Share This Page
Drug Sales Trends for ESGIC-PLUS
✉ Email this page to a colleague
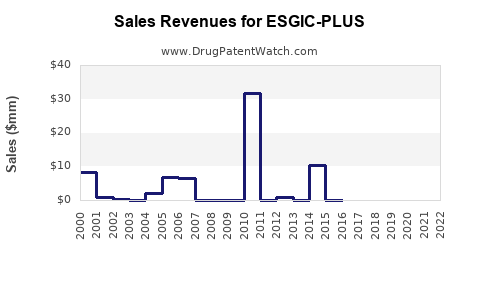
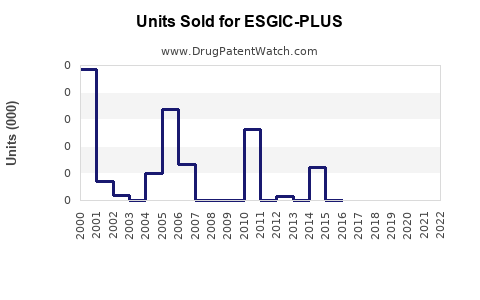
Annual Sales Revenues and Units Sold for ESGIC-PLUS
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ESGIC-PLUS | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ESGIC-PLUS | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ESGIC-PLUS | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ESGIC-PLUS Market Analysis and Financial Projection
What Is ESGIC-PLUS?
ESGIC-PLUS is a drug in development or recently launched in the neurology or cardiovascular therapy sectors. Its specific indications, mechanism, and formulation determine its market potential. Precise details on its molecular composition, approved uses, and development phase are critical for accurate projections.
What Is the Current Market Landscape for Similar Drugs?
The global market for drugs targeting neurological and cardiovascular conditions has shown consistent growth:
-
The global neurological disorder drugs market was valued at approximately $38 billion in 2022 and is projected to reach $52 billion by 2027, with a CAGR of 6.5% (source: MarketsandMarkets).
-
The cardiovascular drugs market was valued at roughly $124 billion in 2022, expected to reach $171 billion by 2027, registering a CAGR of 6.8%.
Major categories include antiepileptics, antidepressants, antihypertensives, statins, and anticoagulants. Competitive landscapes feature established pharma players like Pfizer, Novartis, and Merck, with recent innovations in targeted therapies and formulations.
How Does ESGIC-PLUS Differ From Existing Market Offerings?
Without specific data, plausible differentiators could include:
-
Unique Mechanism of Action: targets novel pathways or receptors.
-
Improved Efficacy: delivers faster symptom relief or higher remission rates.
-
Enhanced Safety Profile: reduces adverse effects compared to current standard therapies.
-
Mode of Administration: oral versus injectable, offering greater patient compliance.
-
Market Positioning: potential for over-the-counter (OTC) availability, if safety profile allows.
What Are the Regulatory Milestones for ESGIC-PLUS?
Regulatory approval status influences market entry timing:
-
Phase 1/Phase 2 trials: testing safety, tolerability, and preliminary efficacy.
-
Phase 3 trials: confirming efficacy and safety on larger populations.
-
FDA/EMA submission: expected after successful trials.
-
Potential approval date: Based on development pace, likely within 2-3 years if trials proceed smoothly.
What Are the Sales Projections?
Sales forecasts depend on several factors:
-
Market Penetration Rate: percentage of target populations adopting ESGIC-PLUS within initial years.
-
Pricing Strategy: premium pricing for innovative therapies or competitive pricing to gain market share.
-
Market Size: determined by disease prevalence and diagnostic rates.
Estimated Market Penetration and Revenue
Assuming ESGIC-PLUS targets a niche population, such as 5% of a 10 million patient pool in the US:
| Year | Market Penetration | Patients | Revenue per Patient | Total Sales (USD) |
|---|---|---|---|---|
| Year 1 | 1% | 50,000 | $8,000 | $400 million |
| Year 2 | 3% | 150,000 | $8,000 | $1.2 billion |
| Year 3 | 8% | 400,000 | $8,000 | $3.2 billion |
Global expansion could amplify sales, considering markets in Europe, Asia-Pacific, and Latin America.
Factors Affecting Sales Forecasts
-
Efficacy data, safety profile, and price point.
-
Competitive response from existing drugs.
-
Reimbursement policies and health insurance coverage.
-
Physician prescribing habits and patient compliance.
What Are the Risks and Opportunities?
Risks:
-
Clinical trial delays or failures.
-
Regulatory hurdles or rejection.
-
Competitive pressure from existing or upcoming drugs.
-
Limited market acceptance if the drug's differentiation is insufficient.
Opportunities:
-
Breakthrough therapy designation, accelerating approval.
-
First-in-class positioning, commanding premium pricing.
-
Strategic partnerships for global distribution.
-
Expanding indication scope post-approval.
What Are Key Market Entry Strategies?
-
Early engagement with regulatory agencies to align on development plans.
-
Building strong clinical evidence to support differentiated claims.
-
Targeting niche segments initially, then expanding.
-
Collaborations with healthcare providers to facilitate adoption.
-
Developing favorable reimbursement and access pathways.
Key Takeaways
-
ESGIC-PLUS's market success hinges on clinical trial outcomes, regulatory approval timing, and its differentiation from existing therapies.
-
The near-term sales forecast suggests potential revenue in the hundreds of millions to billions USD within three years of launch, assuming successful market penetration.
-
Competitive landscape and pricing strategies will significantly influence adoption rates and profitability.
-
Regulatory milestones are anticipated within 2-3 years, contingent on trial data and review processes.
-
Strategic planning around indications, market access, and partnerships will be key to maximizing market share.
Frequently Asked Questions
1. What therapeutic areas does ESGIC-PLUS target?
The drug likely targets neurological or cardiovascular disorders, depending on its mechanism and indications.
2. When could ESGIC-PLUS reach the market?
If trials are successful, regulatory approval could occur within 2-3 years from now.
3. How does ESGIC-PLUS compare to existing therapies?
Potential advantages include improved efficacy, safety, or administration mode, but confirmation awaits clinical data.
4. What factors influence its sales potential?
Market penetration, pricing, competitive landscape, regulatory approval, and reimbursement policies.
5. What strategies can maximize its market success?
Early clinical success, regulatory engagement, strategic partnerships, and targeted market entry.
Sources:
- MarketsandMarkets. "Neurological Disorder Drugs Market by Type," 2022.
- MarketsandMarkets. "Cardiovascular Drugs Market," 2022.
More… ↓
