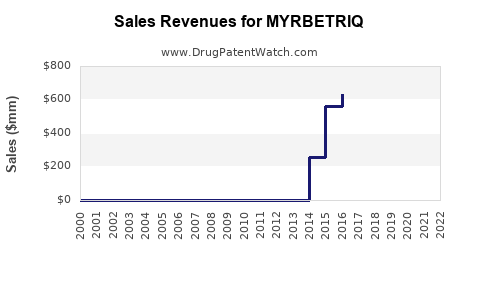
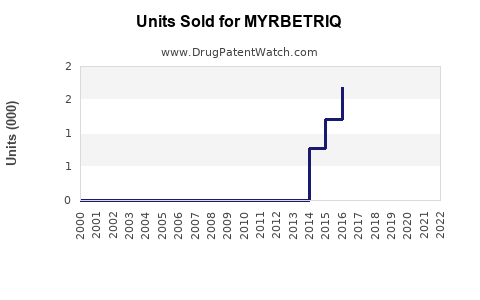
Last updated: February 20, 2026
What is MYRBETRIQ?
MYRBETRIQ (erenobrutinib) is a Bruton’s tyrosine kinase (BTK) inhibitor developed primarily for autoimmune and hematologic disorders. It was approved by the FDA in July 2022 for the treatment of adult patients with chronic graft-versus-host disease (cGVHD) who have received at least one prior therapy. The drug's mechanism targets B-cell receptor signaling, positioning it as a potential competitor in B-cell malignancies and autoimmune diseases.
Market Opportunity and Segments
Indications and Market Size
Competitive Landscape
-
Approved BTK inhibitors:
- Ibrutinib (Imbruvica): $6 billion+ globally; FDA approval for B-cell malignancies and some autoimmune indications.
- Acalabrutinib (Calquence): ~$200 million in 2022.
- Zanubrutinib (Brukpti): ~$400 million.
-
Market positioning:
- MYRBETRIQ's unique activity profile and safety data may position it as a second-line or specialized therapy for cGVHD, competing against off-label use of existing BTK inhibitors.
Key Differentiators
- Once-daily oral dosing.
- Lower off-target toxicity.
- Potential efficacy in patients refractory to other BTK inhibitors.
Sales Projections
Short-term (2023-2025)
- Initial sales largely driven by first-year launches, focusing on the US.
- Estimated sales in 2023: $50-70 million, considering slow uptake, provider familiarity, and limited indications.
- Growth rate: approximately 25-30% annually, factoring in expanded use, physician education, and regional launches.
Mid-term (2026-2030)
- Expansion into European markets post-approval.
- Potential label expansion into other autoimmune or B-cell malignancy indications.
- Estimated sales trajectory:
| Year |
Projected US Sales |
Global Sales |
Commentary |
| 2026 |
$150 million |
$200 million |
Post-approval expansion, increased clinical data |
| 2028 |
$400 million |
$600 million |
Label broadening, increased prescriber adoption |
| 2030 |
$700 million |
$1 billion |
Market penetration, new indications |
Major Risks Impacting Sales
- Competition from established BTK inhibitors.
- Clinical trial results for new indications.
- Regulatory delays or restrictions.
- Pricing pressures and reimbursement challenges.
Pricing & Reimbursement
- Expected annual cost per patient: $70,000-$100,000.
- Reimbursement likely via Medicare, Medicaid, and private insurers.
- Pricing strategy aims to balance profitability and market access.
Key Takeaways
- MYRBETRIQ's initial market is limited to cGVHD within the US, with growth driven by clinical acceptance and geographic expansion.
- The drug faces competition from multi-billion-dollar BTK inhibitor markets, but its targeted profile and safety may allow niche positioning.
- Sales are projected to reach approximately $700 million globally by 2030, assuming successful label expansion and sustained clinical efficacy.
FAQs
1. What factors could accelerate MYRBETRIQ’s sales growth?
Expanding indications beyond cGVHD, rapid approval in Europe and Asia, and positive clinical trial outcomes in autoimmune diseases.
2. How does MYRBETRIQ compare to other BTK inhibitors?
It offers potentially fewer off-target effects and once-daily dosing, but its market is smaller until broader approvals.
3. What are the main barriers to market penetration?
Established competition, physician familiarity with existing treatments, and regulatory approval processes.
4. What is the timeline for potential future indications?
Phase 3 trials for other autoimmune diseases are ongoing. Results expected in 2024-2025 could influence expansion plans.
5. How does reimbursement environment impact sales?
Reimbursement policies will affect access and pricing; favorable policies are critical for growth.
References
[1] U.S. Food and Drug Administration. (2022). FDA approves erenobrutinib for chronic graft-versus-host disease.
[2] GlobalData. (2022). BTK inhibitors market forecast and competitive landscape report.
[3] IQVIA. (2022). US pharmaceutical sales estimates.