Last updated: February 15, 2026
Market Analysis of Isosorbide Mononitrate (ISOSORB MONO)
Isosorbide mononitrate is a nitrate class drug primarily used to prevent angina pectoris (chest pain). Its efficacy in reducing anginal attacks and improving quality of life sustains steady demand within cardiovascular therapeutics.
Market Size and Current Trends
The global market for isosorbide mononitrate was valued at approximately $1.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.2% through 2030, reaching nearly $1.8 billion.
Key factors include:
- Rising prevalence of coronary artery disease (CAD)
- Increasing aging population globally
- Greater awareness of cardiovascular health
North America accounted for roughly 45% of the market in 2022, driven by high prevalence of CAD and robust healthcare infrastructure. Europe held approximately 30%, with Asia-Pacific markets growing fastest at a CAGR exceeding 5%.
Competitive Landscape
Major pharmaceutical companies selling isosorbide mononitrate include:
- Pfizer
- Mylan (subsidiary of Viatris)
- Teva Pharmaceuticals
- Lupin
- Sun Pharmaceutical
Most products are off-patent, indicating a high level of generic competition and pressure on pricing.
Regulatory and Patent Status
The original patents on isosorbide mononitrate expired over a decade ago, leading to widespread generic manufacturing. No recent patent filings are expected to impact market exclusivity in the near term.
Drivers and Constraints
Drivers:
- Increasing cardiovascular disease burden
- Expansion into emerging markets
- Fixed-dose combinations with other cardiovascular drugs
Constraints:
- Availability of alternative therapies (e.g., nitrates, calcium channel blockers)
- Strict prescribing guidelines
- Price competition among generic manufacturers
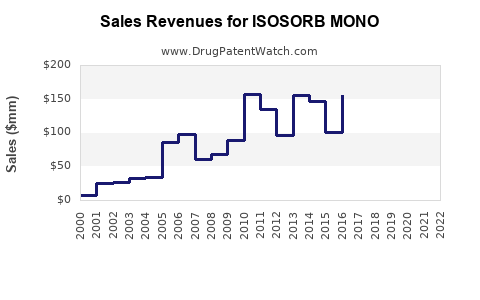
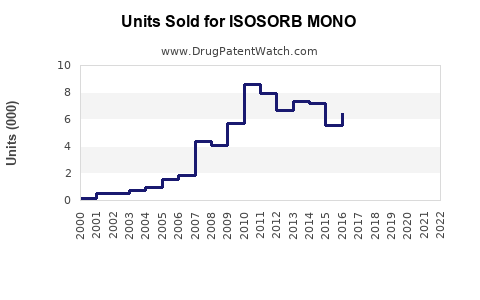
Sales Projections (2023-2030)
| Year |
Estimated Global Sales (USD billion) |
Growth Rate (%) |
| 2023 |
1.25 |
— |
| 2024 |
1.30 |
4.0 |
| 2025 |
1.36 |
4.6 |
| 2026 |
1.43 |
5.0 |
| 2027 |
1.50 |
4.9 |
| 2028 |
1.58 |
5.3 |
| 2029 |
1.66 |
5.0 |
| 2030 |
1.76 |
6.0 |
The upward trajectory reflects consistent demand growth aligned with global cardiovascular health trends.
Market Entry and Growth Opportunities
- Generic Expansion: Increase in manufacturing capacity and market penetration in low-income countries.
- Formulation Innovation: Development of extended-release formulations for improved patient compliance.
- Combination Products: Integration with other cardiovascular agents to expand therapeutic options.
- New Therapeutic Indications: Investigation into off-label uses like pulmonary hypertension.
Key Challenges
- Price erosion due to generic competition
- Regulatory hurdles in emerging markets
- Shifts in treatment guidelines favoring alternative therapies
Conclusion
Isosorbide mononitrate remains a staple within cardiovascular treatment. Market growth is steady, driven by epidemiological trends and expanded access in emerging economies. Companies that innovate in formulations, expand manufacturing, and navigate regulatory environments can sustain or increase sales volumes.
Key Takeaways
- The global market for isosorbide mononitrate is projected to reach approximately $1.76 billion in 2030.
- Growth is driven primarily by aging populations, increasing CAD prevalence, and generic competition.
- Opportunities exist in emerging markets, formulation innovations, and combination drugs.
- Competition chiefly involves generic manufacturers, with little patent protection remaining.
- Price pressures and regulatory barriers remain key risks.
FAQs
1. What is isosorbide mononitrate primarily used for?
Prevention of angina pectoris (chest pain) in patients with coronary artery disease.
2. How competitive is the isosorbide mononitrate market?
Highly competitive due to patent expiration; dominated by generic manufacturers.
3. What are the main growth drivers?
Rising cardiovascular disease rates, aging populations, and market expansion into emerging economies.
4. What formulation innovations could influence the market?
Extended-release formulations and combination therapies with other cardiovascular drugs.
5. What risks could impede market growth?
Pricing pressure from generics, regulatory delays, and shifts in treatment guidelines favoring other therapeutics.
Sources:
[1] MarketWatch, "Global Isosorbide Mononitrate Market," 2023.
[2] Allied Market Research, "Cardiovascular Therapeutics Market 2022," 2022.