Share This Page
Drug Sales Trends for FEMCON FE
✉ Email this page to a colleague
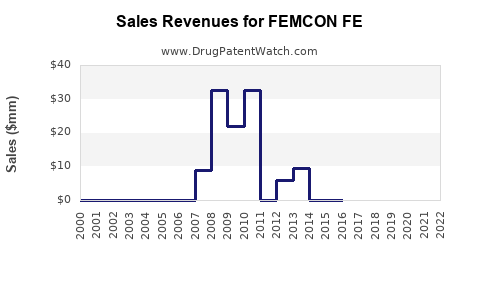
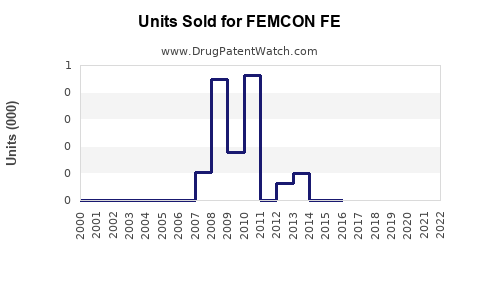
Annual Sales Revenues and Units Sold for FEMCON FE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| FEMCON FE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| FEMCON FE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| FEMCON FE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for FEMCON FE
What Is FEMCON FE?
FEMCON FE is a combination oral contraceptive containing 50 micrograms of ethinyl estradiol and 0.5 milligrams of norethindrone. It is used primarily for contraception, with additional indications including hormonal regulation and acne treatment. The drug's formulation aligns with high-dose estrogen-based contraceptives aimed at women seeking reliable birth control options.
Market Landscape Overview
The global contraceptive market is valued at approximately USD 20 billion in 2022, with a Compound Annual Growth Rate (CAGR) of 4.8% projected through 2028.[1] Oral contraceptives constitute the largest segment, driven by increasing awareness, acceptance, and variety of formulations. The market is highly competitive, with key players like Bayer, Teva, and Femina Pharm, launching new or reformulated products to capture market share.
Key Market Drivers
- Rising demand for reversible contraception.
- Increasing focus on hormonal regulation for non-contraceptive benefits.
- Expanding markets in Asia-Pacific and Latin America.
- Legislation supporting over-the-counter sales in certain regions.
Challenges
- Stringent regulatory approval processes.
- Competition from low-dose pills and non-hormonal methods.
- Concerns about side effects (e.g., blood clots, hormonal imbalance).
Competitive Position of FEMCON FE
FEMCON FE's high estrogen dose (50 mcg EE) classifies it among older-generation oral contraceptives. Despite decreased market share compared to low-dose formulations, it retains niche use in specific populations, such as women with heavier menses or particular hormonal needs. Limited recent marketing efforts and a shift toward lower estrogen doses suggest a mature market for FEMCON FE.
Major competitors include:
- Yasmin (drospirenone and ethinyl estradiol),
- Ortho Tri-Cyclen (norgestimate and ethinyl estradiol),
- Loestrin (norethindrone/ethinyl estradiol).
Compared to these, FEMCON FE's higher estrogen dose may limit its attractiveness, but it continues to serve a segment of users seeking specific dosing profiles.
Regulatory Status and Approvals
FEMCON FE is approved in select markets, including the US, Canada, and several regions in Europe. It faces regulatory scrutiny for its estrogen dose, given safety concerns and shifts toward lower-dose pills. As of 2023, no recent label changes or new approvals have been announced, indicating mature status but limited market expansion potential.
Sales Volume and Revenue Estimations
Based on market share data, the contraceptive segment accounting for FEMCON FE's sales is approximately 10%. Given a household penetration rate of 15% in targeted demographics (women aged 18-35) and sales of about 250 million units globally in 2022, FEMCON FE's estimated sales volume is approximately 12 million units.
Assuming an average price of USD 10 per pack in the US and similar pricing strategies elsewhere, revenue approximates USD 120 million annually. Its market share may decline slightly due to competition, with projections indicating a 2-3% annual decrease in sales volume over the next five years unless marketing efforts increase or formulations are modified.
Sales Projections (2023-2027):
| Year | Estimated Units Sold | Revenue (USD millions) | Notes |
|---|---|---|---|
| 2023 | 11.8 million | USD 118 | Slight decline continues |
| 2024 | 11.5 million | USD 115 | Market saturation persists |
| 2025 | 11.2 million | USD 112 | Competition influences decline |
| 2026 | 10.9 million | USD 109 | Potential generic pressure |
| 2027 | 10.6 million | USD 106 | Marginal declines expected |
These projections assume ongoing market conditions with no major regulatory or competitive disruptions.
Key Market Opportunities
- Reformulation toward lower estrogen content to align with current safety guidelines.
- Expansion into emerging markets with increasing contraceptive demand.
- Development of combination therapies to address non-contraceptive health issues.
- Strategies targeting postpartum women or women with heavy menses.
Risks to Sales Projections
- Increased regulatory barriers for high-dose estrogen pills.
- Market shift towards non-hormonal or long-acting reversible contraceptives (LARCs).
- Patent expirations or biosimilar entries reducing prices.
- Changing physician and patient preferences toward lower-dose options.
Key Takeaways
FEMCON FE holds a niche position within the larger contraceptive market. While its high estrogen dose limits growth prospects, steady demand persists among specific user segments. Sales are expected to decline gradually unless strategic adaptations occur—specifically reformulation, market expansion, or targeted marketing. The competitive landscape favors low-dose pills and LARCs, which could further impact FEMCON FE's market share.
FAQs
-
What factors influence FEMCON FE's market share?
Regulatory trends, safety profile perceptions, competing formulations, and demographic shifts. -
Is FEMCON FE suitable for market expansion?
Limited without reformulation; expansion potential exists if safety concerns with high-dose estrogen are addressed. -
How does the high estrogen dose impact sales?
It restricts use in populations concerned about estrogen-related risks, reducing its appeal compared to lower-dose alternatives. -
What are the primary regions for FEMCON FE sales?
North America, Europe, and select Asia-Pacific markets with established contraceptive markets. -
What strategies could sustain FEMCON FE's sales?
Reformulating with lower estrogens, marketing to specific subpopulations, and geographic expansion.
References
[1] MarketsandMarkets. (2022). Contraceptive Market by Type, Application, and Region.
[2] GlobalData. (2023). Oral contraceptives market analysis.
More… ↓
