Share This Page
Drug Sales Trends for EFFEXOR XR
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for EFFEXOR XR (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
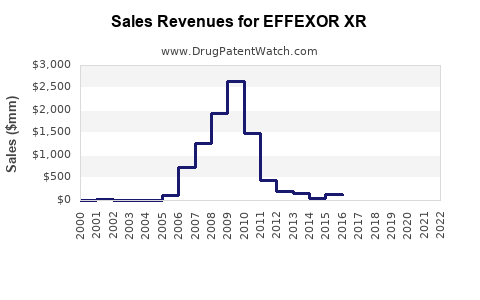
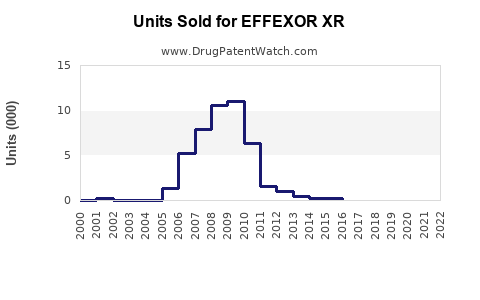
Annual Sales Revenues and Units Sold for EFFEXOR XR
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| EFFEXOR XR | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| EFFEXOR XR | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| EFFEXOR XR | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| EFFEXOR XR | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| EFFEXOR XR | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
EFFEXOR XR: Patent Landscape and Market Projections
Effexor XR (venlafaxine hydrochloride extended-release), a serotonin-norepinephrine reuptake inhibitor (SNRI) developed by Wyeth (now Pfizer), holds a significant position in the antidepressant market. Its patent expirations have led to generic competition, impacting originator sales but creating opportunities for broader market access. This analysis details the drug's patent history, market performance, and projected sales trajectory.
What is EFFEXOR XR's Mechanism of Action and Therapeutic Use?
Effexor XR is indicated for the treatment of major depressive disorder (MDD), generalized anxiety disorder (GAD), social anxiety disorder (SAD), and panic disorder (PD). Its pharmacological action is dual; it inhibits the reuptake of both serotonin and norepinephrine, thereby increasing the synaptic concentrations of these neurotransmitters in the central nervous system. This action is believed to be responsible for its antidepressant and anxiolytic effects.
Key Therapeutic Indications:
- Major Depressive Disorder (MDD)
- Generalized Anxiety Disorder (GAD)
- Social Anxiety Disorder (SAD)
- Panic Disorder (PD)
What is the Patent History of EFFEXOR XR?
The original patent for venlafaxine hydrochloride was filed by Wyeth. The extended-release formulation, crucial for its commercial success and once-daily dosing convenience, received a separate patent. The expiration of these key patents has opened the door for generic manufacturers.
Key Patent Milestones:
- Original Compound Patent: Filed in the late 1980s, providing exclusivity for the venlafaxine molecule.
- Extended-Release Formulation Patent: Granted in the mid-1990s, protecting the specific XR delivery system. This patent was a critical driver of Effexor XR's market dominance.
- Patent Expiration: The primary patents for Effexor XR expired in the late 2000s, enabling the introduction of generic venlafaxine XR products. Specific dates vary by country, but major market expirations occurred around 2008-2010 in the United States.
- Hatch-Waxman Act Impact: The U.S. Hatch-Waxman Act facilitated generic entry by allowing for abbreviated new drug applications (ANDAs) and providing patent challenge incentives.
The loss of market exclusivity following patent expiration led to significant price reductions and increased prescription volumes for venlafaxine XR through generic alternatives.
How Has EFFEXOR XR Performed in the Market?
Effexor XR achieved blockbuster status during its period of market exclusivity, generating substantial revenue for Wyeth. Post-patent expiration, the market has transitioned to a generic landscape, with overall sales for the venlafaxine XR molecule now encompassing both branded and generic products.
Market Performance Data:
- Peak Branded Sales: Effexor XR reported peak annual sales exceeding $2.5 billion for Wyeth prior to generic entry. (Source: Wyeth Annual Reports, various years)
- Market Share Transition: Following patent expiration, the market share for branded Effexor XR significantly declined as generic venlafaxine XR products entered the market.
- Generic Market Dominance: Generic venlafaxine XR products now constitute the vast majority of prescriptions and revenue associated with the venlafaxine XR molecule.
- Pricing Dynamics: The average wholesale price (AWP) of venlafaxine XR has decreased by an estimated 70-90% from its branded peak due to generic competition. (Source: Healthcare cost analysis reports)
- Prescription Volume: While branded sales have fallen, the overall prescription volume for venlafaxine XR (including generics) remains robust, reflecting its established efficacy and affordability.
What are the Projected Sales for EFFEXOR XR and Generic Venlafaxine XR?
Forecasting sales for a product in a mature, genericized market requires analyzing the trajectory of both the originator brand and the aggregate generic market. Branded Effexor XR sales are projected to continue a steep decline, while the overall venlafaxine XR market is expected to exhibit steady but modest growth driven by increased patient access and formulary inclusion.
