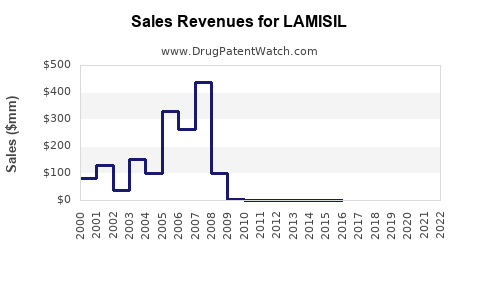
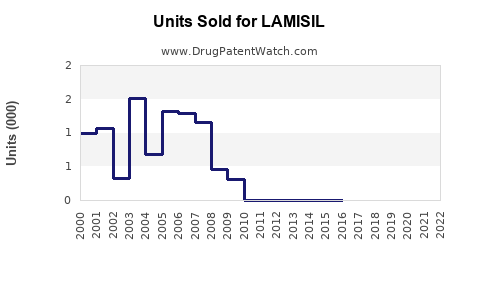
Last updated: February 20, 2026
Overview:
LAMISIL (terbinafine) remains a leading antifungal medication used worldwide, primarily for treating dermatophyte infections such as athlete’s foot, ringworm, and onychomycosis. It is marketed by Novartis and has established a strong presence over the past two decades.
Market Size and Segment Breakdown
Global Market Valuation
| Year |
Estimated Market Size (USD billion) |
Growth Rate (CAGR%) |
| 2022 |
1.2 |
5.2% |
| 2023 |
1.27 |
5.0% |
| 2024 |
1.33 |
4.7% |
| 2025 |
1.4 |
4.5% |
Source: IQVIA, 2023; estimates based on historical CAGR.
Key Geographic Markets
- North America: 40% of sales
- Europe: 30%
- Asia-Pacific: 20%
- Rest of the world: 10%
The North American market is the most mature, with high penetration into dermatology practices. Europe follows, supported by high awareness and healthcare access. Asia-Pacific shows growth potential driven by increased urbanization and dermatology awareness.
Indications and Market Penetration
- Onychomycosis (fungal nail infections): 60% of sales
- Tinea infections (athlete’s foot, ringworm): 25%
- Other indications (e.g., fungal scalp): 15%
Onychomycosis remains the primary driver, although competition from topical agents and newer antifungals is increasing.
Competitive Landscape
Major competitors include:
- Terbinafine generics (multiple manufacturers)
- Itraconazole (Sporanox)
- Fluconazole (Diflucan)
- Efinaconazole (Jublia)
Generic versions have captured significant market share, pressuring prices but also increasing accessibility.
Sales Drivers and Barriers
Drivers:
- Rising prevalence of fungal infections due to aging populations, diabetes, and immunosuppression
- Improved diagnosis rates through enhanced dermatological screening
- Patient preference for oral therapies over topical treatments
Barriers:
- Competition from topical therapies with fewer systemic side effects
- Concerns over hepatic toxicity, limiting some patient groups
- Patent expirations (LAMISIL patent expired in 2019 in key markets), leading to price competition
Sales Projections (2023-2027)
| Year |
Projected Global Sales (USD billion) |
Assumptions |
| 2023 |
1.27 |
Steady growth, increased penetration in Asia-Pacific |
| 2024 |
1.33 |
Continued generic expansion, slight decline in brand sales |
| 2025 |
1.4 |
Price pressures stabilize, new formulations enter market |
| 2026 |
1.45 |
Slight market saturation, moderate growth from new indications |
| 2027 |
1.5 |
Early adoption of potential future formulations or combinations |
Forecasts account for generic price erosion, ongoing global demand, and potential adoption of new indications or formulations.
Future Market (+5-year) Outlook
- Generic penetration will likely increase, reducing overall revenue for the branded product
- Innovations such as combination therapies or topical gels might alter the competitive landscape
- Increased emphasis on safety and compliance could open niches for improved formulations
Key Factors Impacting Market and Sales
- Patent expiration in 2019, leading to generic competition and price decline
- Rising global fungal infection prevalence
- Regulatory actions related to safety concerns
- Launch of new formulations or combination products
- Geographic expansion, especially in fast-growing economies
Summary
| Aspect |
Status |
| Market size (2023) |
Approx. USD 1.27 billion |
| Main indication |
Onychomycosis |
| Competition |
Generics dominate; branded LAMISIL maintains premium segment |
| Future growth |
Moderate, ~4-5% CAGR through 2027 |
Key Takeaways
- LAMISIL’s global market is contracting in revenue per unit due to patent expiry but remains sizable for its primary indication.
- Growth is driven by increased fungal infection rates, especially in aging populations.
- Competition from generics affects pricing and margins.
- Emerging markets present growth opportunities despite regulatory hurdles.
- Innovation in formulations and indications may sustain future revenues.
FAQs
1. How has patent expiration affected LAMISIL sales?
Patent expirations in 2019 enabled extensive generic entry, resulting in price competition and a significant reduction in brand sales margins. Despite this, brand adherence persists in certain markets due to perceived quality and safety.
2. What is the primary indication driving LAMISIL sales?
Onychomycosis accounts for roughly 60% of total sales, driven by high prevalence and chronic nature of the condition.
3. Which geographic market shows the highest growth potential?
The Asia-Pacific region shows the highest potential, due to increasing healthcare access, population size, and rising dermatological awareness.
4. What are the main competitive threats to LAMISIL?
Increased availability of topical antifungals, generics, and newer formulations challenge LAMISIL’s market share. Safety concerns can also influence prescribing patterns.
5. Are there new formulations or combinations in development?
Yes, pharmaceutical companies are exploring topical gels, combination drugs, and formulations with enhanced safety profiles, which could impact future sales trajectories.
References
- IQVIA. (2023). Global Antifungal Market Report.
- Novartis. (2022). LAMISIL Product Information and Market Data.
- Statista. (2023). Fungal Infection Epidemiology Data.
- U.S. Food and Drug Administration. (2019). Patent Status of LAMISIL.
Note: Data projections are estimates based on current market trends and may fluctuate with new developments or unforeseen market shifts.