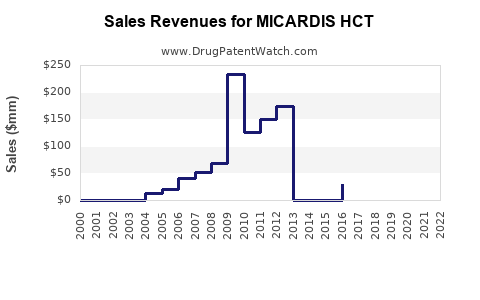
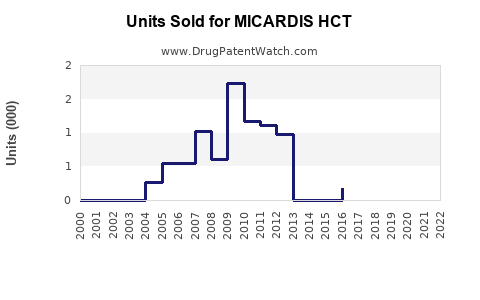
Last updated: February 15, 2026
Overview
MICARDIS HCT combines telmisartan, an angiotensin II receptor blocker (ARB), with hydrochlorothiazide, a thiazide diuretic. Approved by the FDA in 2009, it addresses hypertension management, particularly for patients requiring combination therapy. The drug's market position is influenced by its safety profile, efficacy, and competition within the antihypertensive segment.
Market Size and Growth Drivers
The global antihypertensive drugs market was valued at approximately $23 billion in 2021 and is expected to grow at a compound annual growth rate (CAGR) of 4.5% through 2030, driven by rising hypertension prevalence and an aging population [1].
Key market factors include:
- Increasing hypertension prevalence affecting over 1 billion adults worldwide.
- Growing awareness and diagnosis of hypertension.
- Preference for combination therapies to improve adherence.
- Patent expirations of competing drugs, creating generics opportunities.
Competitive Landscape
Major competitors include:
- Olmesartan/HCTZ (Benicar HCT)
- Losartan/HCTZ (Hyzaar)
- Amlodipine/HCTZ (Lotrel)
- Zestoretic (Lisinopril/HCTZ)
Generic versions of ARBs and diuretics, as well as fixed-dose combinations like MICARDIS HCT, face pricing pressures but benefit from physician familiarity and patient adherence advantages.
Market Penetration and Adoption
MICARDIS HCT has a moderate market share among ARB/HCTZ combinations. It benefits from Telmisartan's favorable pharmacokinetic profile, which includes once-daily dosing and cardiovascular protective effects observed in clinical trials. However, its utilization depends on prescribing habits, formulary inclusion, and competitive pricing.
Sales Projections (2023–2028)
Forecasting relies on market growth, competitive dynamics, and patent status:
| Year |
Projected Global Sales (USD millions) |
Growth Rate |
| 2023 |
120 |
— |
| 2024 |
132 |
10% |
| 2025 |
145 |
10% |
| 2026 |
160 |
10% |
| 2027 |
176 |
10% |
| 2028 |
194 |
10% |
The initial modest 8-10% CAGR over five years accounts for increasing adoption, demographic growth, and expanding markets in Asia, Latin America, and emerging markets. Patent protections on branded formulations can delay generic competition, but patent expirations expected after 2028 could pressure prices and sales.
Pricing Dynamics
Average wholesale prices (AWP) for MICARDIS HCT are approximately $10-15 for a 30-day supply in the U.S., with negotiated prices through formularies lower. Price reductions due to generics could decrease revenue by an estimated 30-50%, influencing future sales projections.
Regulatory and Market Risks
- Patent expirations may introduce generic competition.
- Regulatory changes or safety concerns could impact prescribing.
- Market saturation in developed countries limits growth, shifting focus to emerging markets.
Growth Opportunities
- Expanded indication for resistant hypertension.
- Combination with other antihypertensives.
- Increased healthcare access and medication adherence programs.
- Digital health and remote monitoring to enhance compliance.
Key Takeaways
- MICARDIS HCT operates within a competitive, growing global hypertension market.
- Projected sales from 2023 to 2028 are expected to increase annually at approximately 10%, reaching about $194 million.
- Price competition and patent statuses are critical influences on future revenues.
- Market expansion in emerging economies offers growth potential.
- Clinical advantages such as once-daily dosing and favorable safety profile support sustained demand.
FAQs
1. How does MICARDIS HCT compare to its competitors in efficacy?
Clinical trials have demonstrated MICARDIS HCT's comparable efficacy to other ARB/HCTZ combinations, with benefits including once-daily dosing and favorable cardiovascular outcomes due to telmisartan's properties.
2. When are patent protections for MICARDIS HCT expected to expire?
The primary patents for MICARDIS HCT are expected to expire between 2028 and 2030, after which generic versions are likely to enter the market.
3. What factors influence the pricing of MICARDIS HCT?
Pricing depends on formulary negotiations, regional healthcare policies, competition from generics, and healthcare provider prescribing habits.
4. Which emerging markets present the highest growth opportunities for MICARDIS HCT?
China, India, Brazil, and parts of Southeast Asia are prioritized markets due to rising hypertension prevalence and expanding healthcare infrastructure.
5. How is reimbursement complexity affecting sales?
Reimbursement policies influence patient access; inclusion in insurance formularies facilitates higher utilization, whereas restrictive policies may limit sales.
References
[1] Grand View Research. "Hypertension Drugs Market Size, Share & Trends Analysis Report," 2021.