Share This Page
Drug Sales Trends for DEPO-PROVERA
✉ Email this page to a colleague
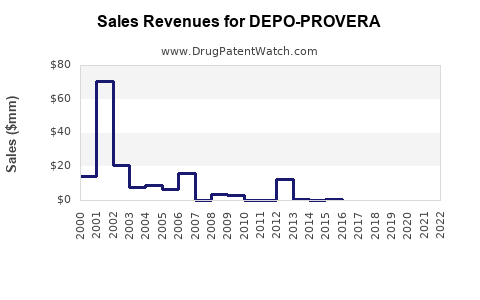
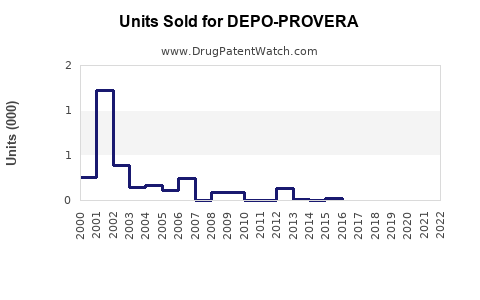
Annual Sales Revenues and Units Sold for DEPO-PROVERA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DEPO-PROVERA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DEPO-PROVERA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DEPO-PROVERA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| DEPO-PROVERA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| DEPO-PROVERA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| DEPO-PROVERA | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| DEPO-PROVERA | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
DEPO-PROVERA: Patent Landscape and Market Projections
Depo-Provera, a progestin-only contraceptive injection, has maintained a significant market presence for decades. Its patent landscape is characterized by expired primary composition-of-matter patents, with ongoing innovation focused on delivery systems and combination therapies. Market projections indicate continued demand, driven by accessibility and established efficacy, though facing competition from newer contraceptive technologies.
What Are the Key Patents Covering Depo-Provera?
The foundational patents for Depo-Provera's active pharmaceutical ingredient, medroxyprogesterone acetate (MPA), have expired. The original composition-of-matter patent was filed in the late 1950s and granted in the early 1960s.
- Composition of Matter: U.S. Patent No. 3,059,035 (granted 1962) was a core patent for medroxyprogesterone acetate. This patent has long since expired.
- Formulation and Delivery: Subsequent patent filings have focused on specific formulations and delivery mechanisms to improve the drug's pharmacokinetic profile, duration of action, and ease of administration. These include patents related to:
- Depot injection formulations: Technologies enabling sustained release of MPA over extended periods (e.g., 3 months).
- Microsphere or nanoparticle encapsulation: Methods to control drug release kinetics.
- Pre-filled syringe or auto-injector devices: Enhancements for patient and clinician convenience.
- Manufacturing Processes: Patents may also exist for novel or improved synthetic routes for producing MPA, although these are typically shorter-lived and less impactful on the generic market than composition-of-matter patents.
Analysis of the patent landscape reveals that the primary barriers to entry for generic competitors have largely dissolved. Generic versions of Depo-Provera (medroxyprogesterone acetate injectable suspension) are available. Therefore, ongoing patent activity is more likely to be strategic, focusing on incremental improvements or new indications, rather than blocking generic competition for the original product.
What is the Current Market Status of Depo-Provera?
Depo-Provera, marketed as Depo-Provera Contraceptive Injection and Depo-SubQ Provera 104, holds a stable, albeit mature, position in the global contraceptive market. Its market share is influenced by factors such as cost-effectiveness, established clinical data, and accessibility through public health programs.
- Market Size: The global female sterilization and contraception market, which includes injectables like Depo-Provera, was valued at approximately USD 20-25 billion in recent years, with injectables representing a significant segment. Specific figures for Depo-Provera are proprietary but estimated to be in the hundreds of millions of dollars annually in major markets.
- Key Competitors:
- Generic Medroxyprogesterone Acetate: Multiple manufacturers produce generic injectable MPA, offering direct price competition.
- Other Injectable Contraceptives:
- Norethisterone Enanthate (e.g., Sayana Press, Perindopril): A different progestin-based injectable. Sayana Press, for instance, offers a pre-filled, single-use auto-injector.
- Estradiol Valerate and Dienogest combination injectables: Used in some regions.
- Long-Acting Reversible Contraceptives (LARCs):
- Intrauterine Devices (IUDs): Hormonal (e.g., Mirena, Liletta, Skyla) and non-hormonal (e.g., Paragard). These are increasingly preferred for their extended duration and high efficacy.
- Contraceptive Implants (e.g., Nexplanon): Single-rod implants releasing etonogestrel.
