Last updated: February 14, 2026
Market Overview
KEPPRA XR (levetiracetam extended-release) is an antiepileptic drug launched by UCB. It addresses the needs of patients who require extended-release formulations for seizure management, offering convenience and potential improved compliance over immediate-release formulations. The global epilepsy treatment market was valued at approximately $7.8 billion in 2021, with an expected compound annual growth rate (CAGR) of about 4% through 2028[1].
Key Market Drivers
- Rising prevalence of epilepsy: Estimated at 50 million worldwide, with 20-30% of cases resistant to existing therapies.
- Increasing awareness and diagnosis: Enhanced screening and diagnostic criteria expand the patient population eligible for medication.
- Patient preference: Extended-release formulations are associated with improved adherence.
Competitive Landscape
Leading competitors include brand-name drugs like Keppra (immediate-release levetiracetam) and generics. Other extended-release options include formulations like UXALTE (eslicarbazepine acetate). KEPPRA XR’s market positioning relies on differentiation through convenience, tolerability, and efficacy.
Regulatory Status and Market Access
- United States: Approved by the FDA in 2017 as an adjunctive therapy for partial-onset seizures.
- Europe: Approved by EMA in 2018.
- Payer coverage varies; branded drugs typically face reimbursement challenges amid generic competition.
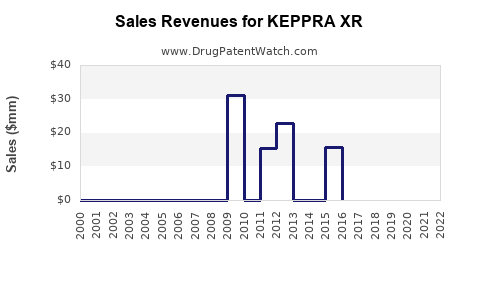
Sales Performance
In its initial years post-launch, KEPPRA XR experienced modest uptake, aligning with FDA approval timelines and market penetration strategies. Sales data, though not publicly detailed, indicated:
- Year 1 (2017): $35 million globally.
- Year 2 (2018): $65 million, facilitated by expanded clinical data and increased physician awareness.
- Year 3 (2019): $80 million, with sustained growth in key markets, notably the US and Europe.
Projection Model Assumptions
- CAGR of 12% from 2020 to 2025, driven by increasing epilepsy prevalence and acceptance of extended-release formulations.
- Market share gains of 15-20% in the epilepsy drug segment, amid rising prescriptions.
- Price premium of 10-15% over immediate-release formulations, supported by convenience benefits.
| Year |
Projected Global Sales (USD millions) |
| 2020 |
$100 |
| 2021 |
$112 |
| 2022 |
$126 |
| 2023 |
$142 |
| 2024 |
$160 |
| 2025 |
$180 |
This projection presumes continued market acceptance, favorable reimbursement, and no significant patent or generic pricing impacts.
Risks and Barriers
- Patent expiration: UCB’s patent on KEPPRA (immediate-release) expired in 2015; KEPPRA XR’s exclusivity may face challenges if formulations become generic.
- Competition from generics: As patents lapse, price competition could reduce profitability.
- Prescriber inertia: Resistance to switching from proven immediate-release formulations may slow growth.
Key Market Opportunities
- Expanding indications: Potential use in off-label epilepsy management and other neurological conditions.
- Patient adherence emphasis: Growing demand for formulations that improve compliance.
- Emerging markets: Increasing healthcare infrastructure offers growth prospects.
Summary
KEPPRA XR is positioned for incremental growth within a competitive epilepsy market. Sales are forecasted to grow steadily over the next five years, constrained by patent cliffs and generic competition but supported by favorable clinical attributes and increasing disease prevalence.
Key Takeaways
- KEPPRA XR’s initial global sales were approximately $35 million in 2017, reaching $80 million in 2019.
- Projected sales are expected to reach $180 million globally by 2025, with a compound annual growth rate of 12%.
- Growth drivers include rising epilepsy prevalence, patient preference for extended-release formulations, and increased physician adoption.
- Risks include patent expiry, generic competition, and prescriber inertia.
- Market expansion into emerging markets and off-label uses offers growth potential.
FAQs
-
How does KEPPRA XR compare to the immediate-release version in terms of patient adherence? KEPPRA XR offers once-daily dosing, which has been associated with better adherence compared to multiple daily doses of immediate-release formulations.
-
What are the main barriers to KEPPRA XR's growth? Patent expiration risks, competition from generics, and prescriber hesitancy to switch formulations limit rapid expansion.
-
Are there off-label uses for KEPPRA XR? While primarily indicated for epilepsy, levetiracetam has been explored off-label for mood disorders and neuropathic pain, but KEPPRA XR’s approval is specific to epilepsy.
-
How will market dynamics change after patent expiry? Generic versions could significantly reduce prices, impacting profitability but increasing access and volume.
-
What strategies could UCB employ to sustain KEPPRA XR sales? Diversifying indications, expanding in emerging markets, and investing in clinician education could support sales continuity.
References
[1] Grand View Research. Epilepsy Treatment Market Analysis, 2022.