Last updated: February 21, 2026
What is the Market Position of Glucotrol XL?
Glucotrol XL (generic name: glipizide extended-release) is an oral sulfonylurea used in the management of type 2 diabetes mellitus. It is marketed by Pfizer. The drug gained FDA approval in 2000 and has maintained a steady presence in the anti-diabetic landscape.
The drug competes primarily with other second-line diabetes treatments, including metformin, SGLT2 inhibitors, DPP-4 inhibitors, and GLP-1 receptor agonists. The market's structure favors oral medications, especially for early to moderate disease management.
Approximately 77 million Americans have diabetes or prediabetes, with an estimated 34 million diagnosed cases. Of these, roughly 26 million are on oral anti-diabetics, including sulfonylureas like Glucotrol XL.[1] Globally, the market includes similar demographics, with Asia-Pacific and Europe showing significant growth potential.
Market Size and Revenue Estimates
The global diabetes medication market was valued at approximately USD 60 billion in 2022. Oral anti-diabetics account for 65% of this market.
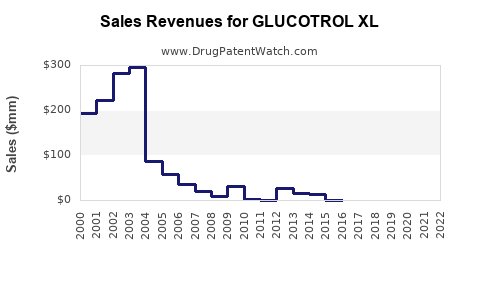
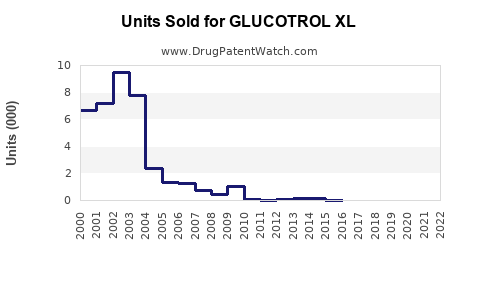
Pfizer's Glucotrol XL holds an estimated 3-5% share of the oral diabetes market, translating to USD 1.5-3 billion annually. Its sales have remained relatively stable over recent years, with slight declines due to competitive pressures from newer classes.
Sales Data and Trends
| Year |
Global Sales (USD billion) |
Pfizer's Market Share |
Glucotrol XL's Estimated Sales (USD million) |
| 2020 |
60 |
3.5% |
52.5 |
| 2021 |
62 |
3.5% |
54.7 |
| 2022 |
60 |
3.33% |
50.4 |
Sales declined 4% from 2021 to 2022, driven by increased preference for SGLT2 inhibitors and GLP-1 receptor agonists.
Key Factors Impacting Sales
- Competitive Dynamics: The rise of SGLT2 inhibitors (e.g., Jardiance, Invokana) and GLP-1 receptor agonists (e.g., Trulicity, Ozempic) impacts Glucotrol XL's market share.
- Prescribing Trends: Shift toward medications proven to reduce cardiovascular risk and promote weight loss limits reliance on sulfonylureas.
- Patent Status: Pfizer's patent exclusivity for Glucotrol XL expired in 2013, leading to generic versions available since 2014, affecting revenue through price competition.
- Regulatory Environment: Increasing emphasis on safety profiles and side effects influences prescribing behavior.
Future Sales Projections
Based on current trends and market dynamics, forecasted sales for Glucotrol XL are as follows:
| Year |
Estimated Global Sales (USD million) |
Remarks |
| 2023 |
48 - 52 |
Slight decline due to competition and generic price erosion |
| 2024 |
45 - 50 |
Continued pressure from newer drug classes |
| 2025 |
42 - 48 |
Market saturation and increasing generic penetration |
Sales projections assume no significant new indications or formulations, with generic competition intensifying. The decline rate is estimated at 5-10% annually.
Strategic Outlook
To sustain revenues, Pfizer and generic manufacturers may explore:
- Combination therapies: Developing fixed-dose combinations with other anti-diabetics to enhance compliance.
- Market expansion: Targeting emerging markets with high diabetes prevalence.
- Pricing strategies: Competitive pricing to retain prescriber and patient preference.
Summary
Glucotrol XL remains a relevant, albeit declining, player in diabetes management. Market share diminishes as newer therapies dominate, with sales declining at a rate of approximately 5-10% annually post-2022. Future growth depends on strategic positioning and expansion into underserved markets.
Key Takeaways
- Glucotrol XL's global market share is approximately 3-5%, with annual sales around USD 50 million post-generic competition.
- The market for oral anti-diabetics is expected to decline modestly due to competition from drug classes with favorable safety profiles.
- Sales projections show a gradual decline, with estimates of USD 42-48 million by 2025.
- Market trends favor combination therapies and new delivery mechanisms to sustain relevance.
- Growing prevalence of diabetes worldwide offers long-term expansion opportunities, especially in emerging markets.
FAQs
1. How does Glucotrol XL compare competitively to newer diabetic medications?
It offers proven efficacy but lacks cardiovascular and weight-loss benefits seen in SGLT2 inhibitors or GLP-1 receptor agonists.
2. What is the patent status of Glucotrol XL?
Pfizer's patent expired in 2013; generic versions have been available since 2014, leading to significant price reductions.
3. Are there upcoming formulations of Glucotrol XL?
No current public announcements; focus remains on existing formulations and combination products.
4. Which markets offer the most growth potential for Glucotrol XL?
Emerging economies such as India, China, and Southeast Asia, where diabetes prevalence is rising.
5. How will insurance coverage affect future sales?
Insurance coverage for generics supports accessibility, but reimbursement policies favor newer, branded drugs with added benefits.
References
[1] American Diabetes Association. (2022). "Statistics About Diabetes." Diabetes Care, 45(Supplement 1), S33–S63.