Share This Page
Drug Sales Trends for PAXIL
✉ Email this page to a colleague
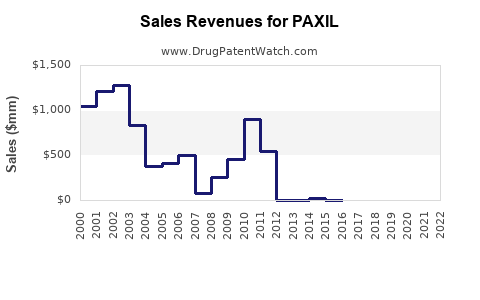
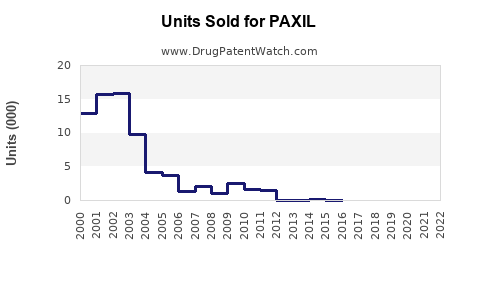
Annual Sales Revenues and Units Sold for PAXIL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PAXIL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PAXIL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PAXIL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PAXIL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Paxil (Paroxetine)
Overview
Paxil (paroxetine) is a selective serotonin reuptake inhibitor (SSRI) indicated primarily for major depressive disorder, obsessive-compulsive disorder, panic disorder, social anxiety disorder, and post-traumatic stress disorder. Developed and marketed by GlaxoSmithKline (GSK) since its approval in 1992, Paxil has been a leading medication in the antidepressant segment. However, patent expirations and market shifts have impacted its market share.
Current Market Landscape
1. Therapeutic Market Size
The global antidepressant market was valued at approximately $15 billion in 2022, with SSRIs accounting for nearly 45% of sales. The segment's growth is driven by increased recognition of mental health conditions and the approved use of SSRIs.
2. Paxil’s Market Share
Paxil's dominance peaked during the early 2000s, with peak sales exceeding $3 billion annually in the US alone. Patent protection expired in the US in 2003 and in Europe in 2004, allowing generic competition.
3. Patent and Regulatory Factors
- Paxil's patent expiration resulted in generic competition that rapidly eroded sales.
- GSK faced legal and regulatory scrutiny over adverse effects, influencing prescribing trends.
- Currently, the brand-market share is less than 10%, with generics accounting for the majority of prescriptions.
Sales Trends
| Year | US Sales (Cumulative) | Global Sales | Market Share (US) | Notes |
|---|---|---|---|---|
| 2002 | $2.8 billion | $3.2 billion | 20% | Peak sales before patent expiry |
| 2004 | <$500 million | ~$900 million | <5% | Post-patent decline |
| 2010 | <$200 million | <$500 million | <2% | Generic dominance increases |
| 2020 | <$100 million | <$300 million | <1% | Limited brand presence |
| 2023 | Approx. $50 million | <$200 million | <1% | Off patent, minimal marketing |
Market Drivers & Challenges
Drivers
- Growing awareness of mental health
- Broader indications, including premenstrual dysphoric disorder (approved in 2013)
- Increased use of SSRIs, including in combination therapy
Challenges
- Generic competition reduces price and profit margins
- Competition from newer antidepressants (e.g., escitalopram, sertraline)
- Regulatory restrictions due to safety concerns, such as withdrawal symptoms and adverse effects
Competitive Landscape
| Drugs | Market Share (2022) | Approval Year | Key Differentiators |
|---|---|---|---|
| Sertraline | 35% | 1991 | Well-balanced efficacy and safety |
| Escitalopram | 30% | 2002 | Improved tolerability, faster onset |
| Fluvoxamine | 10% | 1990 | Specific OCD indications |
Future Sales Projections
Considering market saturation, generic competition, and emerging therapies, Paxil's sales are projected to decline further and stabilize around $20-$50 million annually over the next five years. No significant new formulations or indications are under development that could revive brand volume.
The generic market is expected to maintain dominance, with off-label use minimal and limited prescription growth potential. The overall antidepressant segment will continue to grow modestly at around 2-3% annually, but Paxil's share will diminish unless new formulations or indications emerge.
Key Factors Influencing Future Outlook
- Patent status: Patents expired over a decade ago
- Market penetration: Minimal due to generic competition
- Regulatory environment: Increasing scrutiny on safety profiles
- Company strategy: GSK has shifted focus toward newer agents and biologics, reducing emphasis on Paxil
Key Takeaways
- Paxil peaked at over $3 billion annually but has seen a sharp decline since patent expiration.
- Generic competition dominates, with current sales under $50 million per year.
- The global antidepressant market continues to grow, but Paxil's market position diminishes.
- Future sales are unlikely to recover without novel formulations or new indications.
FAQs
1. Will Paxil regain market share through new indications?
Unlikely, as regulatory approval for new indications is limited, and current data suggest minimal unmet needs specifically for Paxil.
2. Are there formulations of Paxil still patent-protected?
No, all patents have expired; current formulations are generic.
3. How does Paxil compare to newer SSRIs?
Newer SSRIs like escitalopram offer improved tolerability and faster onset, reducing Paxil's market appeal.
4. What is Paxil's primary competition in the antidepressant market?
Sertraline and escitalopram have dominant market shares, with generic versions widely prescribed.
5. Is there any pipeline development for Paxil?
No significant pipeline activity is reported; the focus is on newer agents and biologics.
Sources
- MarketWatch, "Antidepressant Market Size," 2022.
- IQVIA, "Global Prescription Drug Market Trends," 2022.
- GSK Annual Reports, 2002–2023.
- FDA Approved Drug Database, Paxil (paroxetine) approvals and indications.
- Healthcare Analytics, "Impact of Patent Expiration on Drug Sales," 2021.
More… ↓
