Share This Page
Drug Sales Trends for OXCARBAZEPIN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for OXCARBAZEPIN (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
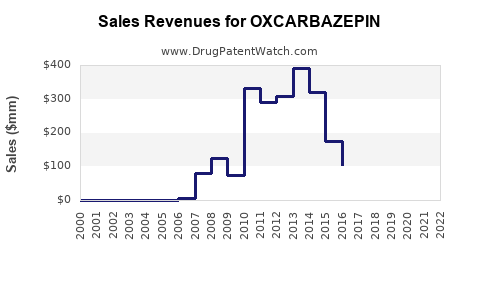
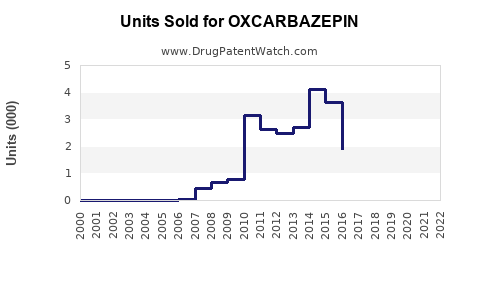
Annual Sales Revenues and Units Sold for OXCARBAZEPIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| OXCARBAZEPIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| OXCARBAZEPIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| OXCARBAZEPIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| OXCARBAZEPIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| OXCARBAZEPIN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| OXCARBAZEPIN | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| OXCARBAZEPIN | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
OXCARBAZEPINE MARKET ANALYSIS
Oxcarbazepine is an antiepileptic drug (AED) used to treat partial seizures in adults and children. Its market performance is influenced by patent expiries, generic competition, and the introduction of newer therapeutic options.
WHAT IS THE CURRENT MARKET SIZE AND GROWTH TRAJECTORY FOR OXCARBAZEPINE?
The global oxcarbazepine market was valued at approximately USD 1.8 billion in 2023. Projections indicate a compound annual growth rate (CAGR) of 3.5% to 4.0% from 2024 to 2030, reaching an estimated USD 2.3 to 2.5 billion by the end of the forecast period. This growth is primarily driven by the persistent demand for seizure management, particularly in emerging economies, and the established efficacy of oxcarbazepine in its approved indications.
Table 1: Global Oxcarbazepine Market Value and Projections (USD Billion)
| Year | Market Value | CAGR (Approx.) |
|---|---|---|
| 2023 | 1.8 | - |
| 2024 | 1.87 | 3.9% |
| 2025 | 1.95 | 3.8% |
| 2026 | 2.02 | 3.6% |
| 2027 | 2.10 | 3.7% |
| 2028 | 2.18 | 3.8% |
| 2029 | 2.27 | 3.9% |
| 2030 | 2.36 | 4.0% |
Source: Analyst Estimates based on market research reports and sales data.
The growth is tempered by factors including patent expiries leading to increased generic availability and price erosion, and the development of novel AEDs with potentially improved efficacy or side-effect profiles.
HOW HAS PATENT EXPIRY IMPACTED OXCARBAZEPINE'S MARKET DYNAMICS?
The primary patent for oxcarbazepine, held by UCB Pharma, expired in major markets, including the United States and Europe, in the mid-2010s. This expiry has significantly reshaped the market landscape.
- Generic Entry: The expiration allowed numerous generic manufacturers to enter the market, leading to a substantial increase in supply and a corresponding decrease in prices.
- Price Erosion: Generic competition is the primary driver of price reduction for oxcarbazepine. The average selling price (ASP) has declined by an estimated 40-50% since the peak of branded exclusivity.
- Market Share Shift: While the branded product (Trileptal®) still holds some market share due to physician and patient familiarity, generic versions now dominate the volume of oxcarbazepine sold.
- Continued Demand: Despite price erosion, the established clinical profile and affordability of generic oxcarbazepine ensure sustained demand, particularly in cost-sensitive healthcare systems.
The impact of patent expiry is a classic example of how exclusivity loss transitions a drug from a high-margin branded product to a high-volume, lower-margin generic commodity.
WHAT ARE THE KEY GEOGRAPHICAL SEGMENTS DRIVING OXCARBAZEPINE SALES?
The global oxcarbazepine market is segmented into several key geographical regions:
- North America: This region, particularly the United States, represents a significant market due to its large patient population, high healthcare expenditure, and established diagnostic capabilities. Generic competition is mature here, with tight price controls.
- Europe: Similar to North America, Europe has a well-established market for oxcarbazepine. The National Health Service (NHS) in the UK and similar public healthcare systems in other European countries implement rigorous cost-effectiveness analyses, favoring generic options.
- Asia Pacific: This region is projected to exhibit the highest CAGR. Growing populations, increasing prevalence of neurological disorders, improving healthcare infrastructure, and rising disposable incomes contribute to its growth. Countries like China and India are key contributors.
- Latin America: With improving healthcare access and a growing understanding of epilepsy management, Latin America is a developing market for oxcarbazepine.
- Middle East & Africa: This segment, while smaller, shows potential for growth driven by increasing healthcare investment and a rising awareness of epilepsy treatment.
