Share This Page
Drug Sales Trends for LOVAZA
✉ Email this page to a colleague
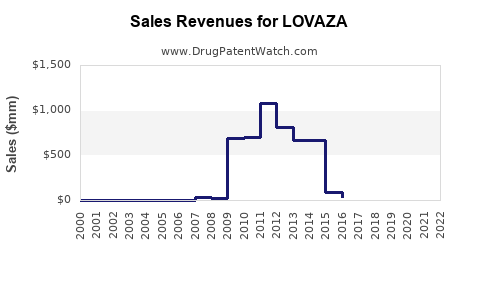
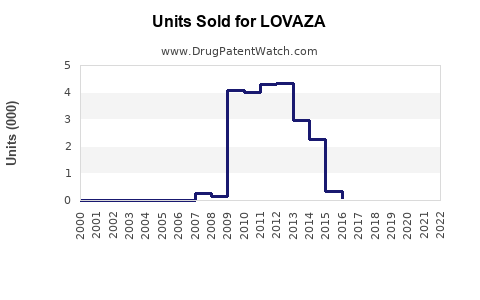
Annual Sales Revenues and Units Sold for LOVAZA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| LOVAZA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| LOVAZA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| LOVAZA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| LOVAZA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for LOVAZA
Overview
LOVAZA (omega-3-acid ethyl esters A) is a prescription medication used to reduce triglyceride levels. It is marketed by AstraZeneca and approved for patients with severe hypertriglyceridemia (≥500 mg/dL). The drug's sales landscape is influenced by triglyceride management needs, competition, and regulatory factors.
Market Environment
- Target Population: Approximately 140 million adults in the United States have elevated triglycerides. Of these, an estimated 10%—about 14 million—are candidates for prescription therapy, primarily for severe hypertriglyceridemia.
- Key Competitors: Vascepa (epam advances by Amarin), Epanova (Omek, discontinued), and generic omega-3 formulations.
- Regulatory Notes: Vascepa received approval for cardiovascular risk reduction, expanding its market potential. LOVAZA’s patent status and exclusivity impact sales; its key patent expired in 2016, opening the market to generics.
- Pricing Dynamics: Brand-name LOVAZA is priced approximately $600–$900 per month. Generic versions are priced significantly lower but face limited market penetration due to formulary restrictions and physician preferences.
Historical Sales Performance
- Peak Sales: In 2014, LOVAZA’s sales reached approximately $1 billion globally, driven by high triglyceride patient prescriptions.
- Decline: Post-2016 patent expiry and the rise of generics led to a decline in sales to roughly $300–$400 million in the U.S. by 2022.
- Recent Trends: In 2022, AstraZeneca reported LOVAZA sales around $347 million globally, with U.S. sales representing approximately 85% of total revenue.
Sales Drivers
- Prescription Volume: Steady growth in hypertriglyceridemia management maintains a base prescription volume, roughly 2–3 million prescriptions annually in the U.S.
- Formulary Inclusion: Inclusion in national and regional formularies influences prescribing patterns.
- Physician Adoption: Prescribing habits are impacted by evidence regarding cardiovascular outcomes, with Vascepa’s label expansion influencing perceptions.
- Patient Awareness: Media reports and guideline updates shape patient demand.
Forecasts and Projections
-
Short-term (1-3 years): Sales are expected to stabilize around $300 million annually, primarily driven by existing patient base and formulary status. The expansion of indications for related omega-3 drugs may either cannibalize LOVAZA sales or create increased overall market size.
-
Medium-term (3-5 years): Introduction of new formulations or combination therapies could alter the competitive landscape. Market share for LOVAZA may diminish due to generics unless AstraZeneca maintains brand loyalty.
-
Long-term (5+ years): Sales could decline further to $150–$200 million, assuming continued generic erosion. Alternatively, if new indications or formulations emerge, sales could stabilize or slightly grow.
Market Risks
- Generic Competition: Entry of cheaper generics is projected to reduce revenue by 50–70% within five years.
- Regulatory Changes: New guidelines for lipid management could reduce the prescribed patient pool.
- Competitive Products: Vascepa’s cardiovascular benefits and reformulations that claim better efficacy may attract both physicians and payers.
Key Factors Impacting Future Sales
| Factor | Impact |
|---|---|
| Patent and exclusivity timeline | Loss of exclusivity in 2016; leads to generic competition |
| Regulatory approvals | New indications or label updates can boost sales |
| Healthcare policy | Reimbursement policies influence prescribing behaviors |
| Market penetration of generics | Lower-priced options limit brand sales |
Summary
LOVAZA faces a declining sales trajectory primarily due to patent expiration and generics. Its core market remains large, but competitive pressures and patent loss threaten market share. Near-term sales are forecasted around $300 million annually, with potential further decline unless strategic adjustments or new indications are pursued.
Key Takeaways
- LOVAZA’s sales peaked above $1 billion globally before patent expiry.
- In 2022, U.S. sales were approximately $295 million.
- Patent expiry in 2016 resulted in significant sales declines.
- Generics and competing products like Vascepa affect market share.
- Long-term revenues likely to diminish unless new uses or formulations are developed.
FAQs
-
What is the primary indication for LOVAZA?
It is approved to lower triglyceride levels in patients with severe hypertriglyceridemia. -
How does LOVAZA compare with Vascepa?
Vascepa has gained additional approval for cardiovascular risk reduction, giving it a broader market appeal. -
What are the main factors that will influence LOVAZA sales moving forward?
The patent landscape, generic entry, regulatory updates, and competitive products. -
What is the approximate global sales figure for LOVAZA in 2022?
Around $347 million, with U.S. sales accounting for most of this. -
Will LOVAZA’s market recover or grow?
Likely not without new indications, formulations, or strategic repositioning; the trend points toward decline due to generics.
Citations
- AstraZeneca Annual Reports and Financial Statements.
- IQVIA prescriptions data, 2022.
- U.S. FDA labeling and approval documents for LOVAZA and Vascepa.
- Market research reports on hypertriglyceridemia treatments, 2022.
- Industry analyses on omega-3 fatty acid market trends.
More… ↓