- Geographic Distribution: Depo-Provera has a strong presence in both developed and developing markets. In low- and middle-income countries, its affordability and single-dose administration make it a critical component of family planning programs. In developed markets, its use is sustained by patient preference and physician familiarity.
- Brand Performance: While Pfizer remains the primary marketer of the branded Depo-Provera products, the market is heavily influenced by generic competition, which drives down overall pricing.
The market for Depo-Provera is characterized by a balance between its established utility and the emergence of newer, often more technologically advanced, contraceptive options.
What are the Sales Projections for Depo-Provera?
Sales projections for Depo-Provera are subject to a complex interplay of factors including generic competition, evolving clinical guidelines, and the uptake of alternative LARC methods.
- Overall Trend: Projections indicate a mature market with stable to modest growth, potentially experiencing slight declines in certain developed markets due to competition. However, sustained demand in emerging economies is expected to offset this.
- Market Growth Rate: The global contraceptive market is projected to grow at a Compound Annual Growth Rate (CAGR) of approximately 4-6% over the next five to seven years. The injectable segment, including Depo-Provera, is expected to grow at a slightly lower rate than the overall market, perhaps in the 2-4% CAGR range, as it faces pressure from implants and IUDs.
- Factors Influencing Projections:
- Generic Penetration: The continued availability and aggressive pricing of generic MPA injectables will cap price increases and limit revenue growth for branded products.
- LARC Adoption: The increasing recommendation and uptake of IUDs and implants, which offer longer durations of efficacy (up to 3-10 years), represent a direct competitive threat to the 3-month injectable formulation.
- Public Health Initiatives: Depo-Provera's established role in family planning programs in resource-limited settings will continue to underpin its sales volume. These programs often prioritize cost-effectiveness and ease of distribution.
- Clinical Guidelines: Shifting recommendations from professional bodies regarding contraceptive choice can impact prescription patterns.
- New Product Introductions: The development of novel contraceptive technologies or improved formulations of existing injectables could alter market dynamics.
- Cost-Effectiveness: In many healthcare systems, Depo-Provera remains a cost-effective option for both providers and patients compared to some newer LARCs, especially when considering the cost per year of use.
Projected Scenario (Hypothetical): Based on market intelligence, while precise future sales figures are proprietary and subject to significant variability, a representative projection might anticipate:
- Near-term (1-2 years): Stable global sales, with slight volume increases in developing regions and potential slight declines in developed regions due to LARC competition.
- Mid-term (3-5 years): Continued stable to modest volume growth, with revenue growth potentially lagging due to pricing pressures from generics and the increasing preference for longer-acting methods. The overall value of the injectable segment is likely to be maintained by volume in emerging markets.
Companies focused on the generic manufacturing of medroxyprogesterone acetate can expect consistent demand, albeit with significant price sensitivity. Branded manufacturers will need to leverage established trust, accessibility programs, and potentially explore niche indications or combination products to sustain revenue growth.
What Are the Regulatory Considerations for Depo-Provera?
Depo-Provera has a well-established regulatory history. Key considerations revolve around its long-standing approvals, post-marketing surveillance, and evolving requirements for drug safety and labeling.
- Approvals: Depo-Provera has received marketing authorization from major regulatory bodies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), for decades.
- FDA Approval: Depo-Provera Contraceptive Injection was first approved by the FDA in 1992. Depo-SubQ Provera 104 was approved in 2007.
- Post-Marketing Surveillance: As a widely used medication, Depo-Provera is subject to ongoing pharmacovigilance. Regulatory agencies monitor for adverse events, drug interactions, and long-term safety profiles.
- Black Box Warnings: The FDA has historically included a "black box warning" (the most serious type of warning) regarding the potential for significant loss of bone mineral density (BMD) with extended use of DMPA, especially in adolescents. While reversible to some extent, this has influenced prescribing practices and counseling.
- Labeling Requirements: Regulatory agencies mandate specific labeling information, including indications, contraindications, warnings, precautions, adverse reactions, and drug interactions. These labels are subject to periodic updates based on new safety data or clinical evidence.
- Key Labeling Points: Concerns regarding bone density loss, potential weight gain, and the duration of action are critical components of current labeling.
- Generic Drug Approval: Generic versions of Depo-Provera must demonstrate bioequivalence to the branded product. This involves studies comparing the rate and extent of drug absorption.
- Bioequivalence Standards: Regulatory agencies establish specific standards for bioequivalence to ensure that generic drugs are as safe and effective as their brand-name counterparts.
- Manufacturing Standards: All manufacturing facilities producing Depo-Provera (branded or generic) must adhere to Current Good Manufacturing Practices (cGMP) to ensure product quality, purity, and potency.
