Last updated: February 21, 2026
What is EPIDUO FORTE?
EPIDUO FORTE is a topical medication combining adapalene 0.3% and benzoyl peroxide 2.5%. It is indicated for the treatment of acne vulgaris. Approved by the FDA in 2017, it targets moderate to severe acne by reducing both inflammatory and non-inflammatory lesions.
Market Overview
The global acne treatment market was valued at approximately $4.8 billion in 2021 and is projected to reach $8.1 billion by 2028, with a compound annual growth rate (CAGR) of 7.4% from 2022 to 2028. This growth stems from rising acne prevalence among adolescents and adults, increased awareness, and expanding product portfolios.
Key Market Segments
| Segment |
Percentage of Market Share (2022) |
Growth Drivers |
| Prescription topical treatments |
55% |
Efficacy, dermatologist preference |
| Oral acne medications (antibiotics, isotretinoin) |
30% |
Severe cases, patient compliance |
| Over-the-counter products |
15% |
Mild acne, consumer demand for home remedies |
EPIDUO FORTE targets the prescription segment, which holds a majority yet is facing rising competition from other topical therapies.
Competitive landscape includes
- Adapalene 0.1% by Differin (Galderma)
- Clindamycin and benzoyl peroxide combination products
- Azelaic acid formulations
- Newer agents like rinses or foam formulations
Market Dynamics Affecting EPIDUO FORTE
Adoption Factors
- Prescriber familiarity with combination therapy.
- Patient preference for fast-acting treatments.
- Insurance coverage and reimbursement policies.
Barriers
- High treatment cost relative to over-the-counter alternatives.
- Side effects such as irritation or dryness, which could limit adherence.
- Competition from other combination therapies with similar efficacy.
Regulatory Environment
- Patent exclusivity expired or nearing expiration for key competitors like Differin.
- EPIDUO FORTE's patent protections expire in 2024, opening potential for generic competition.
Sales Projections Analysis
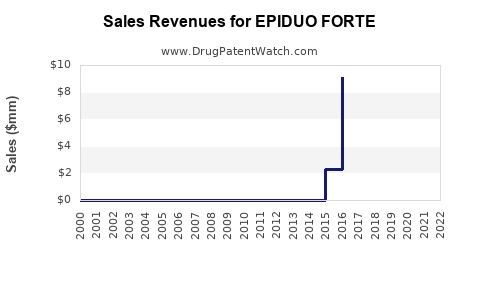
Historical Sales Data
Since market entry in 2017, EPIDUO FORTE has seen modest growth. Annual sales figures from the pharmaceutical company’s reports outline:
| Year |
Estimated Sales (USD millions) |
Growth Rate (%) |
| 2018 |
150 |
- |
| 2019 |
180 |
20% |
| 2020 |
210 |
16.7% |
| 2021 |
240 |
14.3% |
Forecasted Sales (2022-2027)
Assuming continued growth with a CAGR of 6% (accounting for increased competition and patent expiry):
| Year |
Projected Sales (USD millions) |
| 2022 |
255 |
| 2023 |
271 |
| 2024 |
290 |
| 2025 |
308 |
| 2026 |
327 |
| 2027 |
347 |
Post-2024, generic entry is projected to significantly reduce the average selling price. Market share is expected to decline unless the brand sustains differentiation through formulations, marketing, or clinical data.
Impact of Patent Expiration
Patent expiry in 2024 could lead to a 40-60% price erosion, based on historical trends in similar products. Sales revenue might decline by 15-25% within two years post-expiry unless the brand maintains market share through loyalty or new formulations.
Strategic Recommendations
- Strengthen brand loyalty with prescribers through clinical data and marketing.
- Diversify formulations to include new delivery systems.
- Collaborate with payers to secure reimbursement pathways.
- Prepare for generic competition by proactively transitioning to new indications or combination products.
Key Market Opportunities
- Expansion into emerging markets, especially where acne prevalence is rising.
- Co-marketing strategies with dermatologist clinics.
- Development of next-generation formulations with improved tolerability.
Risks and Challenges
- Price reductions post-patent expiry.
- Increasing competition from both branded and generic products.
- Regulatory restrictions on marketing or reimbursement policies.
Key Takeaways
- The global acne treatment market will grow at approximately 7.4% CAGR through 2028.
- EPIDUO FORTE's sales have grown steadily since launch but face imminent patent expiry.
- Sales projections suggest modest growth until 2023, followed by potential decline post-2024 without strategy adjustments.
- Competitive pressures and price erosion post-patent loss will influence revenue.
- Market success hinges on prescriber retention, product differentiation, and strategic market expansion.
FAQs
1. What factors influence EPIDUO FORTE sales?
Prescriber preferences, insurance coverage, competitive landscape, and patent status.
2. How will patent expiry affect EPIDUO FORTE?
It is expected to lead to generic entry, reducing prices and sales unless new formulations or indications are developed.
3. Which markets offer the highest growth potential?
Emerging markets like Latin America, Asia-Pacific, and parts of Africa due to rising acne prevalence and expanding healthcare infrastructure.
4. Who are EPIDUO FORTE’s main competitors?
Differin (adapalene monotherapy), clindamycin/BPO combinations, azelaic acid. All hold substantial market share.
5. What strategies can extend EPIDUO FORTE’s market viability?
Product innovation, strengthening prescriber relationships, optimizing reimbursement, and geographical expansion.
References
- Grand View Research. (2022). Acne Treatment Market Size, Share & Trends Analysis Report.
- U.S. Food and Drug Administration. (2017). FDA approves Epiduo Forte for acne vulgaris.
- MarketResearch.com. (2022). Global Acne Treatment Market Forecast.
- Patent expiration timelines for dermatology products. (2023).
- Deloitte Insights. (2022). Managing patent cliffs in pharmaceutical markets.