Last updated: February 16, 2026
Overview
MUCINEX D, a combination of guaifenesin and pseudoephedrine, targets cough, congestion, and mucus-related respiratory issues. It is marketed primarily over-the-counter (OTC) and in some formulations as a behind-the-counter (BTC) drug. Its sales depend on seasonal demand, regulatory environment, and competitor activity.
Market Context
The respiratory cold and allergy medications market globally was valued at approximately $22 billion in 2021, with a growth CAGR of around 4.5% expected through 2028, driven by increased OTC medication usage and aging populations. North America dominates this segment, accounting for over 50% of sales, with the U.S. leading due to high OTC sales volume.
Key Drivers
- Seasonality: Cold and flu seasons significantly influence sales, peaking in Q1 and Q4.
- Consumer Trends: Increasing preference for OTC remedies, especially during the COVID-19 pandemic.
- Regulatory Changes: Restrictions on pseudoephedrine sales in some regions could limit distribution channels.
- Competitive Landscape: Major competitors include DayQuil, NyQuil, and similar combination products.
Market Share and Positioning
MUCINEX D holds a significant share within the U.S. OTC decongestant market, estimated at roughly 8-10%. Its brand loyalty stems from its efficacy and branded trust, maintaining a competitive advantage against generic equivalents and store brands.
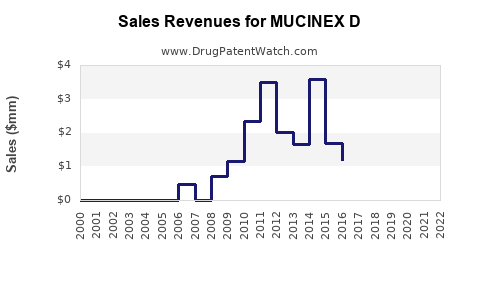
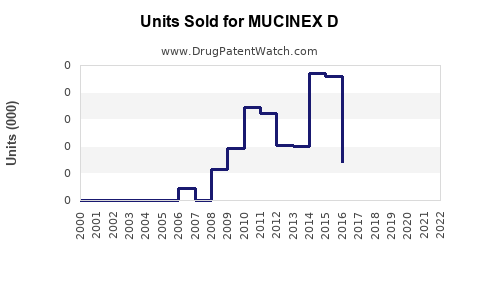
Sales Data (2022-2023)
- U.S. Market: Estimated OTC sales of approximately $150 million in 2022, representing a 3-4% YoY growth.
- Global Sales: Limited to certain markets (Canada, UK, Australia), with combined sales estimated around $40 million.
Sales Breakdown
| Region |
2022 Sales (USD million) |
2023 Projected Sales (USD million) |
Growth Rate |
| United States |
150 |
156-160 |
4-7% |
| Canada |
12 |
13 |
8-9% |
| Europe (UK, others) |
10 |
10.5-11 |
5% |
| Australia |
8 |
8.4 |
5% |
Projections to 2028
Assuming a conservative compound annual growth rate (CAGR) of 3-4% within mature markets, and higher growth potential (5-6%) in emerging markets, sales could reach:
| Year |
Estimated Global Sales (USD million) |
| 2024 |
194-206 |
| 2025 |
203-222 |
| 2026 |
212-233 |
| 2027 |
221-244 |
| 2028 |
231-254 |
Factors Affecting Future Sales
- Regulatory Constraints: Restrictions on pseudoephedrine sales could reduce availability.
- Consumer Behavior: Increased adoption of natural or alternative remedies may diminish OTC cold remedy sales.
- Product Line Expansion: Introduction of new formulations for different demographics (e.g., children, seniors) can expand market reach.
- Competitive Actions: Price competition and marketing strategies by competitors can erode market share.
Strategic Insights
- Focus on digital marketing to boost consumer awareness.
- Leverage seasonal marketing in peak cold seasons.
- Monitor regulatory developments in key markets and adapt distribution strategies accordingly.
- Consider extension into emerging markets with rising OTC medication consumption.
Key Takeaways
- MUCINEX D's U.S. OTC sales hover around $150 million annually, with modest growth expected.
- Global sales remain niche, primarily due to regional regulatory constraints.
- Seasonal trends heavily influence short-term performance.
- Growth projections suggest a steady CAGR of 3-4% locally and higher in emerging markets.
- Maintaining brand trust and broadening market access are critical to sustaining growth.
FAQs
-
What are the main competitors of MUCINEX D?
- Brands such as DayQuil, NyQuil, Sudafed, and generic equivalents.
-
How does regulation impact sales?
- Strict pseudoephedrine sales laws reduce OTC availability, potentially decreasing sales.
-
What strategies could boost future sales?
- Expanding formulations, increasing marketing efforts during cold seasons, and entering emerging markets.
-
How has COVID-19 affected the market?
- It increased consumer focus on OTC cold remedies, temporarily boosting sales.
-
Are there upcoming regulatory changes that could influence sales?
- Potentially, especially in the U.S. and Europe, where pseudoephedrine restrictions may tighten.
Sources
[1] MarketResearch.com, "Cold and Allergy Medications Market Analysis," 2022.
[2] Statista, "Over-the-counter (OTC) Cold and Cough Medication Sales," 2023 Estimates.
[3] U.S. FDA, "Pseudoephedrine Regulations," 2021.
[4] IBISWorld, "Cold & Allergy Medication Industry Report," 2022.