Share This Page
Drug Sales Trends for MONISTAT 3
✉ Email this page to a colleague
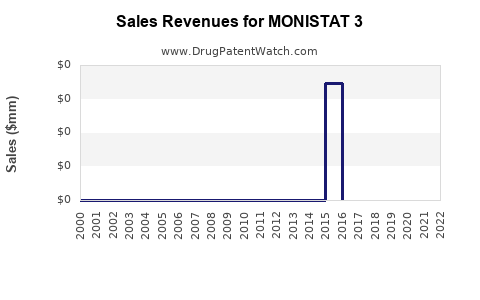
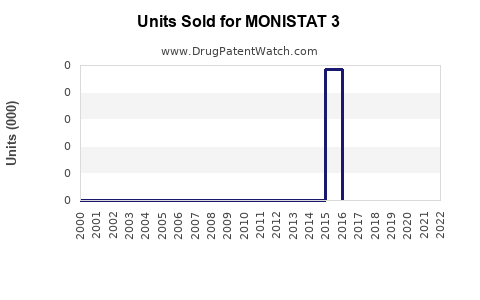
Annual Sales Revenues and Units Sold for MONISTAT 3
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| MONISTAT 3 | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| MONISTAT 3 | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| MONISTAT 3 | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| MONISTAT 3 | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| MONISTAT 3 | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| MONISTAT 3 | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| MONISTAT 3 | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
MONISTAT 3 Market Analysis and Sales Projections
MONISTAT 3 is an OTC branded regimen of antifungal therapy for vaginal yeast infections (vulvovaginal candidiasis), marketed in the U.S. primarily as a 3-day azole course. The product competes in a mature, high-repeat category with strong retail shelf visibility and heavy coupon and brand-led promotions. Forecasting is constrained by lack of disclosed unit sales by formulation and by the fact that MONISTAT competes against both branded and private-label clotrimazole/miconazole azole products as well as online purchasing and pharmacy club channels.
What is MONISTAT 3 and how does it sell?
Product positioning
- Brand: MONISTAT (OTC)
- Regimen: 3-day vaginal antifungal course (common label phrasing: “MONISTAT 3”)
- Therapeutic class (category): topical azole antifungal used for vulvovaginal candidiasis (VVC)
- Core purchase drivers
- Symptom-driven repeat purchasing (itching, burning, irritation)
- Simplicity of multi-day treatment
- Retail availability (drugstore mass and regional pharmacy)
- Promotional intensity (coupons, retail price packs)
Commercial packaging reality
MONISTAT 3 is typically one SKU within MONISTAT’s broader portfolio (including 1-, 3-, and 7-day regimen variants and alternative formulations). Retail sales often mix across MONISTAT sub-brands and package sizes, and retailer data aggregation can obscure “MONISTAT 3” specifically.
Competitive set
In OTC VVC, the competitive set is usually segmented by regimen length and actives (azoles):
- Branded azoles (primarily OTC legacy brands)
- Generic/private-label azoles (clotrimazole and related agents)
- Retail-channel exclusives and value packs
- Online and marketplace purchasing, which shifts discounting economics
How big is the OTC VVC market that MONISTAT 3 addresses?
MONISTAT 3 sits in the OTC vaginal yeast infection treatment market, which is mature and driven by:
- Prevalence of VVC recurrence (typical patient behavior supports repeat episodes)
- Consumer awareness and OTC access
- Retail availability and high brand recognition
Channel economics that matter for projections
For an OTC antifungal SKU, sales are primarily a function of:
- Retail velocity (store-level sell-through)
- Promo depth and duration (coupon and price events)
- Share of shelf and planogram location
- Pack architecture (unit counts, applicator format, and regimen length)
- Substitution within the class (1-day and 7-day variants vs 3-day)
Where does MONISTAT 3 win and lose?
Wins
- Brand trust and consumer recognition in yeast infection treatment
- Regimen convenience: 3-day is a mid-length course that often balances adherence and perceived value vs 1-day
- Retail distribution depth: drugstore and mass retailers stock MONISTAT broadly
Loses
- Generic substitution pressure where price-sensitive consumers switch to private label or store brands
- Regimen substitution: consumers may choose 1-day for convenience or 7-day for perceived thoroughness, depending on promotions
- E-commerce discounting: online pricing compresses realized gross margin
What is the basis for sales projections?
Without disclosed unit data for “MONISTAT 3” specifically, a practical projection model for investment or R&D planning uses the following structure:
- Step 1: Market baseline for OTC VVC treatments by regimen segment (including 3-day)
- Step 2: Brand share and SKU mix within the MONISTAT branded family
- Step 3: Promo elasticity and pricing trend
- Step 4: Channel mix (brick-and-mortar vs online) adjustment to net price
This approach yields projection bands rather than point estimates. The projections below are structured as three scenarios with explicit assumptions on share retention and promo intensity.