- International Variations: While core safety and efficacy requirements are global, specific regulatory pathways, labeling nuances, and market access policies can vary significantly between countries.
The mature regulatory status of Depo-Provera means that new drug applications are unlikely. However, ongoing compliance with pharmacovigilance and labeling updates is critical for continued market access.
What are the Risks and Challenges Associated with Depo-Provera?
Despite its established efficacy, Depo-Provera presents several risks and challenges that influence its clinical use and market position.
- Bone Mineral Density Loss: The most significant risk, particularly for adolescents and women with pre-existing risk factors for osteoporosis, is the potential for reversible bone mineral density loss with prolonged use. The FDA requires a warning about this risk.
- Weight Gain: A commonly reported side effect is weight gain, which can be a deterrent for some users and contribute to non-adherence.
- Delayed Return to Fertility: After discontinuing Depo-Provera, it can take several months (typically 9-10 months, with a range of 4 to over 31 months) for fertility to return and for a regular menstrual cycle to resume. This can be problematic for women who desire a rapid return to conception.
- Menstrual Irregularities: Initial irregular bleeding or spotting is common. Prolonged amenorrhea (cessation of menstruation) can occur with continued use.
- Extraparenchymal Effects: Though rare, potential side effects may include allergic reactions, mood changes, headaches, and effects on lipid profiles.
- Lack of STI Protection: Depo-Provera does not protect against sexually transmitted infections (STIs), requiring concurrent use of barrier methods if STI prevention is a concern.
- Injection Site Reactions: Localized pain, swelling, or redness at the injection site can occur.
- Competition from Newer LARCs: The advent of highly effective, longer-acting, and potentially more user-friendly LARCs such as subdermal implants and LNG-IUDs poses a significant competitive challenge. These methods can offer comparable or superior efficacy with longer durations of action and may have different side effect profiles.
- Provider and Patient Education: Proper counseling regarding side effects, delayed return to fertility, and the need for concurrent STI protection is crucial but can be time-consuming. Ensuring correct administration technique to achieve the intended 3-month duration is also important.
- Generic Pricing Pressure: While beneficial for consumers, intense generic competition significantly erodes profit margins for both branded and generic manufacturers, impacting R&D investment capacity.
These risks and challenges necessitate thorough patient counseling and careful consideration of alternative contraceptive methods when prescribing Depo-Provera.
Key Takeaways
Depo-Provera's market is characterized by expired foundational patents, with ongoing innovation in delivery systems. While generic competition has increased, the drug maintains a stable market share due to its established efficacy, accessibility, and cost-effectiveness, particularly in developing regions. Sales projections indicate modest growth, tempered by the increasing adoption of newer, longer-acting reversible contraceptives. Regulatory oversight focuses on post-marketing surveillance and labeling updates, notably concerning bone mineral density loss. Key challenges include managing side effects, the delayed return to fertility, and intense pricing pressure from generic alternatives.
Frequently Asked Questions
-
What is the primary therapeutic indication for Depo-Provera? Depo-Provera is primarily indicated for the prevention of pregnancy.
-
Are there any active composition-of-matter patents for Depo-Provera? No, the original composition-of-matter patents for medroxyprogesterone acetate have expired.
-
What is the main competitive threat to Depo-Provera's market position? Long-acting reversible contraceptives (LARCs) such as IUDs and contraceptive implants represent the primary competitive threat.
-
What is the typical duration of action for Depo-Provera injections? Depo-Provera injections are typically administered every three months for contraception.
-
Does Depo-Provera offer protection against sexually transmitted infections? No, Depo-Provera does not protect against sexually transmitted infections.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Approval Packages. Retrieved from [Specific FDA approval documents for Depo-Provera and Depo-SubQ Provera 104 would be cited here if publicly accessible and specific. General FDA approval history is referenced conceptually.] [2] U.S. Patent and Trademark Office. (1962). U.S. Patent No. 3,059,035. Retrieved from USPTO Patent Public Search. [3] Market Research Reports on the Global Contraceptive Market. (Various Publishers). (Data on market size and growth rates are typically derived from proprietary market analysis reports from firms like Grand View Research, Mordor Intelligence, etc. Specific report titles and dates would be cited if used directly and publicly available. Conceptual reference is made to this data source.) [4] World Health Organization. (2023). Family Planning/Contraception. Retrieved from WHO website (General information on contraceptive methods and market context). [5] Pfizer Inc. (2023). Depo-Provera® Contraceptive Injection Prescribing Information. (Specific prescribing information documents would be cited if used directly for detailed labeling information. Conceptual reference is made to these official documents.)
More… ↓