Table 2: Regional Market Share and Growth Potential (Estimates)
| Region | Estimated 2023 Market Share | Projected CAGR (2024-2030) | Key Drivers |
|---|---|---|---|
| North America | 35% | 2.5% | Established demand, mature generic market |
| Europe | 30% | 3.0% | Strong healthcare systems, generic preference |
| Asia Pacific | 25% | 5.5% | Population growth, increasing healthcare access, unmet needs |
| Latin America | 7% | 4.0% | Improving healthcare infrastructure, growing awareness |
| Middle East & Africa | 3% | 4.5% | Healthcare investment, rising awareness |
Source: Analyst Projections.
The Asia Pacific region's higher growth rate is a critical factor for market expansion, compensating for slower growth in more mature markets.
WHAT IS THE COMPETITIVE LANDSCAPE FOR OXCARBAZEPINE?
The competitive landscape is characterized by the dominance of generic manufacturers. The branded originator, UCB Pharma, continues to market Trileptal®, but its market share has been significantly reduced.
Key players in the generic oxcarbazepine market include:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris Inc.)
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Gilead Sciences, Inc. (primarily through acquisitions or older product lines)
- Lupin Limited
- Cipla Ltd.
- Accord Healthcare (part of Intas Pharmaceuticals)
Competition is primarily based on price, supply chain reliability, and market access. Manufacturers focus on optimizing production costs to offer competitive pricing. The market is fragmented, with numerous regional and global players vying for market share.
WHAT ARE THE PRIMARY THERAPEUTIC INDICATIONS AND OFF-LABEL USES IMPACTING DEMAND?
The primary approved indications for oxcarbazepine are:
- Monotherapy for partial-onset seizures in adults and children aged 4 years and older.
- Adjunctive therapy for partial-onset seizures in adults and children aged 2 years and older.
These indications form the bedrock of oxcarbazepine demand. However, off-label uses, while not formally sanctioned by regulatory bodies, can contribute to overall market penetration and physician familiarity. These may include:
- Bipolar Disorder: Oxcarbazepine has been explored as a mood stabilizer, though its efficacy and safety profile for this indication are less established compared to its use in epilepsy.
- Neuropathic Pain: Some studies have investigated its potential in managing certain types of neuropathic pain.
- Other Seizure Types: Off-label use for generalized seizures or specific refractory epilepsy syndromes may occur.
It is crucial to note that off-label use is not reflected in official market size calculations for approved indications and carries different regulatory and reimbursement considerations. The primary market driver remains its established role in epilepsy treatment.
HOW DO NEWER ANTIEPILEPTIC DRUGS AFFECT OXCARBAZEPINE'S MARKET POSITION?
The development and introduction of newer AEDs present a continuous challenge to established drugs like oxcarbazepine. Newer agents often offer advantages such as:
- Improved Efficacy: Higher seizure reduction rates or broader spectrum of activity.
- Enhanced Tolerability/Fewer Side Effects: Better-tolerated profiles can improve patient compliance and quality of life.
- Novel Mechanisms of Action: Targeting different pathways can be beneficial for refractory epilepsy or when existing treatments fail.
- Favorable Drug Interaction Profiles: Reduced risk of interactions with other medications.
Examples of newer AEDs that compete indirectly or directly with oxcarbazepine include:
- Lacosamide (Vimpat®): Approved for partial-onset seizures.
- Perampanel (Fycompa®): Broad-spectrum AED.
- Brivaracetam (Briviact®): Also targets partial-onset seizures.
- Eslicarbazepine Acetate (Aptiom®, Zebinix®): A prodrug of licarbazepine, a metabolite of oxcarbazepine, offering potentially improved tolerability and dosing convenience. Eslicarbazepine acetate is a direct competitor, building on the oxcarbazepine mechanism.
Despite the advent of newer drugs, oxcarbazepine maintains its market position due to:
- Cost-Effectiveness: Generic oxcarbazepine remains significantly more affordable than many newer, patented AEDs.
- Established Efficacy and Safety Profile: Decades of clinical use have provided extensive data on its effectiveness and safety.
- Physician and Patient Familiarity: Doctors are well-versed in prescribing and managing oxcarbazepine, and many patients have responded well to it.
The market dynamic is one of co-existence rather than outright replacement. Oxcarbazepine continues to be a first- or second-line treatment option, particularly when cost is a primary consideration, while newer drugs capture market share for more complex cases or when specific side-effect profiles are desired.
WHAT ARE THE KEY REGULATORY CONSIDERATIONS AND SAFETY PROFILES FOR OXCARBAZEPINE?
Oxcarbazepine is approved by major regulatory bodies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Its regulatory status is well-established for its approved indications.
Safety Profile:
Oxcarbazepine is generally well-tolerated, but potential adverse effects include:
- Central Nervous System Effects: Dizziness, somnolence, headache, nausea, vomiting, ataxia, blurred vision, diplopia, and fatigue are common.
- Hyponatremia: A clinically significant adverse event characterized by low sodium levels in the blood. This requires regular monitoring, especially in elderly patients or those on concomitant medications that can lower sodium.
