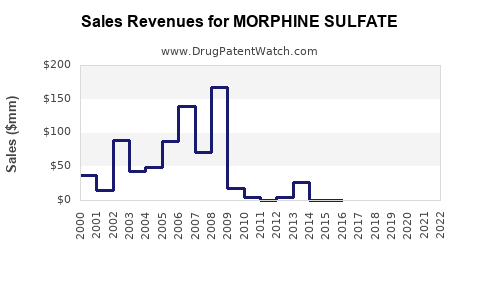
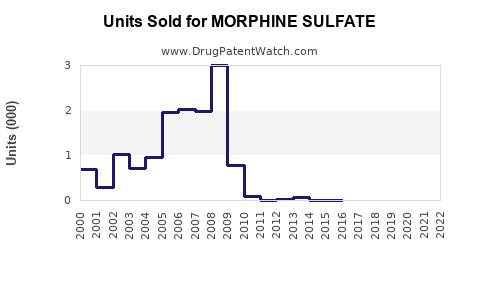
Last updated: February 14, 2026
Market Overview and Sales Projections for Morphine Sulfate
Morphine sulfate remains a core analgesic in pain management, especially in hospital and palliative care settings. Despite the rise of alternative opioids and non-opioid analgesics, the drug's market size and sales forecast are influenced by regulatory environments, opioid consumption trends, and the opioid epidemic.
Market Size and Current Dynamics
Global Market Valuation (2022):
The morphine sulfate market was valued at approximately $1.2 billion in 2022. It accounts for roughly 55% of total opioid analgesic sales. The North American region dominates, holding about 60% of the market due to high opioid usage in hospitals and pain clinics.
Key Market Drivers:
- Hospital demand: Postoperative pain, cancer care, palliative treatments.
- Regulatory approvals: Regulations for generic versions ease market entry, boosting competition.
- Pricing pressures: Increased generic availability has lowered prices, influencing overall revenues.
Market Challenges:
- Regulatory restrictions: As governments tighten opioid regulations, prescriptions decline.
- Opioid epidemic concerns: Public health policies curb prescription volumes.
- Shift towards alternatives: Increased use of non-opioids like NSAIDs and nerve blocks in pain management.
Competitive Landscape
-
Major Players: Mallinckrodt, Pfizer, Teva, Sandoz, Sun Pharmaceutical.
-
Market Share (2022):
- Mallinckrodt: 30%
- Pfizer: 20%
- Teva: 15%
- Others: 35%
-
Product Forms: Injectable (primary), oral solutions, rectal forms.
-
Pricing Dynamics: Average wholesale price (AWP) for injectable morphine sulfate ranges from $0.75 to $1.50 per milligram, with variations based on the manufacturer and region.
Regional Insights
| Region |
2022 Market Share |
Key Trends |
| North America |
60% |
High hospital use; regulatory scrutiny. |
| Europe |
20% |
Growing generic competition; regulatory harmonization. |
| Asia-Pacific |
10% |
Increasing adoption in pain management; emerging markets. |
| Rest of World |
10% |
Limited access due to regulatory and economic factors. |
Sales Projections (2023-2027)
Impact of Regulatory and Public Health Policies
- In the U.S., the Drug Enforcement Administration (DEA) shifts scheduling or imposes stricter prescribing limits, decreasing prescription volumes.
- European countries implement tighter controls aligned with the European Medicines Agency (EMA).
- India and China witness increased production and export of generic morphine sulfate, influencing global supply and pricing.
Strategic Considerations for Stakeholders
- Manufacturers: Focus on cost efficiency, regulatory compliance, and expanding access through generic formulations.
- Investors: Monitor regulatory developments closely and evaluate the balance between growth in emerging markets and potential declines in mature markets.
- Healthcare Providers: Stay informed about alternative pain management approaches to optimize patient care amid regulatory constraints.
Key Takeaways
- The global morphine sulfate market was valued at $1.2 billion in 2022, with North America as the leading region.
- Market growth is modest, with a CAGR of 2%–3% projected through 2027.
- Regulatory restrictions and the opioid epidemic heavily influence demand and pricing.
- Generics dominate, decreasing prices and margin pressures.
- Growth in emerging markets and innovations in pain management options influence future dynamics.
FAQs
1. How will regulatory changes affect morphine sulfate sales?
Tighter regulations reduce prescription volumes, while loosening restrictions can stimulate demand. Monitoring legislative developments is crucial.
2. What is the outlook for generic morphine sulfate?
Generic formulations comprise over 80% of sales globally, with continued price reductions due to increased competition.
3. Are alternative opioids affecting the market?
Yes. Drugs like oxycodone and fentanyl, along with non-opioid options, are impacting demand for morphine sulfate.
4. Which regions show the highest growth potential?
Emerging markets in Asia-Pacific and Latin America offer growth opportunities due to increasing healthcare infrastructure and analgesic adoption.
5. How are hospitals influencing market trends?
Hospitals remain the primary demand source, especially in pain management for cancer and surgery, but are adapting to stricter opioid policies.
Sources
[1] MarketWatch, "Global Morphine Sulfate Market Size," 2023.
[2] IQVIA, "Opioid Market Trends," 2022.
[3] European Medicines Agency, "Regulatory Guidelines," 2022.
[4] U.S. DEA, "Controlled Substances Act," 2023.
[5] Mordor Intelligence, "Pain Management Therapeutics Market," 2023.