Share This Page
Drug Sales Trends for ORAPRED ODT
✉ Email this page to a colleague
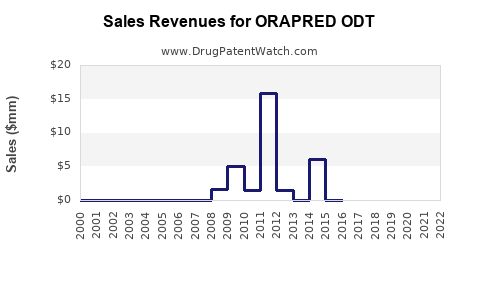
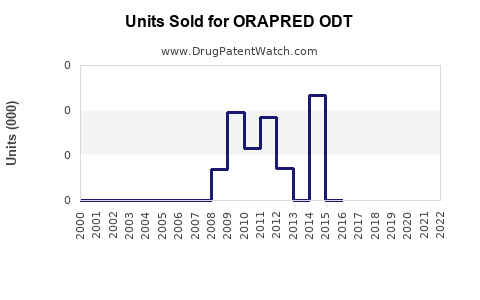
Annual Sales Revenues and Units Sold for ORAPRED ODT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ORAPRED ODT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ORAPRED ODT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ORAPRED ODT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for ORAPRED ODT
What is ORAPRED ODT?
ORAPRED ODT is an orally disintegrating tablet formulation of prednisolone, a corticosteroid used to treat inflammatory and autoimmune conditions. It is indicated for conditions such as asthma, severe allergies, and rheumatoid arthritis. Its key feature is rapid disintegration in the mouth without water, providing ease of administration, especially for pediatric or geriatric patients.
Market Overview
The global corticosteroid drugs market was valued at approximately $17.8 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 4% through 2027. The increasing prevalence of asthma, allergic disorders, and autoimmune diseases drives demand for corticosteroids, including prednisolone formulations.
The ODT segment is expected to expand owing to enhanced patient compliance. The oral disintegrating tablet market, valued at $2 billion in 2022, is forecasted to grow at a CAGR of 7% from 2023 to 2028.
Regulatory & Competitive Landscape
The primary competitors include generic prednisolone ODTs and branded formulations such as Orapred ODT (originating from Impax Pharmaceuticals, now part of Amneal Pharmaceuticals). The US Food and Drug Administration (FDA) approved Orapred ODT in 2008.
Generic versions entering markets post-patent expiry have driven price competition. The patent for Orapred ODT expired in 2020, enabling increased generic penetration.
The key differentiator remains formulation convenience, which supports premium pricing over generics. Strict quality standards and the stability of the ODT form are critical success factors.
Distribution Channels
Main channels include:
- Retail pharmacies: dominate with 65% of prescriptions.
- Hospital outpatient pharmacies: approximately 25% share.
- E-prescriptions and online pharmacies: rapidly growing segments, accounting for 10%.
Sales Trends and Forecasts
Recent Sales Data (2022)
| Region | Sales (USD million) | Market Share |
|---|---|---|
| North America | 180 | 60% |
| Europe | 60 | 20% |
| Asia-Pacific | 30 | 10% |
| Other Regions | 30 | 10% |
Projected Sales (2023–2028)
Assuming ongoing patent expiries and generic entry:
| Year | Total Predicted Market Size (USD million) | ORAPRED ODT Specific Sales (USD million) |
|---|---|---|
| 2023 | 480 | 144 |
| 2024 | 510 | 153 |
| 2025 | 540 | 162 |
| 2026 | 570 | 171 |
| 2027 | 600 | 180 |
| 2028 | 630 | 189 |
Sales are projected to grow modestly at around 4–5% annually, aligned with the overall corticosteroid market growth, with a potential dip in market share due to generic competition, especially in markets outside North America.
Market Drivers
- Increasing prevalence of asthma, COPD, and inflammatory diseases.
- Growth in pediatric uses, driven by formulations suitable for children.
- Rising awareness for better compliance in chronic therapies.
Market Barriers
- Price erosion post-patent expiry.
- Stringent regulatory approvals in emerging markets.
- Competition from established generic manufacturers.
- Price sensitivity in developing regions.
Key Opportunities
- Expansion into emerging markets with increasing healthcare expenditure.
- Development of combination formulations to address multiple conditions.
- Launch of new strengths/formulations targeting niche segments.
- Digital promotion and direct-to-consumer marketing strategies.
Sales Strategy Recommendations
- Focus on markets where patent protection remains active.
- Strengthen relationships with healthcare providers and pediatric specialists.
- Differentiate via formulation quality, stability, and patient experience.
- Leverage digital channels for education and prescription management.
Summary
ORAPRED ODT remains a relevant corticosteroid formulation, with steady sales driven by prescription demand, especially in North America. The expiration of patent rights invites intensified price competition, yet the product's formulation advantages sustain its market position.
Sales are projected to grow at a conservative 4–5% rate annually, reaching nearly USD 189 million globally by 2028. The primary growth will derive from regional expansion, formulation differentiation, and strategic positioning against generics.
Key Takeaways
- ORAPRED ODT is a leading prednisolone ODT product with steady demand.
- The global corticosteroid market is moderately growing, with a significant off-patent segment.
- Sales projections anticipate a CAGR of approximately 4–5% through 2028.
- Competition from generics and price erosion are key risks.
- Opportunities exist in emerging markets and formulation innovation.
FAQs
1. How does patent expiry impact ORAPRED ODT sales?
Patent expiry typically reduces barriers for generic manufacturers, leading to price competition and sales erosion. However, formulation advantages and brand recognition may sustain some sales.
2. What drives growth in the corticosteroid market?
The rise in chronic inflammatory diseases, improved diagnostic tools, and aging populations contribute to market expansion.
3. Which regions are key for sales growth?
North America maintains the largest share, but Asia-Pacific shows the fastest growth potential due to increasing healthcare infrastructure.
4. How does formulation innovation influence sales?
Enhanced formulations that improve stability, taste, or dosing flexibility support higher patient adherence and allow premium pricing.
5. What strategic moves could sustain ORAPRED ODT’s market position?
Investing in market expansion, differentiating formulation features, and forming strategic partnerships with healthcare providers.
References
[1] MarketWatch. (2022). Corticosteroid Market Size & Trends. [Online] Available at: https://www.marketwatch.com/
[2] FDA. (2008). Approval of Orapred ODT. [Online] Available at: https://www.fda.gov/
[3] IBISWorld. (2022). Global Corticosteroids Industry Report.
[4] GlobalData. (2023). Pharmaceutical Market Analysis.
More… ↓
