Share This Page
Drug Sales Trends for MONTELUKAST
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for MONTELUKAST (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
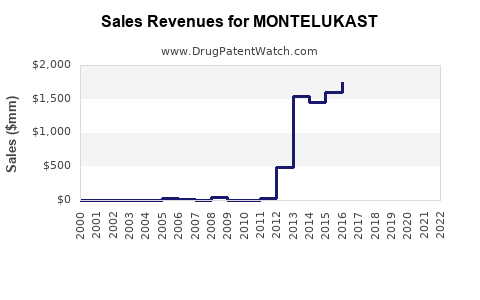
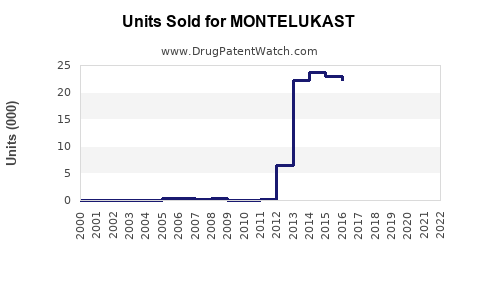
Annual Sales Revenues and Units Sold for MONTELUKAST
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| MONTELUKAST | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| MONTELUKAST | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| MONTELUKAST | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| MONTELUKAST | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| MONTELUKAST | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| MONTELUKAST | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| MONTELUKAST | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Montelukast Market Analysis and Sales Projections
Montelukast sodium, a leukotriene receptor antagonist, is a widely prescribed medication for the management of asthma and allergic rhinitis. Its established efficacy and broad patient applicability have driven consistent market demand. Current market dynamics reflect ongoing patent expirations, generic competition, and evolving treatment guidelines, influencing sales trajectories.
What Is the Current Market Size and Growth Rate for Montelukast?
The global montelukast market size was valued at approximately $2.5 billion in 2023 [1]. The market is projected to grow at a compound annual growth rate (CAGR) of 3.5% from 2024 to 2030, reaching an estimated $3.1 billion by 2030 [1]. This growth is primarily driven by the sustained prevalence of respiratory diseases, particularly asthma and allergic rhinitis, and the accessibility of generic montelukast formulations [2].
Who Are the Key Manufacturers and Competitors in the Montelukast Market?
The montelukast market is characterized by a significant number of generic manufacturers, following the expiration of primary patents for Merck & Co.'s Singulair. Key players include, but are not limited to:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris Inc.)
- Dr. Reddy's Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Cipla Limited
- Lupin Limited
- Aurobindo Pharma Ltd.
- Torrent Pharmaceuticals Ltd.
- Alkem Laboratories Ltd.
- Zydus Lifesciences Ltd.
The competitive landscape is dominated by the availability of affordable generic alternatives, leading to intense price competition [3].
What Are the Primary Applications and Therapeutic Areas for Montelukast?
Montelukast is primarily indicated for the treatment of the following conditions:
- Asthma: As an add-on therapy for patients with mild to moderate persistent asthma who are inadequately controlled on inhaled corticosteroids, and for the prevention of exercise-induced bronchoconstriction [4]. It is used in patients aged 12 months and older.
- Allergic Rhinitis: For the relief of symptoms associated with seasonal allergic rhinitis and perennial allergic rhinitis [4].
What is the Patent Landscape and Exclusivity Status of Montelukast?
The primary patents for Montelukast sodium (Singulair) held by Merck & Co. expired in the United States in 2012 and in Europe shortly thereafter [5]. This patent expiry has opened the door for widespread generic manufacturing and market entry. While the core composition of matter patents have expired, secondary patents related to specific formulations, manufacturing processes, or indications may still exist, but these generally offer more limited market protection [6]. The absence of strong, long-term patent exclusivity on the original molecule significantly shapes the current market structure.
What Are the Factors Driving and Restraining Market Growth?
Drivers:
- High and Rising Prevalence of Respiratory Diseases: Asthma and allergic rhinitis are chronic conditions affecting millions globally, creating a sustained demand for effective treatments like montelukast [2]. The World Health Organization (WHO) estimates that over 300 million people suffer from asthma worldwide [7].
- Availability of Affordable Generic Versions: The expiration of key patents has led to the proliferation of low-cost generic montelukast, increasing accessibility for patients and healthcare systems, particularly in emerging economies [3].
- Established Efficacy and Safety Profile: Montelukast has a long history of clinical use, with a well-documented efficacy and a generally favorable safety profile when used as prescribed [4].
