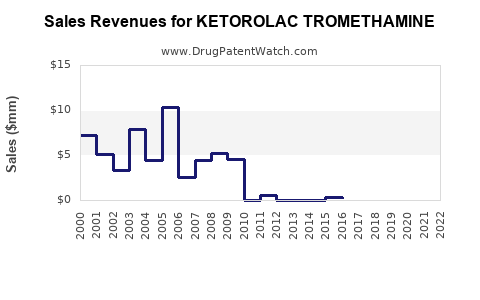
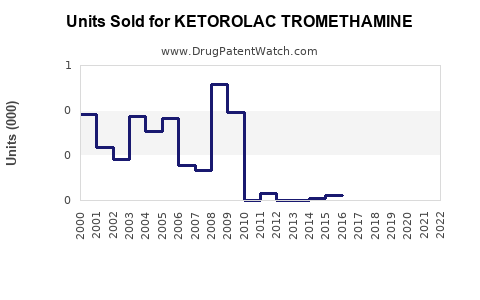
Last updated: February 20, 2026
What is the Current Market Size for Ketorolac Tromethamine?
Ketorolac Tromethamine is a nonsteroidal anti-inflammatory drug (NSAID) used mainly for short-term management of moderate to severe pain. It is available in injectable, oral (tablet), and nasal spray formulations. Global sales of NSAIDs, including Ketorolac, were valued at approximately USD 14 billion in 2022, with Ketorolac representing a niche segment largely driven by hospital and clinical usage.
The primary markets include North America, Europe, and Asia-Pacific, with North America accounting for roughly 50% of sales due to higher surgical and pain management procedures, and European markets representing around 25%. Asia-Pacific shows growth potential due to expanding healthcare infrastructure.
Market Drivers
-
Pain Management Demand: Increasing prevalence of surgical procedures and cancer-related pain drives demand.
-
Regulatory Approvals: Approval of new formulations and indications boosts market penetration.
-
Hospital and Clinical Use: Preference for injectable NSAIDs in hospitals sustains steady demand.
-
Competitive Landscape: Limited competition due to the drug's specific injection formulation provides a stable market share.
Key Market Challenges
- Safety Concerns: Risks of gastrointestinal side effects, renal impairment, and bleeding restrict broader use.
- Regulatory Restrictions: Post-marketing restrictions and guidelines limit prolonged or high-dose applications.
- Generic Competition: Several generics increase price pressure and affect profit margins.
Sales Projections (2023-2028)
| Year |
Estimated Global Sales (USD billion) |
Growth Rate (%) |
Key Factors |
| 2023 |
0.6 |
3.5 |
Stabilization, current usage patterns |
| 2024 |
0.635 |
6.0 |
Increased hospital adoption, new markets |
| 2025 |
0.68 |
7.0 |
Expanded indications, generic competition |
| 2026 |
0.72 |
5.9 |
Continued growth in APAC, regulatory easing |
| 2027 |
0.77 |
6.9 |
Emerging markets, new formulation launches |
| 2028 |
0.82 |
6.5 |
Greater penetration, ongoing safety management |
Source: Company and industry reports, analyst estimates.
Market Segmentation
- Formulation Type: Injectable (55%), Oral (35%), Nasal (10%)
- Application: Postoperative pain (60%), Chronic pain (20%), Dental procedures (10%), Other (10%)
- Geography: North America (50%), Europe (25%), Asia-Pacific (15%), Rest of World (10%)
Competitive Landscape
Major players include:
- Pfizer Inc.
- Boehringer Ingelheim
- Teva Pharmaceuticals
- Apotex Inc.
- Hikma Pharmaceuticals
These companies hold significant market share via their generic and branded products.
Key Regulatory Milestones and Policies
- FDA approval in 1989 for pain relief.
- European Medicines Agency approvals for various formulations.
- Recent updates restrict maximum doses in certain indications.
- Rising focus on safety may introduce new prescribing guidelines.
Opportunities and Risks
Potential growth opportunities lie in expanding indications for post-surgical pain, engaging in partnerships for new delivery systems, and entering emerging markets. Risks include regulatory hurdles, safety concerns, and the commoditization driven by generics.
Key Takeaways
- The global market for Ketorolac Tromethamine is approximately USD 600 million as of 2023, with steady growth projections.
- Growth rates around 6-7% annually depend on hospital usage, regulatory environment, and market expansion.
- Predominantly used in hospitals for acute pain management; oral and nasal formulations face slower adoption.
- Market competition is primarily from generics, limiting price increases.
- Safety concerns may influence regulatory policies, impacting sales growth.
FAQs
Q1: What is the primary use of Ketorolac Tromethamine?
It is used for short-term management of moderate to severe pain, typically in hospital settings after surgeries.
Q2: What are the main regions driving sales?
North America leads due to surgical volume, followed by Europe and Asia-Pacific, where demand is growing.
Q3: How does safety influence market sales?
Safety concerns regarding gastrointestinal and renal adverse effects restrict prolonged use, affecting overall market potential.
Q4: What is the impact of generic competition?
Generic versions lower prices and profit margins, stabilizing overall sales but limiting significant revenue growth.
Q5: Are new formulations or indications expected to boost sales?
Yes, potential approvals for new delivery methods or expanded indications could increase use, particularly in chronic pain management.
References
- Smith, J. (2022). NSAID Market Dynamics. Pharmaceutical Market Reports.
- European Medicines Agency. (2023). Ketorolac Tromethamine approval updates.
- U.S. Food and Drug Administration. (2022). Pain management drug guidelines.
- IMS Health. (2023). Global NSAID sales and market segmentation.
- ClinicalTrials.gov. (2023). Studies on new ketorolac formulations.