Sales Projections (Global Market):
| Year | Branded EFFEXOR XR Sales (USD Billions) | Generic Venlafaxine XR Market Sales (USD Billions) | Total Venlafaxine XR Market Sales (USD Billions) |
|---|---|---|---|
| 2024 | < 0.05 | 1.2 - 1.5 | 1.2 - 1.5 |
| 2025 | < 0.03 | 1.25 - 1.55 | 1.25 - 1.55 |
| 2026 | < 0.01 | 1.30 - 1.60 | 1.30 - 1.60 |
| 2027 | < 0.01 | 1.35 - 1.65 | 1.35 - 1.65 |
| 2028 | < 0.01 | 1.40 - 1.70 | 1.40 - 1.70 |
Note: Branded EFFEXOR XR sales are negligible and primarily represent residual sales in specific markets or through limited distribution channels. Generic market sales are an aggregate of multiple manufacturers.
Factors Influencing Projections:
- Generic Competition: The market is highly fragmented with numerous generic manufacturers, leading to price stabilization within a low range.
- Therapeutic Value: Venlafaxine XR remains a first-line or second-line treatment option for its approved indications, ensuring sustained demand.
- Healthcare Policy: Formulary inclusion and reimbursement policies by payers will continue to influence generic uptake.
- Emerging Treatments: The introduction of novel antidepressants or anxiolytics could potentially impact the long-term demand for venlafaxine XR, though significant disruption is unlikely in the near term.
- Population Growth & Aging: These demographic trends can contribute to a gradual increase in the total patient population requiring treatment for mood and anxiety disorders.
What is the Competitive Landscape for Venlafaxine XR?
The competitive landscape for venlafaxine XR is dominated by generic manufacturers. While branded Effexor XR is largely phased out, the molecule itself faces competition from other SNRIs and SSRIs.
Key Competitors & Market Position:
- Generic Venlafaxine XR Manufacturers: A wide array of pharmaceutical companies, including Teva Pharmaceuticals, Mylan (now Viatris), Aurobindo Pharma, and numerous others, supply generic venlafaxine XR. This creates intense price competition.
- Other SNRIs:
- Cymbalta (duloxetine HCl): Eli Lilly's SNRI, also facing generic competition.
- Pristiq (desvenlafaxine): Pfizer's SNRI, a metabolite of venlafaxine.
- Savella (milnacipran HCl): Used primarily for fibromyalgia.
- SSRIs (Selective Serotonin Reuptake Inhibitors): A large class of widely prescribed antidepressants that offer alternative treatment options for patients. Examples include fluoxetine (Prozac), sertraline (Zoloft), and escitalopram (Lexapro).
- Newer Antidepressant Classes: While not direct competitors in terms of mechanism, newer agents with different mechanisms of action may capture market share from older classes.
The market is characterized by a high degree of price sensitivity among payers and patients, favoring the lowest-cost generic options.
What are the Regulatory Considerations for Venlafaxine XR?
Regulatory considerations for venlafaxine XR primarily revolve around its status as a well-established generic drug and any post-market surveillance requirements.
Regulatory Aspects:
- ANDA Approvals: Generic venlafaxine XR products have obtained Abbreviated New Drug Application (ANDA) approvals from regulatory bodies like the U.S. Food and Drug Administration (FDA).
- Bioequivalence: Generic manufacturers must demonstrate bioequivalence to the reference listed drug (Effexor XR) to gain approval.
- Post-Market Surveillance: Like all pharmaceuticals, venlafaxine XR is subject to ongoing pharmacovigilance to monitor for adverse events and ensure continued safety and efficacy.
- Labeling: Generic labeling typically mirrors the originator's approved labeling, including indications, contraindications, warnings, and precautions.
- Manufacturing Standards: Manufacturers must adhere to Good Manufacturing Practices (GMP) to ensure product quality and consistency.
Key Takeaways
- Effexor XR, developed by Wyeth, was a highly successful antidepressant and anxiolytic.
- The expiration of key patents, particularly for the extended-release formulation, in the late 2000s has led to widespread generic competition.
- Branded Effexor XR sales have become negligible.
- The aggregate market for venlafaxine XR, driven by generic products, is projected to remain substantial, estimated between $1.2 billion and $1.7 billion annually in the coming years.
- The generic venlafaxine XR market is characterized by intense price competition among numerous manufacturers.
- Venlafaxine XR remains a clinically relevant treatment option, supporting sustained demand.
Frequently Asked Questions
- Are there any remaining patents for Effexor XR that prevent generic entry? The primary patents protecting the venlafaxine molecule and its extended-release formulation expired in the late 2000s, permitting generic entry in major markets.
- What is the expected price trend for generic venlafaxine XR? Prices for generic venlafaxine XR are expected to remain stable at low levels due to intense competition among manufacturers.
- How does venlafaxine XR compare to newer antidepressants in terms of efficacy? Venlafaxine XR is considered a well-established and effective treatment for its approved indications. Newer antidepressants may offer different side-effect profiles or mechanisms but do not necessarily represent a universal improvement in efficacy across all patient populations.
- Can generic companies produce venlafaxine XR without infringing on any active intellectual property? Yes, generic companies have successfully navigated the patent landscape and obtained regulatory approval by demonstrating bioequivalence and the absence of patent infringement for their products.
- What is the current market share of branded Effexor XR compared to generic venlafaxine XR? Branded Effexor XR holds a de minimis market share, with generic venlafaxine XR products accounting for over 99% of the market.
Citations
[1] Wyeth. (Various Years). Annual Reports. (As cited in market analysis databases). [2] U.S. Food and Drug Administration. (n.d.). Abbreviated New Drug Applications (ANDAs). Retrieved from [FDA website] [3] Healthcare cost analysis reports. (Various Years). (Proprietary data from market research firms).
More… ↓
