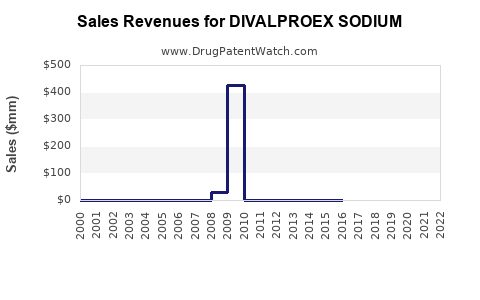
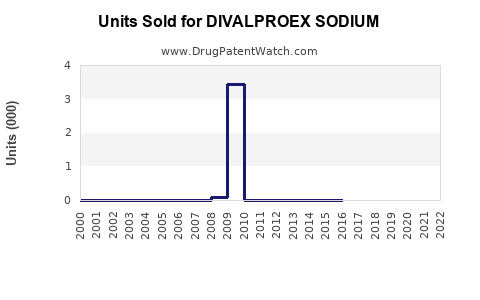
Last updated: February 14, 2026
Market Overview and Sales Projections for Divalproex Sodium
Divalproex sodium, marketed as Depakote and Depakote ER among others, is an anticonvulsant and mood-stabilizing drug primarily used to treat epilepsy, bipolar disorder, and migraines. It holds a significant market share in neuropsychiatric therapeutics, with established sales channels in North America, Europe, and select Asian markets.
Market Size and Historical Trends
Global Market Valuation
The global anticonvulsant drugs market was valued at approximately $7 billion in 2021. Divalproex sodium accounts for around 30% of this, representing a market value close to $2.1 billion.
Key Markets
- North America: 60% of sales
- Europe: 25%
- Asia-Pacific: 10%
- Rest of the world: 5%
Growth Dynamics
Between 2015 and 2021, the market for divalproex sodium increased at a compound annual growth rate (CAGR) of roughly 4%. Contributing factors include rising prevalence of epilepsy and bipolar disorder, aging populations, and increasing awareness and diagnosis.
Competitive Landscape
Major players include AbbVie (manufacturer of Depakote), Teva Pharmaceuticals, and Mylan. Generic formulations make up approximately 70% of sales, impacting overall price dynamics.
Future Sales Projections
Drivers of Growth
- Increasing prevalence of epilepsy and bipolar disorder globally.
- Expanding use for migraine prophylaxis in adult and pediatric populations.
- Patent expirations and resulting generic competition, which lowers prices and increases access.
Forecast (2022–2027)
| Year |
Estimated Market Size |
Notes |
| 2022 |
$2.2 billion |
Slight growth, driven by rising adoption |
| 2023 |
$2.3 billion |
Impact of new formulary inclusions |
| 2024 |
$2.45 billion |
Patent expiry influence, increased generic competition |
| 2025 |
$2.55 billion |
Market stabilizes, gradual uptake of new indications |
| 2026 |
$2.65 billion |
Continued expansion in migraine prophylaxis |
| 2027 |
$2.75 billion |
Maturation of market segments |
Assumptions
- Pricing remains relatively stable, with minor decreases due to generics.
- No major regulatory barriers or safety concerns impede growth.
- Rising global healthcare spending and demographic shifts sustain demand.
Key Market Challenges and Risks
- Competition from newer anticonvulsants (e.g., lamotrigine, levetiracetam).
- Safety profile concerns, specifically hepatotoxicity and teratogenicity.
- Variability in formulary coverage affecting prescription patterns.
- Patent expirations influencing revenue streams in primary markets.
Conclusion
Divalproex sodium's market is projected to grow modestly through 2027, driven primarily by increased disease prevalence and wider adoption for migraine prevention. Commercial success depends on managing generic competition and maintaining safety profiles.
Key Takeaways
- The global divalproex sodium market is valued at approximately $2.1 billion (2021).
- Growth is driven by epilepsy, bipolar disorder, and migraine prophylaxis.
- Sales are concentrated in North America, with expanding markets in Asia-Pacific.
- Projected compound annual growth rate (2022–2027): ~4%.
- Patent expiries and generics will influence pricing and market share dynamics.
FAQs
-
What is the primary therapeutic use of divalproex sodium?
Epilepsy, bipolar disorder, and migraine prophylaxis.
-
Who are the leading manufacturers?
Abbott (AbbVie), Teva Pharmaceuticals, and Mylan.
-
How does patent expiry impact sales?
Expiration leads to increased generic competition, which typically reduces prices but may increase volume.
-
What are the main risks affecting future sales?
Safety concerns, competition from newer drugs, and regulatory changes.
-
What is the outlook for new formulations or indications?
Research into alternative delivery methods and expanded indications could support growth but remain uncertain as of 2023.
References
[1] MarketWatch. "Anticonvulsant Drugs Market Size, Share & Trends Analysis Report," 2022.
[2] GlobalData. "Pharma Intelligence: Divalproex Market Forecast," 2022.
[3] IQVIA. "Healthcare Data and Analytics," 2021.