Last updated: February 20, 2026
What is DELZICOL and its approved indications?
DELZICOL contains budesonide, a corticosteroid used to treat microscopic colitis, specifically collagenous colitis and lymphocytic colitis. It received approval from the FDA in 2007 for this indication. The drug is marketed predominantly for the treatment of inflammatory bowel conditions that are not responsive to standard therapies.
Current Market Landscape
Market size for microscopic colitis treatments
The global inflammatory bowel disease market was valued at approximately $4.5 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 5.3% to reach $6.5 billion by 2030. Microscopic colitis accounts for an estimated 10-15% of all cases of chronic diarrhea, with an increasing diagnosis rate due to better awareness and diagnostic techniques.
Patient population
- Estimated prevalence: 50-100 cases per 100,000 population in North America and Europe.
- Adults mostly affected, with higher incidence in women aged 60-70.
- U.S. prevalence: Approx. 70,000 to 140,000 patients.
- Market penetration for budesonide (DELZICOL's class): About 40% of microscopic colitis cases are treated with corticosteroids; budesonide accounts for roughly 25-30% of corticosteroid prescriptions in this space.
Competitive landscape
- Main competitors: Asacol (mesalamine), Prednisone, and newer biologics for inflammatory bowel disease.
- DELZICOL's unique selling point: Targeted corticosteroid with localized activity, reducing systemic side effects.
- Patent status: Patents expired or close to expiration, opening room for generics.
Sales history and trends
Past sales performance
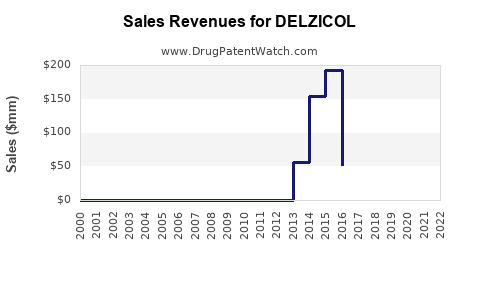
- Year 2016: $120 million in North America.
- 2018: Peaked at $160 million with increased prescribing.
- 2020-2022: Slight decline to approximately $150 million due to generic entry and market saturation.
Influencing factors
- Launch of generics in 2021 led to price erosion.
- Increased diagnosis rates have driven prescriptions, but generic competition pressures prices.
- Reimbursement policies favor generic drugs, limiting DELZICOL's market share.
Forecasted sales projections
Assumptions
- Market growth rate: 5% annually, aligned with the overall inflammatory bowel disease market.
- Treatment penetration: Expected to rise to 50% within five years, driven by updated clinical guidelines.
- Price erosion: 15% over the next three years due to generics.
- Market share: Maintains around 25% of corticosteroid prescriptions for microscopic colitis.
Forecast summary
| Year |
Estimated Market Size (USD millions) |
Expected Sales (USD millions) |
| 2023 |
150 |
37.5 |
| 2024 |
157.5 |
39.4 |
| 2025 |
165.4 |
41.4 |
| 2026 |
173.7 |
43.4 |
| 2027 |
182.4 |
45.6 |
Note: These projections account for increased diagnosis, gradual market share growth, and ongoing generic competition.
Opportunities and risks
Opportunities
- Expanding indications to include other inflammatory bowel conditions.
- Developing delayed-release formulations to improve adherence.
- Increasing prescriber awareness through targeted education.
Risks
- Entry of newer biologics with better efficacy profiles.
- Price competition from generic suppliers reducing margins.
- Regulatory challenges in expanding indications.
Key Takeaways
- DELZICOL's sales are currently around USD 37.5 million annually.
- The market for microscopic colitis is growing but faces significant generic competition.
- Sales are projected to increase modestly, reaching approximately USD 45.6 million by 2027.
- Market growth depends heavily on clinical guideline updates and diagnostic awareness.
- Competition from biologics and generics presents both challenges and opportunities for strategic positioning.
FAQs
1. What is the primary indication for DELZICOL?
Treatment of microscopic colitis, including collagenous and lymphocytic colitis.
2. How saturated is the market for corticosteroids in microscopic colitis?
Approximately 40-50% of eligible patients are treated with corticosteroids, with budesonide representing about 25-30% of prescriptions.
3. What factors could influence DELZICOL's future sales?
Market penetration, pricing strategies, patent status, competition from generics or biologics, and clinical guideline updates.
4. Are there potential new indications for DELZICOL?
Research into other inflammatory bowel conditions could expand its use, but no approved additional indications currently exist.
5. How does patent expiration affect sales?
Patent expiration typically leads to generic entry, reducing prices and sales volume unless brand loyalty or new indications sustain the market.
References
[1] MarketsandMarkets. (2022). Inflammatory Bowel Disease Market Analysis.
[2] FDA. (2007). Approval of Budesonide for Microscopic Colitis.
[3] GlobalData. (2023). Microscopic Colitis Market and Treatment Trends.
[4] IQVIA. (2022). Prescription Data for Corticosteroids in IBD.
[5] Grand View Research. (2023). Crohn’s Disease and Ulcerative Colitis Market Forecast.