Last updated: February 19, 2026
Eliquis (apixaban) is an oral anticoagulant developed by Bristol-Myers Squibb (BMS) and Pfizer. It is used for stroke prevention in non-valvular atrial fibrillation, treatment of venous thromboembolism (VTE), and reduction of risk in orthopedic surgeries. The drug belongs to the direct factor Xa inhibitors class, competing with drugs like Xarelto (rivaroxaban) and Pradaxa (dabigatran).
Global Market Overview
The global anticoagulant market was valued at $10.4 billion in 2022 and is projected to reach $17.8 billion by 2030, expanding at a compound annual growth rate (CAGR) of 7.0% (Research and Markets, 2023). Eliquis accounts for approximately 45% of this market share, making it a dominant product in the sector.
Key Growth Drivers
- Increasing prevalence of atrial fibrillation, VTE, and venous diseases.
- Growing adoption of direct oral anticoagulants (DOACs) over warfarin due to better efficacy and safety profiles.
- Rising geriatric populations globally.
- Expanded clinical indications and approvals, including extended dosing for VTE treatment.
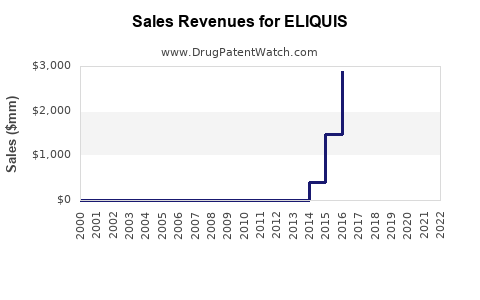
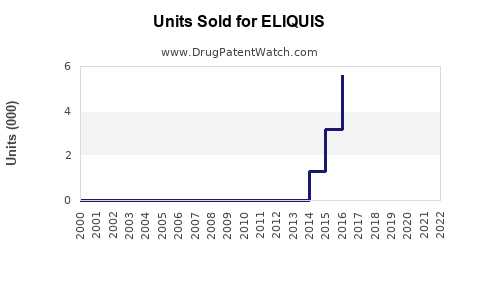
Sales Performance (2022-2023)
| Year |
Global Sales (USD billion) |
Market Share of Eliquis |
| 2022 |
7.6 |
40% |
| 2023 |
8.5 |
42% |
Eliquis sales increased by approximately 12% year-over-year, driven by acceptance in new markets and expanding indications.
Regional Market Distribution
| Region |
2023 Sales (USD billion) |
Market Share |
Key Factors |
| North America |
4.5 |
52% |
High prevalence of atrial fibrillation, widespread physician adoption |
| Europe |
2.2 |
26% |
Growing awareness, regulatory approvals |
| Asia-Pacific |
1.0 |
12% |
Emerging markets, expanding access |
| Rest of World |
0.8 |
10% |
Limited penetration, supply chain issues |
Competitive Landscape
| Drug |
Market Share (2023) |
Key Indications |
Price Range (per course) |
| Eliquis |
42% |
Stroke prevention, VTE |
$380 - $470 |
| Xarelto |
37% |
Same |
$350 - $450 |
| Pradaxa |
12% |
Stroke prevention |
$330 - $410 |
| Edoxaban |
4% |
VTE |
$400 |
Eliquis maintains leadership due to superior safety profile and extensive clinical trial data.
Future Sales Projections
Based on current trends and pipeline advancements, Eliquis' sales are forecasted to grow at a CAGR of 8-10% over the next five years, reaching approximately $13.5 billion to $15 billion in global revenue by 2028.
Key assumptions include:
- Expansion into new indications, including prophylactic uses for other thrombotic conditions.
- Increased adoption in emerging markets driven by regulatory approvals and healthcare infrastructure improvements.
- Increased dosing flexibility and patent protection until at least 2030.
Patent and Regulatory Outlook
Eliquis' primary patents in the U.S. expire around 2026-2028. Patent challenges and biosimilar entrants are anticipated, potentially affecting pricing and market share. Regulatory reviews are ongoing in markets like Japan, India, and China, with approvals expected in the next 1-2 years.
Market Risks
- Competitive pressures from biosimilars and next-generation anticoagulants.
- Regulatory hurdles in emerging markets delaying access.
- Cost containment policies and reimbursement restrictions.
Summary
Eliquis remains a leading anticoagulant with strong sales growth driven by expanding indications, favorable safety profile, and high adoption rates. Future growth hinges on market penetration in Asia-Pacific, pipeline approvals, and management of patent expirations.
Key Takeaways
- Eliquis commands a dominant, growing share of the anticoagulant market.
- Sales reached approximately $8.5 billion in 2023, with projections to surpass $13 billion by 2028.
- Market growth is fueled by increasing prevalence of thrombotic conditions and shifting physician preferences toward DOACs.
- Patent expirations and biosimilar competition could influence future pricing and market share.
- Regional expansion into emerging markets presents significant growth opportunities.
FAQs
1. What are the main competitors to Eliquis?
Xarelto (rivaroxaban) and Pradaxa (dabigatran) are the primary competitors, with Xarelto holding a slightly larger market share.
2. How does Eliquis differentiate itself from competitors?
It has a high safety profile, fewer drug interactions, and demonstrated efficacy in multiple indication types, backed by extensive clinical trial data.
3. When do Eliquis patents expire?
Patent protection in the U.S. is scheduled to expire between 2026 and 2028, opening the market for biosimilar competition.
4. What are the key markets for future growth?
Emerging markets in Asia-Pacific, expansion into new therapeutic areas, and increased use in prophylactic indications.
5. How have recent regulatory decisions affected Eliquis sales?
Approvals in new markets and expanded indications have maintained growth trajectories; delays or restrictions could impact future sales.
References
[1] Research and Markets. (2023). Global anticoagulant market forecast and trends.
[2] Bristol-Myers Squibb. (2022). Eliquis clinical data and commercial update.
[3] IQVIA. (2023). Global pharmaceutical market insights.