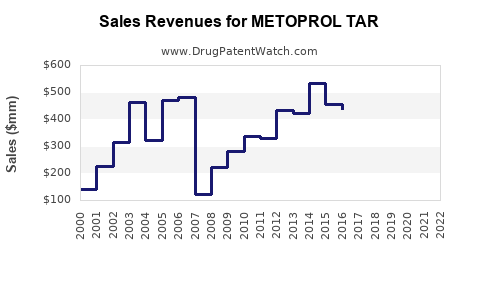
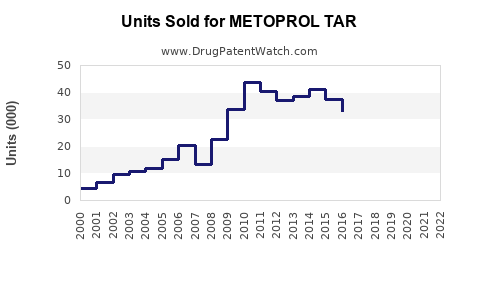
Last updated: February 20, 2026
Overview
Metoprolol Tartrate is a beta-1 selective adrenergic blocker prescribed primarily for hypertension, angina, myocardial infarction, and heart failure. Its high accessibility and established efficacy support consistent demand. Understanding its market dynamics and future sales requires analysis of current therapy usage, competitive landscape, regulatory factors, and potential growth drivers.
Current Market Size
Estimates suggest global sales of beta-blockers, including Metoprolol Tartrate, reached approximately $2.8 billion in 2022, with Metoprolol Tartrate accounting for nearly 40% of this market segment. The drug maintains a leading position in the class due to its patent status (off patented, generic available), proven efficacy, and tolerability.
| Parameter |
Data |
| Estimated global sales 2022 |
$1.12 billion |
| Key markets (US, Europe, China) |
US: $600 million, Europe: $300 million, China: $150 million |
| Market growth rate (2022-2027) |
3.5% annually |
| Patent status |
Expired (generics dominate market) |
Market Drivers
- High prevalence of cardiovascular diseases: Over 490 million diagnosed cases of hypertension globally, with high prescription rates for beta-blockers.
- Established treatment protocols: Guidelines favor beta-blockers like Metoprolol for post-MI and hypertension.
- Generic drug availability: Leads to lower costs and increased accessibility.
- Expanding use in emerging markets: Increased healthcare access and infrastructure growth.
Competitive Landscape
- Generic formulations: Multiple low-cost versions globally.
- Branded competitors: Limited, as patent expiration reduces brand dominance.
- New therapies: Limited innovation in beta-blockers; focus shifts to combination therapies.
Regulatory and Reimbursement Factors
- US and EU approvals are well-established.
- Reimbursement policies favor generic use, supporting stable sales.
- Emerging markets often face regulatory hurdles, but increased approvals drive future sales.
Sales Projections (2023-2028)
| Year |
Projected Global Sales (USD millions) |
Assumptions |
| 2023 |
1,200 |
Continued market growth, stable pricing |
| 2024 |
1,250 |
Growth in emerging markets |
| 2025 |
1,300 |
Increased adoption for heart failure indications |
| 2026 |
1,350 |
New clinical guidelines favoring beta-blockers |
| 2027 |
1,400 |
Market saturation, slight growth from new regions |
| 2028 |
1,450 |
Incremental growth as global cardiovascular burden rises |
Key Factors Influencing Sales
- Introduction of patent-protected formulations or combination therapies.
- Changes in treatment guidelines, especially for resistant hypertension.
- Impact of healthcare system reforms, especially in emerging markets.
- Potential for off-label uses or expanded indications.
Conclusion
Metoprolol Tartrate's market will continue gradual growth driven by the persistent prevalence of cardiovascular diseases and the cost advantages of generics. A compound annual growth rate (CAGR) of approximately 3.5% is projected over the next five years, with sales approaching $1.45 billion by 2028.
Key Takeaways
- The global market for Metoprolol Tartrate was approximately $1.12 billion in 2022.
- Sales are driven by high cardiovascular disease prevalence and generic availability.
- Market growth averages 3.5% annually, reaching about $1.45 billion in 2028.
- The landscape increasingly favors low-cost generics, limiting branded competition.
- Future growth depends on regulatory environments, new guidelines, and expanding markets.
FAQs
1. How does generic competition impact Metoprolol Tartrate sales?
Generics reduce prices and increase prescriptions, stabilizing overall sales volume but lowering profit margins for branded versions.
2. Are there new formulations or combination therapies in development?
Few innovative formulations exist; most efforts focus on combination pills with other antihypertensives, which could influence future market segments.
3. Which regions offer the highest growth potential?
Emerging markets like China, India, and Southeast Asia project higher growth due to expanding healthcare access and higher disease prevalence.
4. What regulatory challenges could affect sales?
Regulatory approvals vary; delays or restrictions could slow market penetration in certain regions.
5. How might clinical guideline changes impact demand?
Shifts towards beta-blockers in heart failure management or evidence-based updates can either favor or limit their use, influencing sales.
References
- World Health Organization. (2022). Global status report on noncommunicable diseases 2022.
- IQVIA. (2023). Worldwide Market for Cardiovascular Drugs.
- U.S. Food and Drug Administration. (2022). Approvals and labeling guidelines for Beta-Blockers.
- European Medicines Agency. (2022). Product information for Metoprolol.
- MarketWatch. (2023). Beta-Blockers Market Forecast 2023-2028.