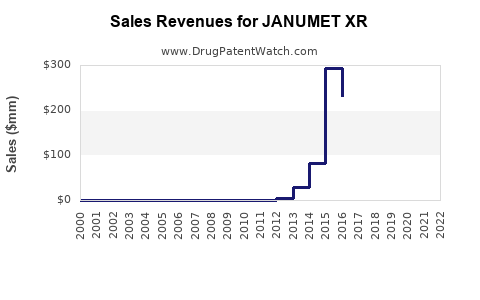
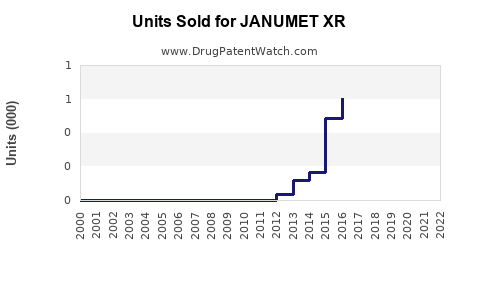
Last updated: February 21, 2026
What is the current market position of JANUMET XR?
JANUMET XR (sitagliptin and metformin extended-release) is approved for managing type 2 diabetes mellitus. It combines a DPP-4 inhibitor with metformin ER. The drug received FDA approval in September 2013. As of 2022, JANUMET XR remains a key player within the oral antidiabetic segment, competing primarily with other fixed-dose combination (FDC) drugs such as Tradjenta (linagliptin), Jardiance (empagliflozin), and passed-means like Jardiamet (empagliflozin + metformin).
Market size and growth dynamics
The global type 2 diabetes drugs market was valued at approximately USD 62.4 billion in 2022. It is forecasted to grow at a compound annual growth rate (CAGR) of 7.2% until 2030. The growth drivers include rising prevalence of diabetes, expanded indication approvals, and increased adoption of combination therapies.
Market segments and drivers
- Oral antidiabetics account for 85% of market volume.
- Fixed-dose combination drugs constitute 40% of prescriptions in this segment.
- The entrance of newer classes (SGLT2 inhibitors and GLP-1 receptor agonists) influences market share dynamics.
- The increase in patient awareness and tighter glycemic control guidelines support oral therapy use.
Competition landscape
Major competitors include:
| Drug Name |
Class |
Launch Year |
Market Share (2022) |
Key Advantages |
| JANUMET XR |
DPP-4 inhibitor + Metformin ER |
2013 |
~12% |
Well-established efficacy, safety |
| Tradjenta (linagliptin) |
DPP-4 inhibitor |
2012 |
9% |
Once-daily dosing, no renal adjustments |
| Jardiance (empagliflozin) |
SGLT2 inhibitor |
2014 |
8% |
Cardiovascular benefits |
| Dapagliflozin (Farxiga) |
SGLT2 inhibitor |
2014 |
2% |
Kidney-friendly profile |
These agents have either expanded the therapeutic options or replaced older drugs in treatment algorithms.
Historical sales performance and projections
From 2015 to 2022, JANUMET/XR contributed steadily to sales growth for Merck & Co., with approximate revenues reaching USD 1.2 billion annually in 2022. The breakdown of sales indicates:
- Genericization pressures are emerging as patent exclusivity for JANUMET XR could expire in 2024 in some territories.
- Market share stability exists owing to brand recognition and clinician familiarity.
- Pricing strategies and reimbursement policies influence sales volume.
Sales projection methodology
Forecasts consider:
- Expected patent expirations starting 2024
- Adoption rates of SGLT2 and GLP-1 therapies
- The impact of reimbursement reforms
- Growth in the global diabetic population (estimated at 537 million in 2021, with a projected 643 million by 2030)
Based on these factors, sales are projected as follows:
| Year |
Sales Estimate (USD billion) |
Notes |
| 2023 |
1.35 |
Slight growth, patent protections remain |
| 2024 |
1.10 |
Patent expiration begins, biosimilar entry |
| 2025 |
0.95 |
Increased competition reduces revenues |
| 2026 |
0.85 |
Market shifts further toward newer therapies |
| 2027 |
0.78 |
Market consolidation, new entrants |
| 2030 |
0.65 |
Continued decline without innovation |
The decline reflects patent expiry impact and market shifts towards SGLT2 inhibitors and GLP-1 receptor agonists, which are preferred for their added benefits on weight and cardiovascular risk.
Strategic factors influencing future sales
- Patent expiration in key markets like the US and EU increases biosimilar competition.
- Line extension or new formulations could temporarily boost sales.
- Expansion into emerging markets will contribute marginally owing to price sensitivity and market access barriers.
- Physician and patient preferences favor drugs with additional benefits, favoring SGLT2 and GLP-1 classes.
Key Takeaways
- JANUMET XR commands a significant share within the DPP-4 inhibitor/metformin combination segment, but faces increased competition and patent expiration pressures.
- The overall oral antidiabetic market will grow, but JANUMET XR's sales are expected to decline gradually after 2024.
- The shift toward drugs with cardiovascular and weight-loss benefits will influence prescribing patterns.
- Patent protection expiration in 2024 may prompt biosimilar entries, impacting revenue.
- Merck's ability to innovate or reposition JANUMET XR for new indications could mitigate sales decline.
Key FAQs
1. When will JANUMET XR lose patent protection?
In the US, patent expiration is projected for late 2023 or early 2024. Patent protections in Europe may vary by country but are expected to follow similar timelines.
2. How does JANUMET XR compare with newer therapies?
It offers proven efficacy for glycemic control but lacks the cardiovascular and weight benefits of SGLT2 inhibitors and GLP-1 receptor agonists, which are increasingly preferred.
3. What percentage of diabetic patients are prescribed FDCs?
Approximately 40% of oral antidiabetic prescriptions are for fixed-dose combinations, favoring drugs like JANUMET XR for adherence.
4. What are the main risks to JANUMET XR sales?
Patent expiry leading to biosimilar competition, shifts in physician preference toward therapies with added benefits, and pricing pressures.
5. Are there expansion opportunities for JANUMET XR?
Moderate potential exists in emerging markets, though price sensitivity and existing competition limit growth. Innovation in formulations or indications could provide incremental sales.
References
- MarketWatch. (2023). Global Diabetes Drugs Market Size, Share & Trends Analysis Report.
- IQVIA. (2022). Prescription data for oral antidiabetics.
- U.S. Food and Drug Administration. (2013). FDA approvals of JANUMET XR.
- EvaluatePharma. (2022). Sales estimates and market forecasts for diabetes drugs.[1]
[1] EvaluatePharma. (2022). "Diabetes drugs sales forecast."