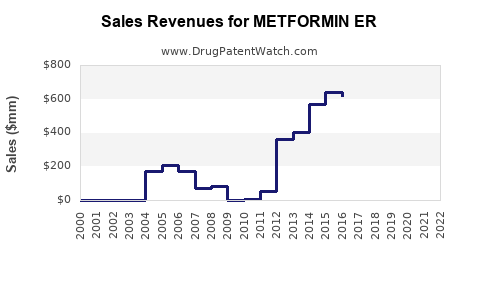
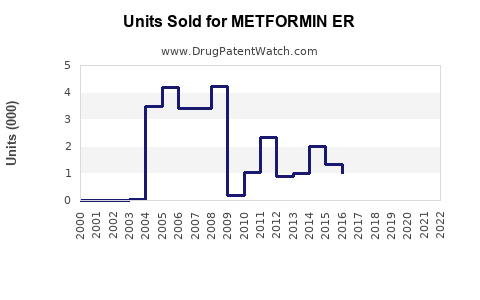
Last updated: February 14, 2026
Metformin ER is a formulary version of metformin designed for once-daily dosing, primarily used in type 2 diabetes management. It accounts for longstanding revenue generated by the metformin class, with growing emphasis on ER formulations due to improved patient adherence.
Market Size and Growth Trends
The global market for diabetes medications, which includes metformin ER, was valued at approximately USD 68.8 billion in 2022, with an annual growth rate of about 9% projected until 2028.[1] Within this, the metformin segment holds a significant share due to its position as a first-line therapy, especially in early-stage type 2 diabetes.
Metformin ER's market share within the larger metformin segment has been increasing, attributed to benefits such as lower gastrointestinal side effects and improved compliance over immediate-release (IR) formulations.
Key Market Drivers
- Prevalence of Type 2 Diabetes: Approximately 415 million adults globally suffer from diabetes, with type 2 accounting for around 90% of cases.[2] The increasing incidence of obesity, sedentary lifestyles, and aging populations expand the diabetic population.
- Guideline Recommendations: Major health organizations, including the American Diabetes Association (ADA), advocate for metformin as the initial pharmacologic treatment.[3] ER formulations are viewed favorably for chronic management.
- Patient Compliance: Once-daily dosing of ER improves adherence compared to multiple IR doses, leading to favored prescribing of ER forms.
Competitive Landscape
Major pharmaceutical companies with FDA approval for metformin ER include:
- AbbVie (Glucophage XR)
- Lupin (Metformin XR)
- Mylan (Metformin XR)
- Zydus Cadila (Metformin ER)
Generic versions dominate the market, with several manufacturers offering bioequivalent products, intensifying price competition and margin pressures.
Market Share and Sales Forecasts (2023–2028)
| Year |
Estimated Global Sales (USD Billion) |
CAGR |
Key Notes |
| 2023 |
2.7 |
4.0% |
Market remains fragmented; increased adoption of ER formulations in developed markets. |
| 2024 |
2.8 |
3.7% |
Continued growth driven by emerging markets and new formulary guidance. |
| 2025 |
3.0 |
3.4% |
Patent expirations for some branded ER products increase generic market share. |
| 2026 |
3.2 |
3.3% |
Rising prevalence of diabetes sustains demand; improved healthcare access in emerging economies. |
| 2027 |
3.4 |
3.1% |
Healthcare system investments support increased diabetes screening and pharmacotherapy. |
| 2028 |
3.6 |
3.0% |
Market stabilizes with a broader generic footprint and increased focus on patient adherence. |
Pricing Dynamics
Generic metformin ER prices range from USD 0.10 to USD 0.20 per tablet, with branded equivalents exceeding USD 0.30, depending on dosage and market. Price sensitivity remains high in low- and middle-income countries, whereas branded products maintain higher margins in developed markets.
Regulatory and Reimbursement Environment
- FDA Approval: Most generic versions of metformin ER have FDA ANDA approvals, facilitating market entry.
- Reimbursement: Insurance coverage varies; in the US, Medicare, Medicaid, and private insurers generally reimburse generic variants, encouraging utilization.
- Patent Status: Some branded ER formulations are patent-expired, which increases the availability of generics.
Risks and Opportunities
- Manufacturing Challenges: Formulating stable ER tablets can be complex; manufacturing quality influences market share.
- Regulatory Changes: New guidelines on diabetes treatment favoring combination therapies could affect monotherapy sales.
- Emerging Markets: Rapid urbanization and increased healthcare infrastructure support expanding use.
Key Takeaways
- The global market for metformin ER is projected to grow from USD 2.7 billion in 2023 to USD 3.6 billion in 2028.
- Demand is driven by the rising prevalence of type 2 diabetes, guideline endorsements, and improved patient compliance.
- Generic competition and low price points characterize the market, especially outside developed economies.
- Market growth will slow gradually, stabilizing around 3% annually, with emerging markets offering growth opportunities.
- Regulatory environment favors generics, but manufacturing quality remains vital.
FAQs
Q1: What factors influence the sales of metformin ER?
The primary drivers are the global increase in type 2 diabetes prevalence, prescribing guidelines favoring metformin, and patient adherence advantages of ER formulations. Price competition also impacts sales volume.
Q2: Who are the leading producers of metformin ER?
Major players include AbbVie (Glucophage XR), Lupin, Mylan, and Zydus Cadila, with several other generic manufacturers operating globally.
Q3: How does patent status affect the market?
Most branded ER formulations' patents have expired, allowing generics to enter and drive prices downward, improving access but reducing branded sales margins.
Q4: What are the key challenges facing metformin ER sales?
Market saturation in mature markets, manufacturing complexities, and potential shifts towards combination therapies or novel agents pose risks.
Q5: Which regions are expected to see the most growth in metformin ER sales?
Emerging markets in Asia, Latin America, and Africa are expected to see increased adoption due to rising diabetes prevalence and expanding healthcare infrastructure.
Citations:
- [1] Grand View Research. "Diabetes Drugs Market Size, Share & Trends Analysis Report." 2022.
- [2] International Diabetes Federation. "IDF Diabetes Atlas," 10th edition, 2021.
- [3] American Diabetes Association. "Standards of Medical Care in Diabetes—2023." Diabetes Care, 2023.