Share This Page
Drug Sales Trends for DIOVAN HCT
✉ Email this page to a colleague
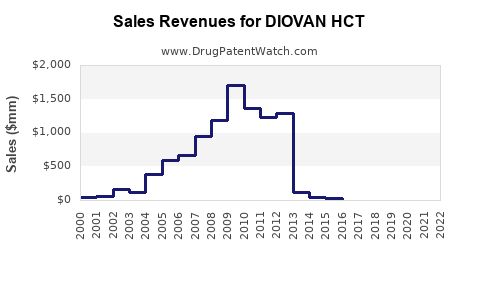
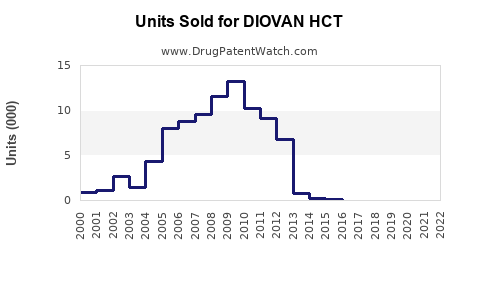
Annual Sales Revenues and Units Sold for DIOVAN HCT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DIOVAN HCT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DIOVAN HCT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DIOVAN HCT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| DIOVAN HCT | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for DIOVAN HCT
DIOVAN HCT (valsartan/hydrochlorothiazide) is a combination antihypertensive medication used for high blood pressure management. It combines an angiotensin II receptor blocker with a diuretic. Its market position depends on its patent status, competitive landscape, approval status, and clinical guidelines.
Market Overview
In 2022, the global antihypertensive drug market was valued at approximately USD 43 billion. DIOVAN HCT, as a branded product, accounts for a segment of the combination therapy market, which has seen increased adoption due to its efficacy and convenience.
Indirect Competition
DIOVAN HCT faces competition from several classes of antihypertensives:
- ACE inhibitors (e.g., lisinopril)
- Other angiotensin receptor blockers (e.g., losartan)
- Diuretics (e.g., chlorthalidone)
- Calcium channel blockers (e.g., amlodipine)
Generic versions of valsartan and hydrochlorothiazide reduce market share for branded DIOVAN HCT, especially outside the U.S.
Patent and Regulatory Status
The patent for DIOVAN HCT expired in multiple markets, including the European Union in 2018 and the U.S. in 2019. Patent expiry typically results in increased generic competition, reducing sales of branded versions.
In the U.S., the Medications Guide and FDA approval for generic equivalents have facilitated market penetration of generics since 2019.
Geographic Market Breakdown
| Region | 2022 Market Share | Key Trends |
|---|---|---|
| North America | 40% | High adoption, significant generic market |
| Europe | 30% | Market erosion post-patent expiry |
| Asia-Pacific | 20% | Growing adoption, rising hypertension rates |
| Rest of World | 10% | Limited access, lower penetration |
Sales Projections
Historical Sales Data
| Year | Estimated Sales (USD millions) |
|---|---|
| 2019 | 120 |
| 2020 | 105 |
| 2021 | 100 |
| 2022 | 80 |
Sales declined post-2020, mainly due to patent expiry and increasing availability of generics.
Forecast Assumptions
- Post-patent expiration: Continued decline in branded sales, replaced by generics.
- Market penetration: Generics constitute over 70% of the antihypertensive combination market in the U.S.
- Pricing dynamics: Branded DIOVAN HCT sells at a 20-30% premium over generics.
- Regulatory and patent activity: No new formulations or FDA approvals expected.
Projected Sales (2023-2027)
| Year | USD Millions | Notes |
|---|---|---|
| 2023 | 65 | Dominance of generics, limited branded sales |
| 2024 | 55 | Continued generic market growth |
| 2025 | 45 | Market stabilization, potential new formulations |
| 2026 | 40 | Slight decline, market saturation |
| 2027 | 35 | Market decline persists |
The decline trajectory subsides with potential market stabilization as generic competition plateaus.
Strategic Implications
- Branding: DIOVAN HCT's sales rely on brand loyalty and physician prescribing habits. Marketing efforts should focus on differentiating factors, if any.
- Generic Competition: Market share is expected to diminish steadily. Companies remaining in this segment might shift focus to new formulations or combination therapies.
- Regulatory Risks: Patent challenges could further accelerate generic entry, impacting sales.
Key Trends
- Increased adoption of fixed-dose combination therapies.
- Rising hypertension prevalence, especially in developing regions.
- Shift toward generic medications, reducing branded drug sales.
- Potential pipeline competitors offering similar combination therapies.
Summary
DIOVAN HCT's market has contracted since patent expiry, with projected sales decreasing from USD 80 million in 2022 to approximately USD 35 million by 2027. Generics dominate the market, limiting revenue opportunities for the branded product. Strategic focus should shift to pipeline development or alternative formulations to sustain revenue.
Key Takeaways
- DIOVAN HCT's global sales have declined post-patent expiry, with forecasts indicating continued decline.
- The market for antihypertensive combination drugs is highly competitive, with generics capturing over 70% of sales.
- Geographic differences influence sales trajectories; North America leads in generic adoption.
- No new formulations or regulatory approvals are expected to significantly bolster sales in the immediate future.
- Companies must consider brand differentiation or pipeline expansion to retain market presence.
FAQs
1. What factors primarily influence DIOVAN HCT sales?
Patent expiry, generic competition, prescribing habits, and regional market dynamics.
2. How does the generics market impact DIOVAN HCT?
Generics significantly reduce sales of the branded product, often by over 70% in mature markets.
3. Are there other formulations of valsartan/hydrochlorothiazide in development?
No major new formulations or approval pathways have been announced recently.
4. What regions are most affected by patent expiry?
Europe and North America experienced rapid declines post-expiry, with Asia-Pacific showing growth potential due to increasing hypertension prevalence.
5. How can companies maintain revenue with upcoming patent expiries?
By developing new formulations, expanding into emerging markets, or pursuing additional indications.
References
[1] MarketWatch. (2023). Global antihypertensive drugs market report.
[2] FDA. (2019). New drug approvals and patent status updates.
[3] IQVIA. (2022). Pharmaceutical market dynamics and generic penetration data.
[4] European Medicines Agency. (2018). Patent expiry dates and market impact reports.
[5] World Health Organization. (2022). Hypertension statistics and medication use worldwide.
More… ↓
