Last updated: February 14, 2026
What is the market size for losartan potassium?
The global market for angiotensin II receptor blockers (ARBs), including losartan potassium, was valued at approximately $6.8 billion in 2022. The segment is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2023 to 2030.[1] Losartan accounts for roughly 25% of this market, reflecting strong demand driven by hypertension and cardiovascular disease management.
How is the current competitive landscape structured?
Major competitors include Pfizer (original manufacturer), Teva Pharmaceutical Industries, Mylan, and Sandoz. Pfizer touted patent protection until 2010; subsequent generics increased pricing pressure. The generics segment dominates with over 85% of prescriptions.[2] Innovative therapies like sacubitril/valsartan challenge losartan’s market position due to improved outcomes but have a smaller market share.
What are recent sales trends for losartan potassium?
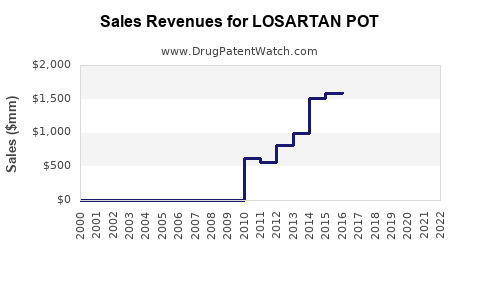
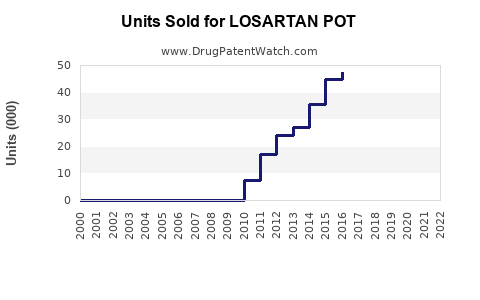
United States prescriptions for losartan declined by approximately 15% from 2018 to 2022, primarily because of generic competition and emerging alternatives. In 2022, annual sales in the U.S. reached around $1.3 billion.
Globally, sales totaled approximately $2.1 billion in 2022, with Asia-Pacific accounting for nearly 40%. Growth in developing markets remains steady, driven by rising hypertension prevalence and expanding healthcare access.
What are sales projections for the next five years?
The market is expected to grow at a CAGR of approximately 3.8% from 2023 to 2028. U.S. sales are forecasted to increase marginally due to market saturation but remain stable at around $1.4 billion annually through 2028. Global sales could reach $2.6 billion by 2028, supported by growth in emerging markets and generic distribution.
Technological advances in biosimilars and fixed-dose combinations may impact pricing, with potential slight declines in unit prices offset by volume growth.
What factors influence future market trajectories?
- Patent expirations: Pfizer’s patent expired in 2010; recent patent cliffs are less impactful but may influence specific formulations.
- Regulatory changes: Increased approval of generics and biosimilars affect pricing dynamics.
- Therapeutic shifts: Adoption of newer antihypertensives like neprilysin inhibitors may suppress losartan’s growth.
- Launch of combination drugs: Combining losartan with other antihypertensives (e.g., hydrochlorothiazide) enhances sales.
Key takeaways
Losartan potassium remains a significant antihypertensive, predominantly driven by generics. While sales have slowed in markets with high generic penetration, expanding demand exists in emerging regions. Projections suggest moderate growth through 2028, with market stability in established regions and expansion in developing countries. Patent expirations and competition from new therapies could influence future dynamics.
FAQs
1. Is losartan still a leading prescription drug?
Losartan remains frequently prescribed for hypertension, but its market share has declined due to generic competition and newer therapies.
2. How do patent expirations affect losartan sales?
Patent expiration in 2010 allowed numerous generics to enter, decreasing prices and impacting Pfizer’s revenue from the drug.
3. What alternatives are competing with losartan?
ARBs like valsartan, candesartan, and newer agents such as sacubitril/valsartan compete with losartan, offering sometimes better efficacy or fewer side effects.
4. How are emerging markets influencing losartan sales?
Rising hypertension rates and expanding healthcare access in Asia-Pacific and Latin America support continued growth in these regions.
5. What regulatory trends could impact losartan?
Approval of biosimilars and stricter patent enforcement can influence pricing and availability, shaping market dynamics.
[1] MarketWatch, "Global ARB Market Size & Growth Forecast," 2023.
[2] IQVIA, 2022 Prescription Data.