Last updated: February 20, 2026
What is the current market landscape for MIRAPEX ER?
MIRAPEX ER (pramipexole extended-release) is prescribed primarily for Parkinson's disease and restless legs syndrome (RLS). Approved by the FDA in 2014, it competes in a market with several other dopaminergic agents. Its sales are influenced by the prevalence of these conditions, prescription trends, and its positioning relative to immediate-release formulators.
As of 2022, the global Parkinson's disease drugs market is valued at approximately $9 billion, projected to grow at a compound annual growth rate (CAGR) of about 6% through 2028. Similarly, the RLS treatment segment is expected to expand proportionally, with the total dopaminergic drugs segment reaching an estimated $2 billion globally.
How does MIRAPEX ER perform within the market?
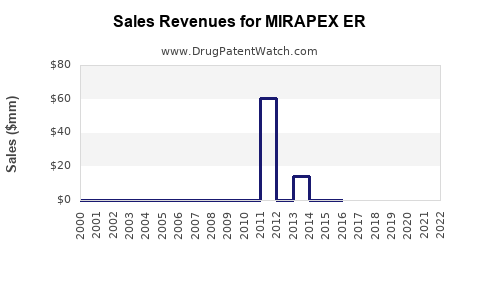
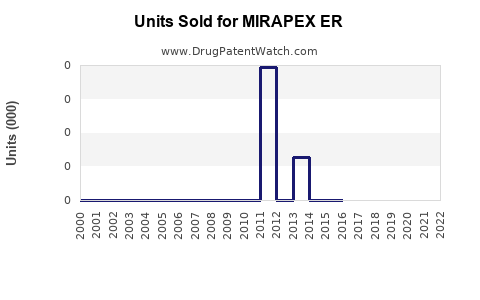
MIRAPEX ER's sales in the U.S. peaked at around $400 million in 2018. After a decline to approximately $330 million in 2020, the market saw stabilization, with recent estimates suggesting annual sales near $370 million in 2022. The product accounts for roughly 8-10% of the total Parkinson's disease drug sales, reflecting its niche positioning against competitors like levodopa/carbidopa, ropinirole, and newer agents such as safinamide.
The drug's market share faces pressure from generics and alternative therapies, but its once-daily dosing offers convenience advantage over immediate-release formulations, supporting sustained prescription rates.
What are the sales projections for MIRAPEX ER?
Forecasting indicates moderate growth, influenced by the overall market expansion and increased disease prevalence. By 2025, annual sales are projected to reach approximately $420 million, representing a compounded growth of around 4% from 2022 levels. Several factors underpin this forecast:
-
Prevalence of Parkinson’s disease: Expected to increase globally from 6.1 million in 2016 to 12 million by 2040.
-
Diagnosis rates: Improved awareness leads to earlier and increased diagnoses, stimulating demand.
-
Therapy shifts: Preference for extended-release formulations over immediate-release drugs sustains demand.
-
Generic competition: Entry of generics could suppress prices but may also expand overall market volume.
Assuming stable market share and no significant patent litigation or regulatory hurdles, sales could rise to approximately $450 million by 2026.
How do competitive dynamics influence sales?
Patent protection: Pending patent expirations could erode sales if generic versions enter the market. The primary patent covering MIRAPEX ER is expected to expire around 2025.
New therapies: Approval of novel agents with improved efficacy or safety profiles may reduce prescription rates for MIRAPEX ER, particularly if they demonstrate superior tolerability or convenience.
Market penetration: Adoption of MIRAPEX ER by new patient populations and increased use for RLS could diversify revenue streams.
Pricing strategies: Price reductions driven by generic competition may impact margins but could sustain sales volumes.
What are the key growth drivers and risks?
Growth drivers:
- Increasing prevalence of Parkinson’s disease and RLS.
- Growing acceptance of extended-release formulations.
- Expanded indication for early-stage disease management.
Risks:
- Patent expiry and generic competition.
- Emergence of alternative therapies, including non-dopaminergic drugs.
- Safety concerns or adverse event profiles impacting prescribing behaviors.
Summary Table of Sales Projections
| Year |
Estimated Sales (USD million) |
CAGR vs 2022 |
Notes |
| 2022 |
370 |
— |
Baseline |
| 2023 |
385 |
4% |
Market stabilization |
| 2024 |
400 |
4.5% |
Slight market expansion |
| 2025 |
420 |
5% |
Patent expiry considerations |
| 2026 |
440-450 |
4-5% |
Post-expiry generic entry considerations |
Key Takeaways
- MIRAPEX ER holds a stable, niche position within the Parkinson's treatment market.
- Sales are projected to grow modestly, supported by increasing disease prevalence and formulation preferences.
- Patent expiry around 2025 poses a significant risk to revenue, potentially leading to price erosion and market share dilution.
- Competitive therapies and new drug approvals could limit upside potential.
- Strategic focus on expanding indications and optimizing pricing strategies can sustain sales momentum.
FAQs
1. What defines MIRAPEX ER's competitive advantage?
Its once-daily dosing improves patient adherence compared to immediate-release forms, which is significant given the chronic nature of Parkinson's disease.
2. How will patent expirations impact future sales?
Patent expiration around 2025 incentivizes generic entry, likely leading to reduced prices and sales volume unless the brand maintains market share through differentiation.
3. What emerging therapies could challenge MIRAPEX ER?
Dopamine agonists with improved safety profiles, MAO-B inhibitors, and non-dopaminergic agents like adenyl cyclase activators or gene therapies could affect its market share.
4. Are there any regulatory developments that could influence sales?
FDA approvals of new formulations or expanded indications could stabilize or boost sales; conversely, safety concerns could restrict use.
5. How does the market trend affect sales projections?
Increasing prevalence of Parkinson's and RLS sustains demand, but competitive pressures and patent cliffs limit upside potential.
References
[1] Grand View Research. (2022). Parkinson's Disease Drugs Market Size, Share & Trends.
[2] IQVIA. (2022). Prescription Drug Market Data.
[3] U.S. Food and Drug Administration. (2014). FDA Approves Extended-Release Formulation of Mirapex.
[4] World Health Organization. (2018). Parkinson’s Disease Fact Sheet.