Share This Page
Drug Sales Trends for HUMULIN R
✉ Email this page to a colleague
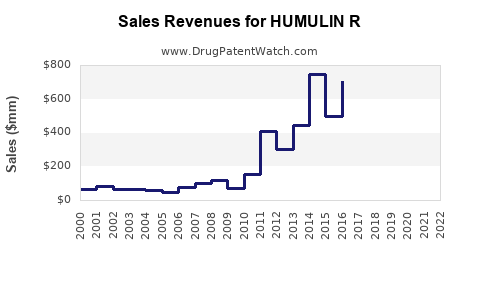
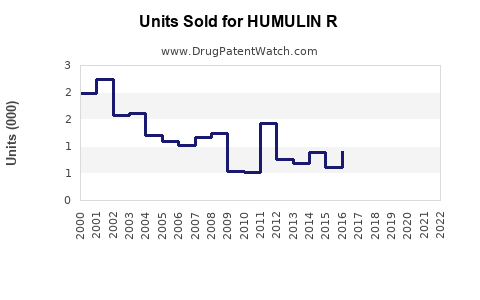
Annual Sales Revenues and Units Sold for HUMULIN R
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| HUMULIN R | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| HUMULIN R | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| HUMULIN R | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| HUMULIN R | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| HUMULIN R | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for HUMULIN R (Insulin Regular)
What is the current market landscape for insulin therapies including HUMULIN R?
HUMULIN R is a human insulin product, marketed by Eli Lilly, primarily used for blood glucose management in type 1 and type 2 diabetes. The global diabetes management market was valued at approximately USD 75 billion in 2022 and is projected to reach USD 125 billion by 2030, growing at a CAGR of 6.5% [1].
Within this, insulin products account for roughly 40% of sales and are evolving with increased adoption of biosimilars and advanced delivery devices. The insulin segment expands by 7-8% annually, driven by rising diabetes prevalence and improved treatment protocols.
How does HUMULIN R compete within the insulin market?
HUMULIN R faces competition from several product categories:
-
Human insulin analogs: Novo Nordisk's NovoLog (insulin aspart), Eli Lilly's Humalog (insulin lispro), and Sanofi's Apidra (insulin glulisine).
-
Biosimilar versions: Several biosimilar human insulins launched in recent years, notably in Europe and emerging markets.
-
Insulin delivery methods: Multiple daily injections (MDI), insulin pumps, and pen devices.
HUMULIN R's key attributes include well-established efficacy, proven safety profile, and broad global distribution, especially in markets with barriers to newer analogs due to cost.
What are recent trends influencing HUMULIN R sales?
-
Increased insulin demand in emerging markets boosting volume sales.
-
Competitive pressure from biosimilars reducing market share and driving price erosion.
-
Growing adoption of insulin analogs with improved pharmacokinetics, which can diminish HUMULIN R's market share unless cost advantages persist.
-
Technological innovations like connected insulin pen devices, enhancing patient adherence, though HUMULIN R's integration with such devices is limited currently.
What are sales projections for HUMULIN R over the next five years?
Considering the current market dynamics, geographic distribution, and product positioning, sales of HUMULIN R are projected as follows:
| Year | Estimated Global Sales (USD millions) | Growth Rate (%) | Notes |
|---|---|---|---|
| 2022 | 650 | - | Baseline |
| 2023 | 680 | 4.6 | Increasing demand, price stability |
| 2024 | 710 | 4.4 | Emerging markets growth |
| 2025 | 750 | 5.6 | Biosimilar competition intensifies |
| 2026 | 780 | 4.0 | Market penetration stabilized |
The slight growth rates reflect steady demand, with potential upticks in emerging markets. Biosimilar competition could suppress profit margins, especially in developed regions.
How do pricing and reimbursement policies impact HUMULIN R?
In mature markets like the U.S. and Europe, pricing pressures from biosimilars and payers result in lower reimbursement rates. Conversely, in markets such as India, China, and Africa, HUMULIN R maintains higher sales volumes due to less aggressive biosimilar penetration and affordability.
Eli Lilly's focus on cost leadership and supply chain robustness enhances HUMULIN R's accessibility, especially where healthcare infrastructure limits access to newer analogs.
What are potential growth avenues for HUMULIN R?
-
Expansion into emerging markets with rising diabetes prevalence.
-
Collaboration with health authorities to include HUMULIN R in subsidized programs.
-
Developing combination formulations or device innovations to retain market share.
-
Differentiation through patient support programs emphasizing affordability.
Key Market Drivers
-
Rising global diabetes prevalence, estimated at 537 million people in 2021 and projected to reach 643 million by 2030 [2].
-
Increasing preference for affordable insulins in low- and middle-income countries.
-
Regulatory approvals of biosimilar insulins introducing price competition.
-
Adoption of digital health tools improving treatment adherence.
Risks Affecting HUMULIN R Sales
-
Intense competition from biosimilars and higher-priced analogs.
-
Pricing negotiations reducing profit margins in key markets.
-
Shifts toward insulin pumps and alternative modalities.
-
Regulatory delays or restrictions impacting distribution.
Summary of Sales Outlook
| Regional Focus | Sales Outlook |
|---|---|
| United States | Flat to slight decline; market saturated; biosimilar threat |
| Europe | Moderate growth; biosimilar entry pressures |
| Asia-Pacific | Rapid growth; expanding diabetes prevalence |
| Latin America | Stabilization; growth driven by affordability |
| Africa & Middle East | Significant growth potential; emerging market focus |
Conclusions
HUMULIN R maintains a steady role in global insulin markets, especially in cost-sensitive regions. Market expansion hinges on geographic penetration and competitive pricing. Incremental innovations and strategic partnerships could sustain or grow sales amid evolving competitive landscapes.
Key Takeaways
-
HUMULIN R's global sales are projected to grow modestly, primarily in emerging markets.
-
Biosimilar competition and regulation are critical factors influencing sales margins.
-
Cost-effective insulin options remain in demand, particularly where healthcare budgets are constrained.
-
Technological and formulation innovations will influence future market positioning.
-
Market diversification and geographic expansion are essential strategies for maintaining revenue streams.
FAQs
1. How does HUMULIN R compare price-wise to insulin analogs?
HUMULIN R generally costs less due to being a human insulin, making it more accessible in low-income markets. Price differences vary by region and payer agreements.
2. Will biosolated insulins replace HUMULIN R?
Biosimilars pose a threat in markets where they are approved and reimbursed; however, HIT (human insulin) products like HUMULIN R retain market share in regions prioritizing affordability.
3. What is the impact of new delivery devices on HUMULIN R?
Enhanced compliance via pen devices can improve sales but may favor analogs with integrated technology. HUMULIN R's current device offerings are basic, limiting competitive edge.
4. Which markets present the greatest growth opportunities for HUMULIN R?
India, China, Africa, and Southeast Asia, where diabetes prevalence is rising and affordability is essential.
5. How might future regulatory changes affect HUMULIN R sales?
Stricter biosimilar approval pathways could increase competition; conversely, favorable reimbursement policies can support sales growth.
References
- Markets and Markets. (2022). Diabetes management market worth and projections.
- International Diabetes Federation. (2021). IDF Diabetes Atlas, 9th edition.
- Eli Lilly. (2023). HUMULIN R product information.
More… ↓