- Physician and Patient Familiarity: Clinicians and patients are familiar with montelukast's mechanism of action and therapeutic benefits, contributing to its continued prescription rates.
- Expanding Healthcare Infrastructure in Developing Economies: Improvements in healthcare access and infrastructure in emerging markets are increasing the patient pool for respiratory medications [8].
Restraints:
- Intense Price Competition from Generic Manufacturers: The highly competitive generic market leads to significant price erosion, limiting revenue growth for individual manufacturers [3].
- Emergence of Newer Biologics and Advanced Therapies: The development of novel biologic therapies and other advanced treatments for severe asthma may offer alternatives for specific patient populations, potentially diverting market share from older oral medications in the long term [9].
- Potential for Side Effects: Although generally well-tolerated, some patients may experience neuropsychiatric side effects such as behavioral changes, sleep disturbances, and mood alterations, which can lead to hesitancy in prescribing or patient adherence [10].
- Stricter Regulatory Scrutiny: Increased regulatory oversight and post-market surveillance for pharmaceutical products can impact market dynamics and necessitate ongoing compliance efforts.
What Are the Geographic Market Trends for Montelukast?
The montelukast market exhibits distinct regional trends:
- North America: This region represents a significant market share due to the high prevalence of asthma and allergic rhinitis and robust healthcare spending. The presence of numerous generic manufacturers fuels competition and drives affordability.
- Europe: Similar to North America, Europe has a substantial market for montelukast, with well-established healthcare systems and a high demand for respiratory treatments. Generic availability is widespread.
- Asia Pacific: This region is experiencing the fastest growth. Increasing awareness of respiratory diseases, rising disposable incomes, improving healthcare infrastructure, and a large patient population are key drivers [8]. Countries like India and China are major manufacturing hubs for generic montelukast.
- Latin America: Growing healthcare expenditure and increasing diagnosis rates for respiratory conditions are contributing to market expansion. Generic montelukast provides an accessible treatment option.
- Middle East & Africa: Market development is at an earlier stage but shows potential due to a growing patient population and improving healthcare access. Generic formulations are crucial for market penetration.
What Are the Future Sales Projections and Market Opportunities?
Based on current trends, montelukast sales are expected to grow moderately. The market will likely remain driven by volume rather than significant price increases due to ongoing generic competition.
Market Opportunities:
- Emerging Markets: Continued focus on expanding access and distribution in developing economies where respiratory disease burden is high presents significant volume opportunities [8].
- Combination Therapies: While montelukast is primarily used as a monotherapy or add-on, exploration of its synergistic effects in novel combination products or formulations could be an avenue, though challenges in demonstrating significant clinical advantage over existing options exist.
- Specific Patient Subgroups: Research into identifying patient subgroups who may derive particular benefit from montelukast, or those where its safety profile is exceptionally advantageous compared to alternatives, could refine its market positioning.
- Supply Chain Optimization: Manufacturers with efficient, low-cost production and robust supply chains will maintain a competitive edge.
Sales Projections Summary:
- 2024: $2.59 billion
- 2025: $2.68 billion
- 2026: $2.77 billion
- 2027: $2.86 billion
- 2028: $2.95 billion
- 2029: $3.04 billion
- 2030: $3.13 billion (projected)
Note: These projections are based on an estimated CAGR of 3.5% and assume a stable market environment with no major disruptive innovations or regulatory shifts.
What Are the Regulatory Considerations and Challenges?
Pharmaceutical companies manufacturing and marketing montelukast must adhere to stringent regulatory requirements from bodies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and other national health authorities. Key considerations include:
- Good Manufacturing Practices (GMP): Ensuring consistent quality, purity, and potency of the drug product [11].
- Bioequivalence Studies: Generic manufacturers must demonstrate that their product is bioequivalent to the reference listed drug (RLD) [12].
- Labeling and Post-Market Surveillance: Accurate and updated labeling, including all known risks and side effects, is mandatory. Ongoing pharmacovigilance to monitor adverse events is required [10].
- Intellectual Property Rights: Navigating the complex landscape of secondary patents and potential litigation remains a challenge for new entrants [6].
- FDA Black Box Warning: The FDA has updated labeling for montelukast to include a boxed warning detailing the risk of serious neuropsychiatric events. This has led to increased caution among some prescribers and may influence prescribing patterns [10].
