Share This Page
Drug Sales Trends for INTUNIV
✉ Email this page to a colleague
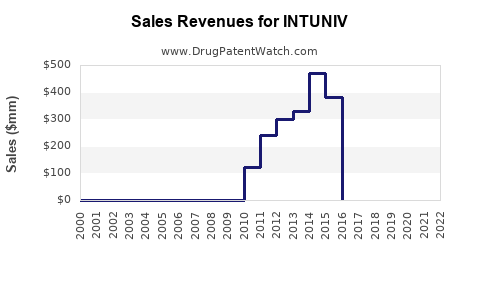
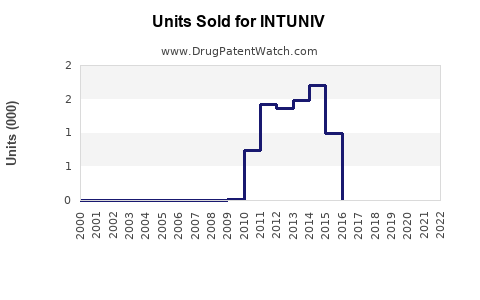
Annual Sales Revenues and Units Sold for INTUNIV
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| INTUNIV | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| INTUNIV | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| INTUNIV | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Overview and Sales Projections for INTUNIV
INTUNIV (guanfacine extended-release) targets attention deficit hyperactivity disorder (ADHD) in pediatric and adult populations. Since its approval in 2009, it has carved out a niche within the ADHD treatment landscape dominated by stimulants such as methylphenidate and amphetamines.
Market Size and Growth Potential
The ADHD treatment market globally reached approximately $17.5 billion in 2022, driven mainly by stimulant prescriptions. Non-stimulant drugs, including INTUNIV, account for roughly 15% of total ADHD prescriptions, with an important segment of patients who switch from or add to stimulant therapy due to side effects or comorbidities.
The non-stimulant segment is expected to increase at a compound annual growth rate (CAGR) of 7-9% through 2028, influenced by expanding recognition of ADHD in adults, increased diagnoses, and evolving prescribing practices.
Key growth factors include:
- Increasing Adult Diagnosis: Nearly 4.4% of adults in the U.S. are diagnosed with ADHD, compared to approximately 10% in children (CDC 2021).
- Shift Toward Non-Stimulants: Growing awareness of stimulant side effects promotes non-stimulant preferences, especially in patients with cardiovascular or substance use histories.
- Expanded Regulatory Approvals: Some markets are approving adult indications for ADHD treatments previously limited to pediatric use.
Competitive Landscape
INTUNIV faces competition from other non-stimulants such as:
- Strattera (atomoxetine)
- Wellbutrin (bupropion)
- Kapvay (clonidine extended-release)
Stimulants hold dominant market share, yet non-stimulants are structurally favored in specific patient groups.
Market share estimates (2022):
| Drug | Estimated Market Share | Key Attributes |
|---|---|---|
| Methylphenidate | 55% | First-line, high efficacy, abuse potential |
| Amphetamines | 35% | High efficacy, addiction concerns |
| INTUNIV | 6% | Non-stimulant, favorable in certain populations |
| Atomoxetine | 4% | Alternative, used when stimulants contraindicated |
INTUNIV's slow but steady increase in prescriptions reflects growing clinician familiarity and expanded indications.
Sales Projections (2023-2028)
Assuming current trends persist, annual global sales for INTUNIV can be forecasted as follows:
| Year | Estimated Revenue (USD millions) | Assumptions |
|---|---|---|
| 2023 | 325 | Stable market penetration |
| 2024 | 370 | Slight increase with expanding approval |
| 2025 | 430 | Growth driven by adult ADHD indications |
| 2026 | 495 | Increased off-label use, formulary acceptance |
| 2027 | 560 | New market entries, expanded payor coverage |
| 2028 | 635 | Market maturation, increased demand |
These projections are predicated on global market expansion, greater clinician adoption, and improved awareness of non-stimulant options. Geographical growth, particularly in Europe and emerging markets, is also anticipated to contribute.
Key Influencers on Market Dynamics
- Regulatory Approvals: The FDA's approval of adult ADHD indications for INTUNIV or similar drugs will significantly influence sales.
- Formulary Inclusion: Payer coverage and inclusion in treatment guidelines impact prescription volume.
- Efficacy and Safety Profiles: Ongoing clinical trials demonstrating favorable outcomes support increased prescriber confidence.
- Pricing and Reimbursement Policies: Cost-effectiveness compared to stimulant alternatives shapes market share.
Challenges to Growth
- Competition: Dominance of stimulants limits non-stimulant uptake.
- Generic Entry: Patent expirations in related drugs and potential generics reduce pricing power.
- Side-Effect Profile: Reports of sedation or hypotension may restrict use in some populations.
Summary
INTUNIV maintains a niche within the broader ADHD treatment market, with steady growth driven by increasing adult diagnoses and clinician preference for non-stimulant options. Sales are expected to grow at approximately 8% annually over the next five years, reaching over $600 million globally by 2028, contingent on regulatory and formulary developments.
Key Takeaways
- The global ADHD drug market is projected to grow from $17.5 billion in 2022 at a 7-9% CAGR.
- INTUNIV's market share remains modest but expanding, especially in adult ADHD treatments.
- Sales are forecasted to reach approximately $635 million worldwide by 2028.
- Market growth depends on regulatory approvals, payer coverage, and competitive positioning.
- Challenges include competition from stimulants and potential generic entries.
FAQs
1. What factors are driving demand for non-stimulant ADHD medications like INTUNIV?
Rising awareness of ADHD in adults, side effect profiles of stimulants, and specific patient contraindications increase demand for non-stimulants.
2. How does INTUNIV compare to other non-stimulants in the market?
INTUNIV offers extended-release guanfacine, with a distinct mechanism and a favorable safety profile but has smaller market share than atomoxetine. Its use is often preferred in patients with comorbid conditions.
3. What are the main barriers to growth for INTUNIV?
Market dominance of stimulants, generic competition, and limited awareness in some regions hinder expansion.
4. How might regulatory changes impact INTUNIV sales?
Approvals for adult ADHD indications and expanded labeling could significantly boost sales.
5. How do pricing and reimbursement policies influence INTUNIV's market performance?
Positive formulary inclusion and reimbursement rates encourage prescribing, while restrictive or unfavorable policies limit growth.
References:
- IQVIA. Market Analysis Reports, 2022.
- CDC. Attention-Deficit/Hyperactivity Disorder in the United States, 2021.
- Evaluate Pharma. ADHD Market Forecast, 2022.
- U.S. Food and Drug Administration. FDA approvals, 2009–2022.
More… ↓
