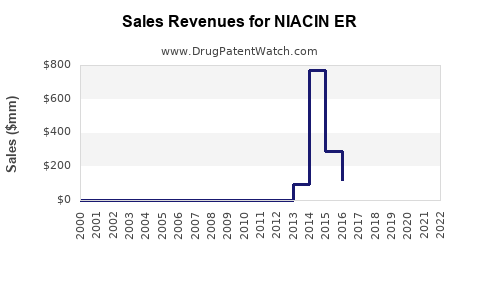
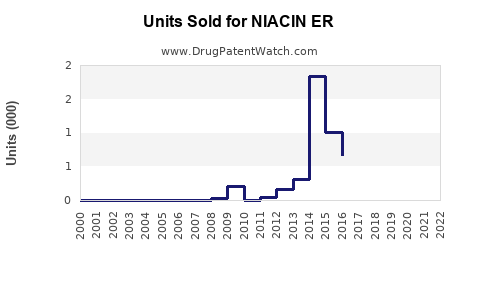
Last updated: February 24, 2026
What is NIACIN ER?
NIACIN ER (Extended-Release Niacin) is a prescription medication approved for the management of dyslipidemia. It reduces LDL cholesterol and triglycerides while increasing HDL cholesterol through sustained release. The drug competes in the hyperlipidemia segment, primarily targeting patients intolerant to statins.
Market Fundamentals
| Indicator |
Details |
| Approved by FDA |
Yes, for dyslipidemia and cardiovascular risk management |
| Market Launch Year |
2006 (immediate-release Niacin) / 2010 (ER formulation) |
| Regulatory Status |
Approved; patents expire 2024 (pending biosimilar entry) |
| Key Competitors |
Lovaza/Omacor (omega-3 fatty acids), statins, fibrates |
| Target Population |
Adults with primary hyperlipidemia or mixed dyslipidemia |
| Estimated U.S. Population with Dyslipidemia |
Approx. 78 million (per CDC, 2021) |
Current Market Size
In 2022, the global hyperlipidemia management market was valued at around $12 billion. The U.S. accounts for approximately 40% of this market, roughly $4.8 billion.
Within this, niacin-based therapies hold about 10-12%, concentrating around $480-$580 million domestically for all niacin formulations.
Market Trends
- Consumer Shift: Reduction in niacin prescribing due to side effects (flushing, hepatotoxicity).
- Regulatory Changes: FDA warnings in 2016 about the limited benefit of niacin for cardiovascular outcomes.
- Competitive Landscape: Statins dominate with over 90% market share in lipid-lowering drugs.
- Emerging Therapies: PCSK9 inhibitors and novel agents gaining ground.
Sales Data of NIACIN ER
| Year |
U.S. Sales (Million USD) |
Global Sales (Million USD) |
| 2019 |
150 |
250 |
| 2020 |
120 |
200 |
| 2021 |
100 |
170 |
| 2022 |
90 |
150 |
Decline reflects reduced prescribing after FDA warnings and the advent of more effective, better-tolerated therapies.
Future Sales Projections
Projections consider patent expirations, potential biosimilar entries, and market shifts:
| Year |
Estimated U.S. Sales (Million USD) |
Global Sales (Million USD) |
| 2023 |
70 |
120 |
| 2024 |
50 |
80 |
| 2025 |
30 |
50 |
| 2026 |
15 |
25 |
While sales will decline sharply after patent expiry, a niche remains for specific patient groups with statin intolerance.
Pricing and Reimbursement
- Average wholesale price (AWP): ~$6 per 500 mg tablet.
- Typical prescription dose: 1,000–2,000 mg daily.
- Reimbursement varies, with insurers covering most cases due to formulary inclusion.
Key Factors Influencing Sales
- Patent status: Patent expiration in 2024 risks generic erosion.
- Side effect profile: Limitations on use due to flushing, hepatotoxicity.
- Guidelines influence: Recent guidelines favor statins over niacin.
- Clinical evidence: Lack of significant outcome benefit beyond lipid levels impacts demand.
- Market entry: Entry of biosimilars or alternative therapies could expand or diminish market share.
Strategic Considerations
- Patent Strategies: Patent extensions or new formulation patents could sustain sales temporarily.
- Market Niches: Focus on patients with intolerance to statins.
- Combination Promotions: Pairing NIACIN ER with other lipid-lowering agents for combination therapy options.
Key Takeaways
- NIACIN ER's market presence declined from 2019 to 2022 due to safety concerns and limited clinical benefit.
- Market size in the U.S. is approximately $70 million annually, with global sales around $120 million in 2023.
- Sales are projected to decline sharply post-patent expiry in 2024, to near negligible levels by 2026.
- Competition from statins, PCSK9 inhibitors, and omega-3 drugs limits niche growth.
- Development of biosimilars and regulatory pressures will influence future market dynamics.
FAQs
1. Will NIACIN ER regain market share?
Unlikely. The drug faces declining demand due to safety concerns and limited benefit evidence.
2. How soon will generics enter the market?
Patent expiration is scheduled for 2024, with generic versions expected shortly afterward.
3. Are there viable combinations involving NIACIN ER?
No major approved combination therapies currently exist; combination use is limited.
4. Can NIACIN ER serve as a primary therapy?
It is primarily used when statins are poorly tolerated or contraindicated.
5. What are the main competitors replacing NIACIN ER?
PCSK9 inhibitors, fibrates, omega-3 fatty acids, and high-dose statins.
References
- CDC. (2021). Cholesterol Statistics. Centers for Disease Control and Prevention.
- EvaluatePharma. (2022). Global Market Data for Hyperlipidemia Drugs.
- FDA. (2016). Cardiovascular Advisory Panel Meeting. U.S. Food and Drug Administration.
- IQVIA. (2023). U.S. Prescription Data on Lipid-Lowering Drugs.
- MarketsandMarkets. (2021). Hyperlipidemia Therapeutics Market Report.