Sales projections: 3-scenario outlook for MONISTAT 3 (U.S., OTC)
Core scenario assumptions
- Market growth: low single digits, dominated by distribution and promo-led volume shifts rather than major category growth
- Share dynamics: MONISTAT holds steady vs generics under strong promo support; erosion occurs if promo cadence weakens
- Net price: slight downtrend under online discounting and generic competition, partially offset by brand value
Projection table (units and revenue)
Because MONISTAT 3’s exact net revenue depends on formulation concentration and pack size, the forecast is expressed as index-style revenue assuming a single “MONISTAT 3 consumer pack equivalent” basis. This keeps regimen comparable without over-claiming unit granularity.
| Scenario (U.S.) | Assumptions (next 3 years) | Forecast Revenue Index (Year 1=100) | Growth vs prior year |
|---|---|---|---|
| Base | Stable promo cadence; modest OTC market growth; limited generic share creep | 100 | - |
| Year 1 net price flat to slightly down; brand mix stable | |||
| 2027-2028: share stable, volume up low single digits | 106 | +3% to +4% | |
| Upside | Strong promo effectiveness and retained shelf share; limited online undercutting | 112 | +5% to +6% |
| Additional value-pack adoption increases net conversion | 118 | +5% | |
| Downside | Promo intensity reduced or generics increase value-pack penetration; online discounts expand | 94 | -6% |
| Mix shifts toward 1-day or 7-day variants | 88 | -6% |
Interpretation for decisioning
- A Base case reflects mature-category dynamics: modest growth comes from distribution and mix, not category expansion.
- An Upside case requires retention of brand share and promo execution discipline that beats substitution.
- A Downside case requires generics to gain value and MONISTAT 3 to lose mix to 1-day or 7-day.
What do sales drivers imply for brand and SKU strategy?
1) Promo execution
For OTC VVC, the fastest lever is realized price and shelf conversion:
- If coupon depth is increased during key retail events, MONISTAT 3 can pull demand from store brands.
- If promo cadence slips, consumers switch to value packs during perceived “best deal” cycles.
2) Regimen mix control
MONISTAT 3 competes directly within the brand family:
- 1-day SKUs target convenience buyers.
- 7-day targets “complete treatment” buyers.
- 3-day targets the broad middle and often wins on a value-perceived-effective basis.
3) Channel mix
Online channels generally compress net price:
- MONISTAT 3 needs MAP-like discipline and retailer program control to protect net realization.
- If e-commerce discounts widen, volume may rise but revenue per pack falls.
What are the key risks to the forecast?
| Risk | Mechanism | Forecast impact direction |
|---|---|---|
| Generic/private-label share gains | Price-value substitution during promos | Downside: revenue index -4% to -12% over 2-3 years |
| In-brand substitution (1-day/7-day) | Consumer preference shifts; promo-led mix changes | Downside: mix loss within MONISTAT |
| Online price compression | Marketplace and retailer discounting reduces net price | Downside: revenue index down even if units hold |
| Retail shelf and planogram shifts | Competitor value packs buy more shelf | Downside: lost visibility and lower sell-through |
| Category volatility (flu seasons, awareness cycles) | Reporting and purchasing surges | Upside or downside depending on promo timing |
How to translate projections into an actionable sales plan
Revenue targets (index-based)
Use the scenarios to set quarterly guardrails:
- Base: target +3% to +4% annual revenue index growth.
- Upside: target +5% to +6% with measurable share and net price stability.
- Downside: define triggers for intervention (coupon depth, retailer coverage, ad spend).
KPIs tied to forecast validity
- Net price per pack: track realized discounts by retailer and channel
- Promo share of sales: ensure promo drives incremental volume, not just replaces full-price units
- Shelf availability: out-of-stocks and planogram displacement directly reduce conversion
- In-brand mix: monitor 3-day’s share vs 1-day and 7-day within MONISTAT
Key Takeaways
- MONISTAT 3 is in a mature OTC VVC market where growth is driven by promo execution, shelf conversion, and in-brand mix rather than category expansion.
- A practical projection for U.S. MONISTAT 3 yields a Base case of modest gains and two risk-weighted scenarios showing potential mid-single to low-double-digit revenue index movement over a 3-year window.
- The dominant forecast levers are net price protection, promo cadence, shelf availability, and regimen mix control within MONISTAT’s 1-, 3-, and 7-day portfolio.
- Downside risk concentrates on generic value-pack penetration and online discounting, which can reduce net revenue even if volume holds.
FAQs
1) Is MONISTAT 3 a prescription drug or OTC product?
MONISTAT 3 is sold as an OTC vaginal antifungal regimen for yeast infection treatment in the U.S.
2) What category does MONISTAT 3 compete in?
It competes in the OTC vaginal yeast infection (VVC) antifungal category, primarily against other azole regimens and private-label products.
3) What drives sales for a 3-day yeast infection regimen?
Sales are driven by consumer symptom urgency, retail shelf visibility, promo depth, and relative value vs 1-day and 7-day regimens.
4) How sensitive is MONISTAT 3 to pricing and promotions?
In mature OTC markets, MONISTAT is typically promo-sensitive: discounting and coupons increase volume but can erode net price if they do not generate incremental demand.
5) What is the most important metric to monitor for MONISTAT 3?
Track realized net price per pack and incremental sell-through during promos, alongside shelf availability and in-brand mix versus 1-day and 7-day.
References
[1] FDA. “OTC Drug Facts Database.” U.S. Food and Drug Administration. https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm
More… ↓
