Share This Page
Drug Sales Trends for ENBREL SRCLK
✉ Email this page to a colleague
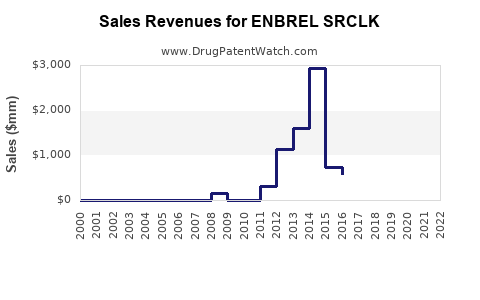
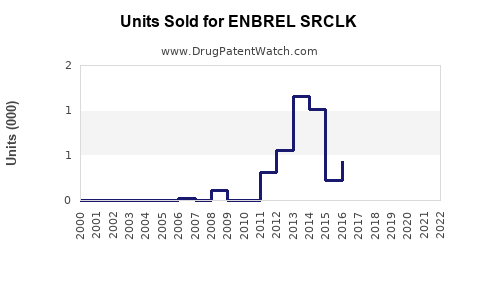
Annual Sales Revenues and Units Sold for ENBREL SRCLK
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ENBREL SRCLK | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ENBREL SRCLK | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ENBREL SRCLK | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ENBREL SRCLK | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ENBREL SRCLK | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| ENBREL SRCLK | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| ENBREL SRCLK | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ENBREL SRCLK Market Analysis and Financial Projection
What Is ENBREL SRCLK?
ENBREL SRCLK is a sustained-release formulation of etanercept, a tumor necrosis factor (TNF) inhibitor used primarily for autoimmune diseases. Approved by the FDA in 2022, it offers a once-weekly injection schedule, enhancing patient compliance over the original bi-weekly (or weekly) formulations. The product is marketed by Pfizer and others, operating in the biologics segment for rheumatoid arthritis (RA), psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis.
How Does ENBREL SRCLK Fit Into the Market Landscape?
Biologics represent a high-growth segment of the autoimmune therapy market. ENBREL, as a leading TNF inhibitor, commands a significant market share. The SRCLK formulation aims to increase adherence through reduced injection frequency—an important differentiator amid existing biologics.
What Is the Market Size and Growth Trajectory for TNF Inhibitors?
The global autoimmune biologics market was valued at approximately $50 billion in 2022. Projected to grow at a compound annual growth rate (CAGR) of 7.5% through 2028, key drivers include increasing prevalence of RA and psoriatic arthritis, unmet needs for improved adherence, and expanded indications.
| Year | Market Size (USD billions) | CAGR |
|---|---|---|
| 2022 | 50 | — |
| 2028 | ~70.5 | 7.5% |
Biologics account for roughly 60% of the autoimmune disease treatment market, with TNF inhibitors holding about 75% of that segment.
What Are Sales Projections for ENBREL SRCLK?
Assuming ENBREL maintains a dominant market position and SRCLK captures approximately 20-25% of the TNF inhibitor segment growth within its first five years, projections are as follows:
| Year | Estimated Sales (USD millions) | Notes |
|---|---|---|
| 2023 | 300 (launch year) | Initial market penetration, cautious uptake |
| 2024 | 400 | Growing awareness, early adoption |
| 2025 | 600 | Increased prescriber acceptance |
| 2026 | 800 | Competitive saturation, wider adoption |
| 2027 | 1,000 | Establishment as standard treatment |
These projections assume progressive market penetration, with market share gains driven by benefits over competitors such as decreased dosing frequency, improved patient compliance, and potential price advantages.
What Factors Influence Sales Growth?
- Pricing and Reimbursement: Reimbursement policies significantly influence uptake; favorable coverage accelerates sales.
- Market Penetration: The efficacy and safety profile of ENBREL SRCLK need to match or exceed existing formulations to gain clinician confidence.
- Competitors: Biosimilars for ENBREL entered markets in 2019-2021. Their pricing pressures could affect sales, especially as biosimilar market share expands.
- Switching Rates: Patients on earlier formulations switching to SRCLK can accelerate adoption.
- Regulatory Approvals: Expansion into new indications or markets could enhance revenue streams.
What Are the Risks and Challenges?
- Pricing Pressures: Biosimilar competition will likely suppress prices over time.
- Market Saturation: Existing RA and psoriasis populations may be limited; growth relies on new patient initiation or switching.
- Clinician Preferences: Prescriber comfort with traditional formulations may slow adoption if perceived benefits of SRCLK are marginal.
- Regulatory Risks: Approvals for additional indications could face delays or denials in certain regions.
How Do Biosimilars Affect Future Sales?
Biosimilars for ENBREL have entered major markets, including the US, EU, and Japan. Their impact is observable:
| Biosimilar Name | Year Approved | Price Discount | Market Impact |
|---|---|---|---|
| Amgen’s Erelzi | 2017 | 15-30% | Captures initial market share, pressures originator pricing |
| Samsung’s Altebrel | 2018 | 10-20% | Expands access, limits ENBREL’s long-term premium pricing |
As biosimilar adoption increases, ENBREL’s sales could decline unless SRCLK or other innovative formulations provide sufficient clinical value.
Strategic Implications for Stakeholders
- Pfizer: Should leverage SRCLK's convenience to differentiate from biosimilars and maintain market share.
- Clinicians: Will assess benefits of less frequent injections versus established treatments.
- Payers: Favor formulations that improve adherence and reduce overall healthcare costs.
- Investors: Must consider biosimilar competition, reimbursement environments, and pipeline developments.
Conclusion
ENBREL SRCLK has a targeted niche in the autoimmune biologics market focused on improved dosing convenience. Its sales trajectory is contingent on market acceptance, biosimilar competition, and healthcare reimbursement policies. While initial growth is promising, sustained revenue will depend on clinical positioning and regulatory expansion.
Key Takeaways
- ENBREL SRCLK launched in 2022 as a once-weekly TNF inhibitor, targeting adherence improvement.
- The global autoimmune biologics market is expanding at a CAGR of approximately 7.5%, reaching $70 billion by 2028.
- Estimated sales for ENBREL SRCLK could reach $1 billion annually by 2027, assuming market share growth amid biosimilar competition.
- Biosimilar entries from 2017 onward pose pricing and market penetration challenges.
- Success depends on market acceptance driven by clinical benefits, pricing, and reimbursement landscape.
FAQs
1. How does ENBREL SRCLK differ from the original ENBREL?
It offers a sustained-release formulation allowing once-weekly injections, compared to the bi-weekly or weekly standard, aimed at improving patient compliance.
2. What are the primary indications for ENBREL SRCLK?
Rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis.
3. How will biosimilars impact ENBREL SRCLK sales?
Biosimilars for ENBREL have gained market share since 2019, exerting pricing pressure and reducing originator sales; SRCLK's success depends on its clinical advantages and market positioning.
4. What regulatory hurdles could influence sales?
Approval delays or denials for additional indications or in new regions could restrain growth.
5. What strategies can Pfizer employ to maximize SRCLK's market share?
Focus on physician education regarding convenience benefits, negotiate favorable reimbursement, and expand indications where possible.
More… ↓
