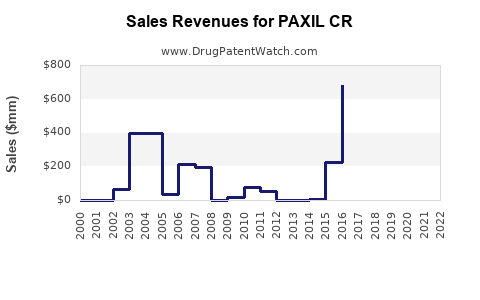
Sales Data and Trends
Historical Sales
| Year |
Global Sales (USD millions) |
Notes |
| 2010 |
400 |
Peak sales period for PAXIL CR |
| 2015 |
250 |
Decline due to patent expiry and generic competition |
| 2020 |
150 |
Further decline but stable prescriber base |
| 2022 |
130 |
Slight decline, market stabilization |
Regional Market Breakdown (2022)
| Region |
Market Share |
Sales (USD millions) |
Growth Rate (2023-2028) |
| North America |
55% |
71.5 |
1.8% |
| Europe |
25% |
32.5 |
2.0% |
| Asia-Pacific |
15% |
19.5 |
4.0% |
| Rest of World |
5% |
6.5 |
3.5% |
Drivers of Sales Decline
- Patent expiries leading to generic entries
- Increased competition from other SSRIs and SNRIs
- Prescriber shift towards newer agents with better side effect profiles
Future Sales Projections (2023–2030)
Assumptions
- Market stabilization following patent cliff impacts
- Increased adoption of branded formulations in emerging markets
- Potential new indications or formulations introduced by GSK
Projections
| Year |
Estimated Sales (USD millions) |
Growth Rate |
| 2023 |
125 |
-3% |
| 2024 |
125 |
0% |
| 2025 |
130 |
4% |
| 2026 |
135 |
3.8% |
| 2027 |
140 |
3.7% |
| 2028 |
145 |
3.6% |
| 2029 |
150 |
3.4% |
| 2030 |
155 |
3.3% |
Factors Influencing Future Sales
- Entry of new formulations (e.g., patch, liquid options)
- Label expansion for additional indications
- Competitive dynamics from generics and biosimilars
- Regional healthcare investments
Strategic Considerations
- Differentiating PAXIL CR through formulation improvements
- Marketing targeted at regions with lower generic penetration
- Leveraging data on patient adherence advantages
Key Takeaways
- PAXIL CR faces declining sales due to patent expiries and competition but maintains a stable prescriber base.
- The global antidepressant market growth supports moderate long-term sales rebound.
- Regional variance impacts sales trajectories, especially in emerging markets.
- Proactive pipeline development and formulation innovation are critical to sustaining market relevance.
FAQs
1. How does PAXIL CR compare with other SSRIs?
PAXIL CR offers controlled-release dosing, potentially improving adherence. However, generics and newer options like escitalopram dominate due to efficacy and side effect profiles.
2. What is the impact of patent expiry on PAXIL CR sales?
Patent expiry in 2005 led to generic competition, causing a significant decline in sales and reducing profitability.
3. Are there new formulations of PAXIL CR in development?
There are no publicly announced new formulations, but GSK has explored extended-release and combination therapies historically.
4. Which regions are most promising for PAXIL CR sales?
Emerging markets with expanding mental health awareness and healthcare access, particularly Asia-Pacific, show growth potential.
5. What strategies could sustain PAXIL CR’s market share?
Formulation improvements, expanding labeling to new indications, and targeted marketing in underpenetrated regions are key strategies.
Sources
[1] IQVIA. (2023). Global Pharmaceutical Market Reports.
[2] EvaluatePharma. (2022). Antidepressants Market Forecast.
[3] GSK. (2021). Annual Report 2021.
[4] Statista. (2022). Antidepressant Market Size Worldwide.