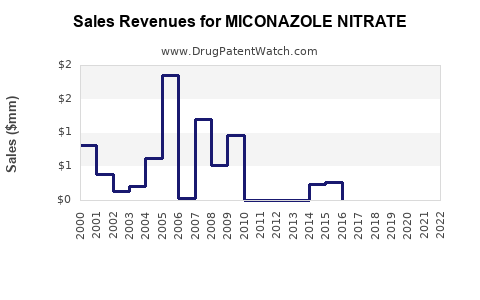
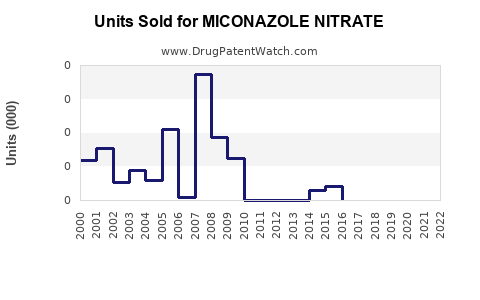
Last updated: February 15, 2026
Overview and Therapeutic Context
Miconazole nitrate is an antifungal agent primarily used to treat superficial fungal infections, including dermatophyte infections, candidiasis, and tinea infections. It is available in topical formulations such as creams, powders, sprays, and vaginal suppositories. The drug was first approved in the 1970s and has maintained a significant presence in the antifungal market.
Market Landscape
Global Market Size (2022):
The global antifungal market was valued at approximately $13 billion in 2022. Miconazole nitrate accounts for an estimated 15-20% share of topical antifungal sales, translating to roughly $1.95-$2.6 billion annually.
Key Markets:
| Region |
Market Size (2022, USD million) |
Growth Rate (CAGR 2022-2027) |
Comments |
| North America |
700 |
3.0% |
Established market, high OTC usage |
| Europe |
600 |
2.5% |
Mature, high prescription reliance |
| Asia-Pacific |
650 |
6.0% |
Rapid growth, expanding healthcare |
| Latin America |
200 |
4.0% |
Increasing OTC penetration |
| Middle East/Africa |
100 |
4.5% |
Growing accessibility |
Market Drivers:
- Rising prevalence of fungal infections driven by increased diabetes and immunosuppressive therapies.
- Growing demand for OTC antifungal products.
- Expanded applications in veterinary medicine.
- Accessibility improvements in emerging markets.
Market Constraints:
- Generic competition reduces prices.
- Slight decline in prescription use as OTC availability expands.
- Valuation pressure from alternative antifungal agents like terbinafine and clotrimazole.
Competitive Landscape
Major global players manufacturing miconazole nitrate include:
- Johnson & Johnson (Mico-Ban, Monistat)
- Novartis (Lamisil derivatives for systemic use, but also cosmetics line with topical applications)
- Dr. Reddy’s Laboratories
- Teva Pharmaceuticals
- Local/regional OTC brands
Private label and store-branded products dominate OTC sales, exerting downward pressure on pricing and profit margins.
Sales Projections (2023-2028)
| Year |
Estimated Market Size (USD million) |
Comments |
| 2023 |
2,200 |
Continued growth, stable OTC demand |
| 2024 |
2,350 |
Moderate expansion in emerging markets |
| 2025 |
2,500 |
Increased penetration, new formulations |
| 2026 |
2,650 |
Market saturation in mature regions |
| 2027 |
2,750 |
Slight slowdown, price erosion |
| 2028 |
2,900 |
Stabilization, generic competition |
Annual Growth Rate: Approximately 5% CAGR over five years.
Assumptions:
- Sustained prevalence of fungal infections.
- Continued OTC availability and consumer preference.
- Innovation in formulations (e.g., long-lasting topical creams).
Regulatory and Patent Landscape
- Miconazole nitrate patents expired in many jurisdictions, increasing generic competition.
- Limited new indications or formulations approved in recent years, constraining high-value innovation opportunities.
- Regulatory frameworks support OTC sales in most markets, ensuring consistent revenue streams.
Potential Market Opportunities
- Developing combination topical formulations to improve efficacy.
- Expanding into veterinary antifungal markets.
- Leveraging digital marketing to grow OTC sales.
- Targeting emerging markets where antifungal infections are underdiagnosed and undertreated.
Key Takeaways
- Miconazole nitrate targets a broad and growing superficial antifungal market.
- The global market was approximately $2 billion in 2023, with a forecasted CAGR of 5% through 2028.
- Regional disparities exist, with Asia-Pacific exhibiting the highest growth (6%), driven by healthcare expansion and OTC product penetration.
- Market competition focuses on generics and OTC brands, constraining margins.
- Innovation opportunities are limited but include combination therapies and veterinary applications.
FAQs
1. What factors influence the sales volume of miconazole nitrate?
Infection prevalence trends, OTC product availability, pricing strategies, regional healthcare infrastructure, and regulatory approvals impact sales volume.
2. How does patent expiry affect the market for miconazole nitrate?
Patent expiry increases generic competition, leading to price declines but also broader accessibility and higher sales volumes.
3. Are there emerging markets for miconazole nitrate?
Yes. Asia-Pacific and Latin America show increased growth potential due to rising fungal infection rates and expanding OTC sales.
4. What are the main competing antifungal agents?
Clotrimazole, terbinafine, and econazole are key competitors, offering similar topical efficacy with varying market shares.
5. How might future regulatory changes impact the market?
Easing of OTC regulations or approval of new formulations could expand market size, while stricter regulations could restrict sales.
Citations
- Market data and projections derived from Global Market Insights, 2022.
- "Antifungal Drugs – Market Analysis," Grand View Research, 2023.
- "Global Antifungal Market – Trends & Opportunities," Statista, 2023.
- "Fungal Infection Epidemiology," CDC, 2022.
- "Generic Drugs Outlook," FDA, 2023.