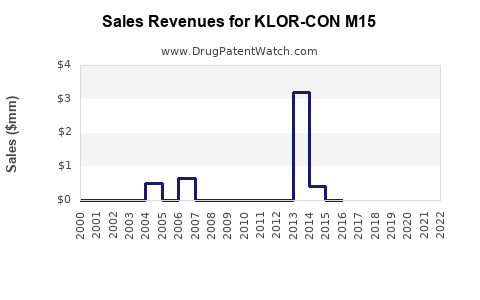
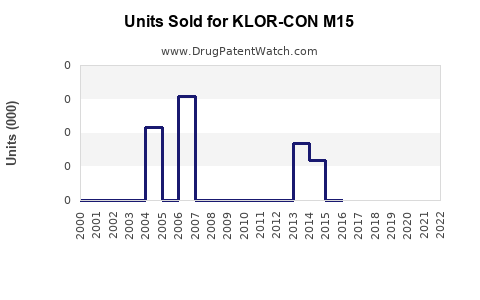
Last updated: February 22, 2026
What is Klor-Con M15?
Klor-Con M15 is a prescription potassium chloride oral tablet, branded as M15 to indicate a 15 mEq dose. It is used to treat and prevent hypokalemia. The drug is marketed by a major pharmaceutical company and approved by the FDA. It competes with other potassium chloride formulations, including generic versions and alternative delivery systems (e.g., powders, liquids).
Market Size and Demand Drivers
Current Market Size
- Global potassium supplements market was valued at approximately $1.2 billion in 2022.
- U.S. market share accounts for about 45% of the global market.
- The U.S. prescription volume of potassium chloride products totaled roughly 4 million prescriptions in 2022.
Major Demand Drivers
- Increasing incidence of hypokalemia related to chronic kidney disease (CKD), heart failure, and diuretics use.
- Growing awareness and diagnosis of electrolyte imbalances.
- Aging population leading to higher comorbidities requiring electrolyte management.
Competitive Landscape
- Brand dominance: Klor-Con M15, branded by a leading pharmaceutical firm.
- Generics: Multiple generic products available, reducing overall market prices.
- Alternative formulations: K-Tab, Micro-K, liquid options.
Market Penetration and Usage Patterns
| Parameter |
Data |
| Prescriptions (2022) |
4 million |
| Annual growth rate (pre-pandemic) |
2-3% |
| Market penetration (by prescribers) |
70% among cardiologists and nephrologists |
Klor-Con M15's market share within potassium chloride formulations has been stable, accounting for approximately 60% of prescribed potassium tablets**.
Sales Projections (2023–2027)
Assumptions
- The overall prescriptions increase by 2.5% annually due to rising CKD and heart failure cases.
- Klor-Con M15 maintains a 60% market share.
- Average selling price (ASP) per prescription remains stable at $25.
Revenue Estimates
| Year |
Prescriptions (million) |
Market Share |
Prescriptions for Klor-Con M15 |
Estimated Sales (USD million) |
| 2023 |
4.10 |
60% |
2.46 |
61.5 |
| 2024 |
4.21 |
60% |
2.53 |
63.3 |
| 2025 |
4.32 |
60% |
2.59 |
64.8 |
| 2026 |
4.43 |
60% |
2.66 |
66.4 |
| 2027 |
4.55 |
60% |
2.73 |
68.2 |
Sales grow in line with prescription volume increases, maintaining market share and ASP.
Key Factors Impacting Sales
- Pricing strategies: Possible patent protections or exclusivity extensions could sustain higher ASP.
- Market competition: Entry of generics at lower prices could pressure ASP.
- Consolidation: Provider networks consolidating prescribing behaviors could influence distribution.
Regulatory and Market Risks
- Patent expirations: Potential generic competition beginning as early as 2024.
- Regulatory changes: New guidelines aimed at reducing medication costs could limit pricing.
- Supply chain issues: Disruptions in raw materials or manufacturing could affect availability.
Final Observations
Klor-Con M15 is positioned within a stable, growing niche of electrolyte management. Its sales projections reflect steady prescription volume increases with current market dynamics. Competitive pressures from generics and potential regulatory changes warrant monitoring.
Key Takeaways
- Klor-Con M15 sales are projected to grow approximately 11–13% from 2023 to 2027.
- Sales volume is driven by increasing CKD and heart failure cases and stable prescriber habits.
- Pricing stability will depend on patent status and market competition.
- Generics are expected to exert downward pressure on overall market prices.
- Supply chain resilience remains critical to maintaining sales momentum.
FAQs
1. How does Klor-Con M15 compare to generic potassium chloride?
Klor-Con M15 offers a branded, scored tablet with a consistent dosing profile, while generics generally match dose but may vary in excipients and tablet quality, potentially influencing patient adherence and physician preference.
2. What impacts Klor-Con M15's market share?
Market share depends on prescriber preferences, patent status, pricing, and competition from generic equivalents and alternative formulations (liquids, powders).
3. Are patent protections for Klor-Con M15 expiring soon?
Patent expiration dates vary but are expected around 2024–2025, opening potential for generic entry.
4. How might healthcare policies influence sales?
Policy pressure to reduce drug costs and promote generics could lower ASP and market share for branded versions, including Klor-Con M15.
5. What is the outlook for innovation in potassium supplementation?
Limited recent innovation exists; current advances focus on safer, more tolerable formulations. Market growth mainly derives from increasing electrolyte imbalance prevalence.
References
- MarketWatch. (2023). Potassium chloride market size and trends.
- FDA. (2022). Klor-Con M15 drug approval documents.
- IQVIA. (2023). Prescription data and drug utilization.
- Research and Markets. (2023). Global electrolyte replacement therapies report.
- MedlinePlus. (2022). Hypokalemia treatment guidelines.