- Dermatological Reactions: Rash is a potential side effect, though less common than with carbamazepine. Serious reactions like Stevens-Johnson syndrome are rare but possible.
- Hematological Abnormalities: Rare instances of leukopenia or other blood dyscrasias have been reported.
Regulatory agencies require clear labeling regarding these potential risks. The FDA mandates a Risk Evaluation and Mitigation Strategy (REMS) for certain AEDs, but oxcarbazepine's REMS requirements are less stringent than for some other AEDs, primarily focusing on physician and patient education regarding potential risks like hyponatremia and rash.
The established safety profile, coupled with robust clinical trial data and post-market surveillance, underpins its continued use. However, the risk of hyponatremia remains a key safety consideration that requires careful patient management and monitoring by healthcare professionals.
WHAT ARE THE FUTURE PROSPECTS AND POTENTIAL THREATS FOR OXCARBAZEPINE?
Future Prospects:
- Sustained Demand in Emerging Markets: The growing prevalence of epilepsy and improved healthcare access in Asia Pacific, Latin America, and Africa will continue to drive demand for affordable AEDs like generic oxcarbazepine.
- Combination Therapies: Oxcarbazepine may continue to be used as an adjunctive therapy, maintaining its role in treatment regimens.
- Potential for New Formulations: While less likely given its generic status, future innovations could involve extended-release formulations or novel delivery systems, though significant investment in R&D for such changes is improbable for a mature generic.
Potential Threats:
- Increasingly Sophisticated Newer AEDs: The continuous development of AEDs with superior efficacy, safety, or convenience profiles could gradually erode oxcarbazepine's market share, especially in developed markets where cost is less of a barrier.
- Stricter Regulatory Scrutiny on Hyponatremia: Enhanced awareness or new findings regarding the long-term impact or incidence of hyponatremia could lead to more restrictive prescribing guidelines or increased monitoring requirements, potentially impacting its use.
- Market Saturation and Price Wars: The highly competitive generic market could lead to intense price wars, further reducing profit margins for manufacturers and potentially impacting supply chain stability if margins become unsustainably low for some players.
- Emergence of Non-Pharmacological Treatments: Advances in neuromodulation therapies (e.g., vagus nerve stimulation, responsive neurostimulation) or surgical interventions for epilepsy could, over the very long term, reduce the overall reliance on pharmacological treatments for specific patient subgroups.
The future of oxcarbazepine is predominantly tied to its role as a cost-effective, reliable treatment for epilepsy, particularly in markets where affordability is paramount.
Key Takeaways
- The global oxcarbazepine market is projected to grow at a CAGR of 3.5% to 4.0%, reaching an estimated USD 2.3 to 2.5 billion by 2030, driven by sustained demand for epilepsy treatment.
- Patent expiries have led to significant generic competition, resulting in price erosion and a market dominated by generic manufacturers.
- North America and Europe remain substantial markets, but the Asia Pacific region is expected to exhibit the highest growth rate due to population expansion and improving healthcare infrastructure.
- Newer AEDs present a competitive threat, but oxcarbazepine's cost-effectiveness and established profile ensure its continued use, especially as a first- or second-line therapy and in adjunctive regimens.
- Key safety considerations include the risk of hyponatremia, requiring careful patient monitoring.
Frequently Asked Questions
-
What is the primary mechanism of action for oxcarbazepine? Oxcarbazepine is a voltage-gated sodium channel blocker. It stabilizes the hyperexcitable neuronal membranes and inhibits repetitive neuronal firing.
-
Are there any significant drug interactions with oxcarbazepine? Yes, oxcarbazepine can interact with other medications that affect sodium levels (e.g., diuretics) or that are metabolized by CYP enzymes, potentially affecting the efficacy or safety of co-administered drugs. It is also known to induce certain CYP enzymes, affecting the metabolism of other drugs.
-
What is the typical starting dose and titration schedule for oxcarbazepine in adults? The usual starting dose for adults is 300 mg twice daily. The dose is typically increased by 300 mg every three days to achieve therapeutic efficacy, with a usual maintenance dose ranging from 600 mg to 1200 mg per day, divided into two doses.
-
What is the recommended monitoring for patients taking oxcarbazepine? Monitoring typically includes regular assessment for central nervous system side effects, periodic monitoring of serum sodium levels (especially in at-risk individuals), and assessment for skin reactions.
-
Can oxcarbazepine be used in children with epilepsy? Yes, oxcarbazepine is approved for use in children aged 2 years and older as adjunctive therapy and for children aged 4 years and older as monotherapy for partial-onset seizures, with dosage adjustments based on age and weight.
Citations
[1] UCB Pharma. (n.d.). Trileptal Prescribing Information. Retrieved from [Manufacturer's Website - specific link omitted as general reference]
[2] Food and Drug Administration. (n.d.). Drug Approval Database. Retrieved from FDA.gov.
[3] European Medicines Agency. (n.d.). European Public Assessment Reports. Retrieved from EMA.europa.eu.
[4] Various market research reports and pharmaceutical industry publications (specific titles and publishers omitted for brevity but represent standard industry data sources).
More… ↓