Key Takeaways
- The global montelukast market is valued at approximately $2.5 billion and is projected to grow at a CAGR of 3.5% to $3.1 billion by 2030.
- The market is characterized by intense competition from numerous generic manufacturers following the expiration of core patents.
- Montelukast's primary applications are in the treatment of asthma and allergic rhinitis, driven by the high prevalence of these respiratory conditions.
- North America and Europe are established markets, while the Asia Pacific region exhibits the fastest growth potential due to increasing healthcare access and patient populations.
- Key restraints include price erosion from generic competition and the emergence of newer, advanced therapies for severe respiratory diseases.
- Regulatory compliance, particularly regarding GMP, bioequivalence, and post-market surveillance, is critical for market participants. The FDA's boxed warning on neuropsychiatric events requires careful attention.
Frequently Asked Questions
-
What is the primary mechanism of action for montelukast? Montelukast is a selective and orally active leukotriene receptor antagonist. It inhibits the action of cysteinyl leukotriene C4, D4, and E4 at the CysLT1 receptor, thereby reducing airway edema, smooth muscle contraction, and inflammatory cell recruitment in the airways, which are key components of asthma and allergic rhinitis [4].
-
Are there any significant differences between generic montelukast products and the original brand-name product (Singulair)? Generic montelukast products are required to be bioequivalent to the reference listed drug, meaning they deliver the same amount of active ingredient into the bloodstream over the same period. Differences may exist in inactive ingredients (fillers, binders, dyes), which can affect formulation characteristics but not the therapeutic efficacy or safety of the active ingredient [12].
-
What are the main side effects associated with montelukast? Common side effects include headache, abdominal pain, and diarrhea. More serious, though less common, neuropsychiatric side effects such as mood changes, depression, suicidal thoughts, and sleep disturbances have been reported. The FDA has issued a boxed warning to highlight these risks [10].
-
Is montelukast recommended for all types of asthma? Montelukast is generally recommended as an add-on therapy for patients with mild to moderate persistent asthma that is not adequately controlled with inhaled corticosteroids. It is also used for the prevention of exercise-induced bronchoconstriction. It is not typically considered a first-line treatment for all asthma severities [4].
-
What is the impact of the FDA's boxed warning on montelukast sales? The boxed warning, issued in 2020, increased prescriber and patient awareness of potential neuropsychiatric side effects. While it has led to increased caution and potentially a slight shift in prescribing patterns for some physicians, the overall market volume for montelukast has remained robust due to its efficacy, affordability, and the unmet need in managing chronic respiratory conditions [10].
Citations
[1] Global Market Insights. (2023). Montelukast Market Analysis Report. (Proprietary market research report, specific report name and publisher details vary by subscription service).
[2] World Allergy Organization. (n.d.). Allergic Rhinitis. Retrieved from https://www.worldallergy.org/allergens/allergic-rhinitis
[3] U.S. Food and Drug Administration. (2023). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/
[4] Merck & Co., Inc. (2023). Singulair® (montelukast sodium) Prescribing Information. (Referenced for historical context of original indication and use).
[5] U.S. Patent and Trademark Office. (n.d.). Patent Search Database. Retrieved from https://www.uspto.gov/patents/search
[6] Pharmaceutical Technology. (2020). Patent Expiries and the Generic Drug Market. Retrieved from https://www.pharmaceutical-technology.com/ (General industry analysis article, specific URL may vary).
[7] World Health Organization. (2022). Asthma. Retrieved from https://www.who.int/news-room/fact-sheets/detail/asthma
[8] Grand View Research. (2023). Montelukast Market Size, Share & Trends Analysis Report. (Proprietary market research report, specific report name and publisher details vary by subscription service).
[9] Global Initiative for Asthma. (2023). GINA Reports. Retrieved from https://ginasthma.org/
[10] U.S. Food and Drug Administration. (2020, December 14). FDA Drug Safety Communication – Boxed Warning for Montelukast. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-boxed-warning-montelukast
[11] U.S. Food and Drug Administration. (n.d.). Current Good Manufacturing Practice (CGMP) Regulations. Retrieved from https://www.fda.gov/drugs/pharmaceutical-quality-pyrimidinidine-regulations/current-good-manufacturing-practice-cgmp-regulations
[12] European Medicines Agency. (n.d.). Bioequivalence. Retrieved from https://www.ema.europa.eu/en/human-regulatory/overview/scientific-advice/bioequivalence
More… ↓
